Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below : {:("Concentration/M", 0.001, 0.010, 0.020, 0.050, 0.100),(10^(2) xx k//S m^(-1), 1.237, 11.85, 23.15, 55.53, 106.74):} Calculate Lambda_(m) for all concentration and draw a plot between Lambda_(m) and C^(1//2). Find the values of Lambda_(m)^(@). |
Answer» Solution :`1 S cm^(-1) = 100 S m^(-1)`. 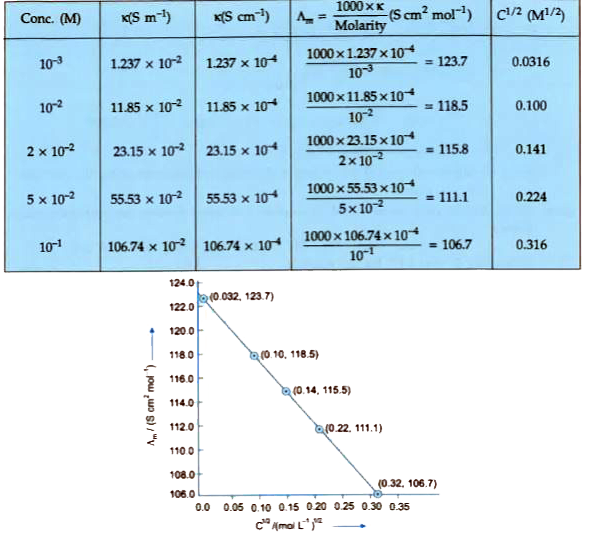 The graph is EXTRAPOLATED to MEET Y-axis. It meets the Y-axis at 124.05. `Lambda_(m)^(@)`= Intercept on Y-axis = 124.05 S `cm^(2) mol^(-1)` |
|
| 2. |
The conductivity of sodium chloride at 298K has been determined at different concentrations and the results are given below: Calculate Lamda_(m) for all concentration and draw a plot between Lamda_(m) and c^((1)/(2)). Find the value of Lamda_(m)^(@). |
Answer» Solution :* Here, the value of given concentration and concentration and conductivity and the values of `c^((1)/(2))` and `Lamda_(m)` re given in the following table.  * Plotting the graph, from the calculation of above table `c^((1)/(2))` is on X-axis and its respective value of `Lamda_(m)` on the Y-axis, we will get following graph.  * This graph having negative slopping linear line. Value of `Lamda_(m)^(0)" S "m^(2)MOL^(-1)`: Value of `c^((1)/(2))=0` which is the value of `Lamda_(m)^(0)` `k=1,237xx10^(-1)S " "m^(-1),c=0.001M` Then, `k=1.237xx10^(-4)" S "cm^(-1),c^((1)/(2))=0.0316M^((1)/(2))` `therefore Lamda_(m)=(k)/(c)=(1.237xx10^(-2)" S "cm^(-1))/(0.001" mol "L^(-1))XX(1000cm^(3))/(L)` `=123.7" S "cm^(2)mol^(-1)` Given, `k=11.85xx10^(-2)" S "m^(-1),c=0.010M` Then, `k=11.85xx10^(-4)" S "cm^(-1),c^((1)/(2))=0.01M^((1)/(2))` `=118.5" S "cm^(2)mol^(-1)` Given, `k=23.15xx10^(-2)" S "m^(-1),c=0.020M` Then, `k=23.15xx10^(-4)" S "cm^(-1),c^((1)/(2))=0.1414M^((1)/(2))` `therefore Lamda_(m)=(k)/(c)=(23.15xx10^(-4)" S "cm^(-1))/(0.020" mol "L^(-1))xx(1000cm^(3))/(L)` `=115.8" S "cm^(2)mol^(-1)` Given, `k=55.53xx10^(-2)" S "m^(-1),c=0.050M` Then, `k=55.53xx10^(-4)" S "cm^(-1),c^((1)/(2))=0.2236M^((1)/(2))` `therefore k=(k)/(c)=(55.53xx10^(-4)" S "cm^(-1))/(0.050" mol "L^(-1))xx(1000cm^(3))/(L)` `=111.1" S "cm^(2)mol^(-1)` Given, `k=106.74xx10^(-4)" S "m^(-1),c=0.3162M` `therefore Lamda_(m)=(k)/(c)=(106.74xx10^(-4)" S "cm^(-1))/(0.100" mol "L^(-1))xx(1000cm^(3))/(L)` `=106.74" S "cm^(2)mol^(-1)` Now, we have the following data:  Since the line interrupts `Lamda_(m)` at `124.0" S "cm^(2)` `mol^(-2),Lamda_(m)^(@)=124.5" S "cm^(2)mol^(-1)`. |
|
| 3. |
The conductivity of NaCl at 298K has been determined at different concentrations and the results are given below: {:("Concentration/M",0.001,0.010,0.020,0.050,0.100),(10^(2)xxkappa//S" "m^(-1),1.237,11.85,23.15,55.53,106.74):} Calculate wedge for all concentrations and draw a plot between wedge and c^(1//2). find the value of wedge^(8). |
|
Answer» Solution :1 S `cm^(-1)=100" S "m^(-1)` `(1" S "cm^(-1))/(100" S "cm^(-1))=1` (UNIT conversion FACTOR)  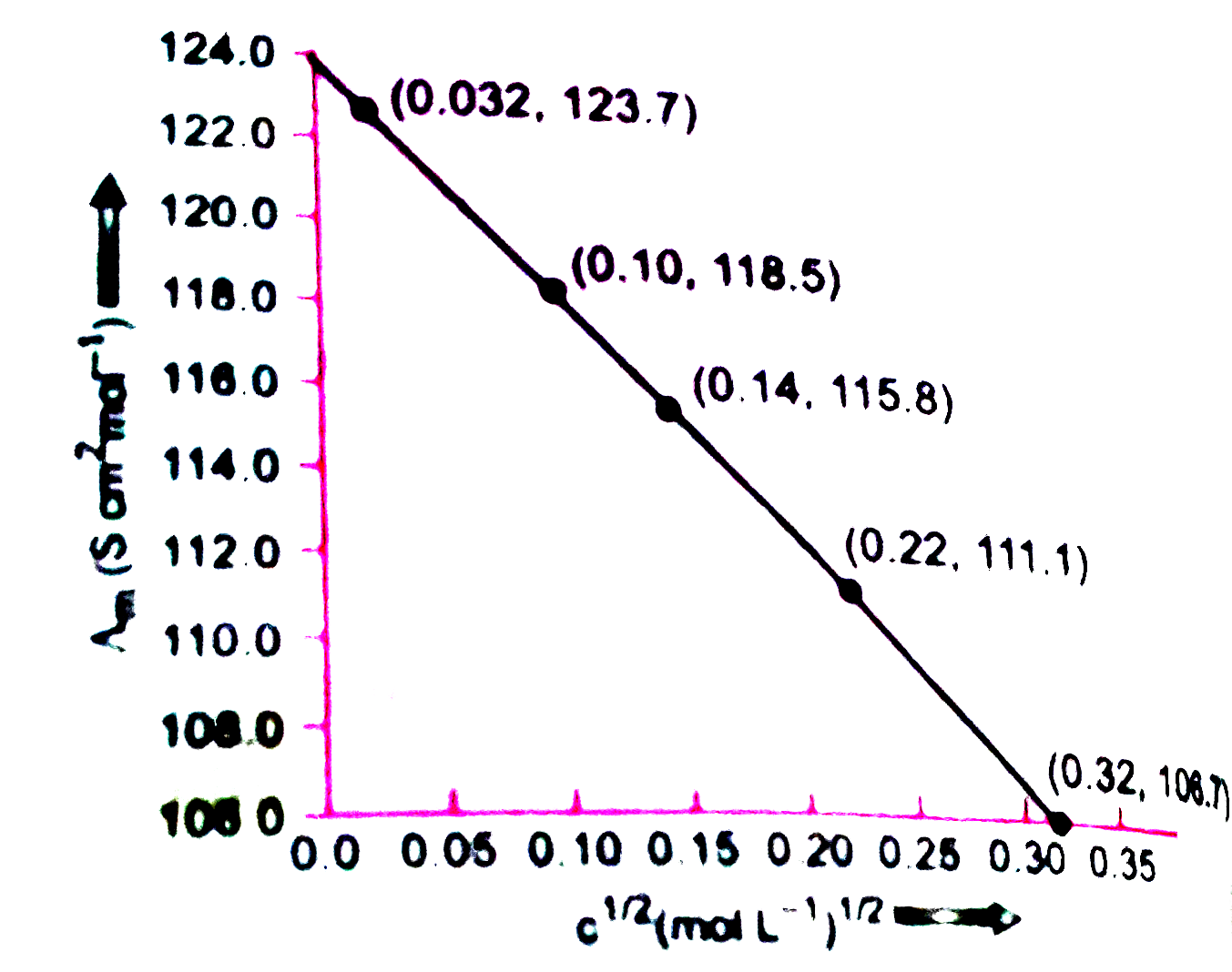 `(AA)^(@)=`intercept on the `(AA)_(m)` axis=124.0 S `cm^(2)mol^(-1)` (on extrapotation to zero CONCENTRATION). |
|
| 4. |
The conductivity of pure water in a conductivity cell with electrodes of cross sectional area 4 cm^2 and 2 cm apart is 8xx10^(-7)S cm^(-1) What current would flow through the cell under an applied potential difference of 1 volt ? |
|
Answer» |
|
| 5. |
The conductivity of pure water in a conductivity cell with electrodes of cross sectional area 4 cm^2 and 2 cm apart is 8xx10^(-7)S cm^(-1) What is resistance of conductivity cell ? |
|
Answer» |
|
| 6. |
The conductivity of saturated solution of AgCl is found to be 1.86 xx 10^(-6) ohm^(-1)cm^(-1) and that of water is 6xx 10^(-8) ohm^(-1)cm^(-1). If lamda^0 AgCl is |
|
Answer» `1.3 xx 10^(-5)` |
|
| 7. |
The conductivity of ____________ semiconductor is increased by adding an appropriate amount of suitable ____________________. |
| Answer» SOLUTION :IMPURITY | |
| 8. |
The conductivity of metals increases with: |
|
Answer» INCREASE in temperature |
|
| 9. |
The conductivity of intrinsic semiconductors can be increased by adding a suitable impurity. This process is called __(P)__. This can be done with an impurity which is __(Q)_ rich or deficient as compared to the semiconductor. Such impurities introduce __(R)__ defects in them. Electron rich impurities result in __(S)__ type semiconductors while electron deficit impurities result in __(T)___ type semiconductors . |
|
Answer» <P>P-doping , Q-proton , R-point , S- p , T-n |
|
| 10. |
The conductivity of a solution containing 1.0 g of anhydrous BaCl, in 200 cm of the solution has been found to 0.0058 S cm^(-1). Calculate the molar conductivity and equivalent conductivity of the solution. |
|
Answer» `Lambda_(m) = (0.0058 xx 1000)/(0.0240) = 241.67 OHM^(-1) cm^(2) MOL^(-1)` NORMALITY of solution = `(1//104 xx 1000)/(200) = 0.0480 N` `Lambda = (0.0058 xx 1000)/(0.0480) = 120.83 ohm^(-1) cm^(2) "EQUIV"^(-1)` |
|
| 11. |
The conductivity of a solution containing 1 gram of anhydrous BaCl_(2) in 200 cm^(3) of water has been found to be 0.0058 S cm^(-1). What are the molar conductivity and equivalent conductivity of the solution? (At. Wt. of Ba=137 and Cl=35.5) |
|
Answer» Solution :We are given: Conductivity `(kappa)=0.0058" S "cm^(-1)` Molar MASS of `BaCl_(2)=137+2xx35.5=208" G "mol^(-1)` As 1 gram of `BaCl_(2)` is present in 200 `cm^(3)` of the solution, therefore, molar concentration (C) `=(1)/(208)xx(1)/(200)xx1000" mol "L^(-1)=0.0240" mol "L^(-1)` `therefore`Molar conductivity, `wedge_(m)=(kappaxx1000)/(c_(m))=(0.0058" S "cm^(-1)xx1000cm^(3)L^(-1))/(0.0240" mol "L^(-1))=241.67" S "cm^(2)mol^(-1)` Further in case of `BaCl_(2)`, equivalent weight`=("Mol. wt")/(2)=(208)/(2)=104` `therefore` Concentration of the solution in gram equivalent per litre `(c_(EQ))=(1)/(104)xx(1)/(200)xx1000=0.0480` `therefore`Equivalent conductivity, `wedge_(eq)=(kappaxx1000)/(c_(eq))=(0.0058" S "cm^(-1)xx1000cm^(3)L^(-1))/(0.0480" g "eqL^(-1))=120.83" S "cm^(2)eq^(-1)`. |
|
| 12. |
The conductivity of a saturated solution of BaSO_(4) at 298 K , is found to be 3.758 xx 10^(-6) ohm^(-1) cm^(-1) and that of water used is 1.36 xx 10^(-6) ohm^(-1) cm^(-1) . Molar ionic conduntances at infinite dilution for Ba^(2+) and SO_(4)^(2-) ions are 110 and 136.6 ohm^(-1) cm^(2) mol^-1 respectively . Calculate the solubility of BaSO_(4) at 295 K (Atomic masses : Ba = 137 , S = 32 , O = 16) |
|
Answer» `= 3.758 xx 10^(-6) - 1.36 xx 10^(-6)` `= 2.398 xx 10^(-6) ohm^(-1) cm^(-1)` `Lambda_(m)^(@) = 110 + 136.6 = 246.6 ohm^(-1) cm^(2) mol^(-1)` Molar MASS = `137 + 32 + 4 xx 16 = 233` Solubility = `(kappa xx 1000)/(Lambda_(m))` `(2.398 xx 10^(-6) xx 1000)/(246.6)` `= 9.724 xx 10^(-6) mol L^(-1)` or `= 9.724 xx 10^(-6) xx 233 = 2.266 xx 10^(-3) G L^(-1)` |
|
| 13. |
The conductivity of a saturated solution of AgCl at 288 K is found to be 1.382xx10^(-6)" S "cm^(-1). Find its solubility. Given ionic conductances of Ag^(+) and Cl^(-) at infinite dilution are 61.9 S cm^(2)mol^(-1) and 76.3"S "cm^(2)mol^(-1). respectively |
|
Answer» SOLUTION :`wedge_(m)^(@)(AgCl)=lamda_(ag^(+))^(@)+lamda_(Cl^(-))^(@)=61.9+76.3=138.2" S "cm^(2)mol^(-1)` Solubility `=(kappaxx1000)/(wedge_(m)^(@))=(1.382xx10^(-6)xx1000)/(138.2)=10^(-5)" mol "L^(-1)=10^(-5)xx143.5" G "L^(-1)=1.435xx10^(-3)g" "L^(-1)`. |
|
| 14. |
The conductivity of a saturated solutior of a sparingly soluble salt MX_2 is found to be 4 xx 10^(-5) Omega^(-1) cm^(-1)." If "lambda_(m)^(oo) (1/2 M^(2+))=50 Omega^(-1) cm^(2) mol^(-1) and lambda^(oo) (X^(-))=50 Omega^(-1) cm^(2) mole^(-1), the solubility product of the salt is about |
|
Answer» `2 xx 10^(-10)M^(3)` |
|
| 15. |
The conductivity of a metal depends upon |
|
Answer» nature and structure of the metal. |
|
| 16. |
The conductivity of a 0.01 M solution of acetic acid at 298 K is 1.65 xx 10^(-4) S cm^(-1). Calculate the molar conductivity (Lambda_(m)) of the solution. |
|
Answer» |
|
| 17. |
The conductivity of 0.20M solution of KCl at 298K is 0.0248 S cm^(-1) . Calculate its molar conductivity. |
|
Answer» SOLUTION :`"MOLAR conductivity"=(kxx1000)/(M)=("0.0248 S cm"^(-1)XX"1000 cm"^(3)L^(-1))/("0.2 mol L"^(-1))` `="124.0 S cm"^(2)"mol"^(-1)` |
|
| 18. |
The conductivity of a 0.01 M solution of acetic acid at 298 K is 1.65 xx 10^(-4 )S cm^(-1).Calculate molar conductivity (Lambda_(m))of the solution. |
|
Answer» SOLUTION :Use the relation, `Lambda_(m) = (1000 k)/C`, where C stands for CONCENTRATION SUBSTITUTING the values, we get `Lambda_(m) = (1000 xx 1.65 xx 10^(-4))/0.01 = 16.5 S cm^(2) MOL^(-1)` |
|
| 19. |
The conductivity of a 0.01 M solution of a 1:1 weak electrolyte at 298 K is 1.5 times 10^(-4)" S " cm^(-1). i) molar conductivity of the solution ii) degree of dissociation and the dissociation constant of the weak electrolyte Given that lambda_("cation")^(@)=248.2" S "cm^(2)mol^(-1) lambda_("anion")^(@)=51.8" S "cm^(2)mol^(-1) |
|
Answer» SOLUTION :Given C = 0.01 M `""lambda_("cation")^(@)=248.2" S "cm^(2)mol^(-1)` `K=1.5 times 10^(-4)" S " cm^(-1) " "lambda_("anion")^(@)=51.8" S "cm^(2)mol^(-1)` (i) Molar conductivity `wedge_(m)^(@)=(""^(@)(SM^(-1)) times 10^(-3))/(C("in M"))mol^(-1)m^(3)` `" "^(@)=1.5 times 10^(-4)" S " cm^(-1)` `""=(1.5 times 10^(2) times 10^(-3))/(0.01)S" "mol^(-1)m^(2)` `1cm^(-1)=10^(2)m^(-1)` `""=1.5 times 10^(-3)" S "m^(2)mol^(-1)` `""=1.5 times 10^(2)` (ii) Degree of DISSOCIATION `alpha=WEDGE^(@)/wedge_(infty)^(@)` `wedge_(infty)^(@)=lambda_("cation")^(@)+lambda_("anion")^(@)` `" "=(248.2+51.8)" S "cm^(2)mol^(-1)` `" "=300" S " cm^(2)mol^(-1)` `" "=300 times 10^(-14)" S "m^(2)mol^(-1)` `alpha=(1.5 times 10^(-3)" S"m^(2)mol^(-1))/(300 times 10^(-4)Sm^(2)mol^(-1))` `alpha=0.05` |
|
| 20. |
The conductivity of a 0.01 M solution of a 1:1 weak electrolyte at 298 K is 1.5xx10^(-4)Scm^(-1). (i) molar conductivity of the solution (ii) degree of dissociation and the dissociation constant of the weak electrolyte Given that lambda_("cation")^(@)=248.2" S cm"^(2)" mol"^(-1) lambda_("anion")^(@)="51.8 S cm"^(2)" mol"^(-1) |
|
Answer» Solution :(i) Molar conductivity `C=0.01M` `kappa=1.5xx10^(-4)"S cm"^(-1)(or)kappa=1.5xx10^(-2)"S m"^(-1)` `Lambda_(m)=(kappaxx10^(-3))/(C )"Sm"^(-1)"mol"^(-1)m^(3)=(1.5xx10^(-2)xx10^(-3))/(0.01)"Sm"^(2)"mol"^(-1)` `Lambda_(m)=1.5xx10^(-3)"Sm"^(2)"mol"^(-1)` (ii) Degree of dissociation `ALPHA=(Lambda_(m))/(Lambda_(oo))(or)alpha=(Lambda_(m))/(Lambda_(m)^(@))` `Lambda_(oo)^(@)=lambda_("cation")^(@)+lambda_("anion")^(@)` `=(248.2+51.8)"S cm"^(2)"mol"^(-1)` `="300 S cm"^(2)"mol"^(-1)` `=300xx10^(-4)"S m"^(2)"mol"^(-1)` `alpha=(1.5xx10^(-3))/(300xx10^(-4))=0.05` `K_(a)=(alpha^(2)C)/(1-alpha)` `=((0.05)^(2)(0.01))/(1-0.05)` `K_(a)=2.63xx10^(-5)` |
|
| 21. |
The conductivity of 10^(-3) mol/L acetic acid at 25 °C is 4.1 xx 10^(-5) cm^(-1) . Calculate its degree of dissociation,Lambda_(m)^(@) if for acetic acid at 25 °C is 390.5 S cm^(2) mol^(-1). |
|
Answer» Solution :Apply the following relation: `Lambda_(m) = (1000 k)/C` Substituting the values in the above EQUATION, we have `Lambda_(m) =(1000 xx 4.1 xx 10^(-5))/10^(-3) = 41 S cm^(2) mol^(-1)` Degree of dissociation is given by the following equation: `alpha = Lambda_(m)^(C )/Lambda_(m)^(@) = 41/(390.5)= 0.105` or `10.5%` |
|
| 22. |
The conductivity of 0.20M solution of KCl of 298K is 0.0248 S cm^-1. Calculate its molar conductivity. |
|
Answer» SOLUTION :`A_m=(K times 1000)/(MOLARITY)` `=(0.0248 S cm^-1 times1000cm^3 L^-1)/(0.20 mol L^-1)` `=124 S cm^2 mol^-1` |
|
| 23. |
The conductivity of 0.20M solution of KCL at 298K is 0.025 S cm^-1. Calculate its molar conductivity. |
|
Answer» SOLUTION :CONCENTRATION of solution =0.20 M Conductivity =`0.025 S cm^-1` Molar conductivity `A_m` `=("Conductivity (K) TIMES 1000")/("Concentration of solution ")` `=(0.025 times 1000)/0.20` `=125 S cm^2 mol^-1` |
|
| 24. |
The conductivity of 0.20 M solution of KCl at 298K is 0.0248 S cm^(-1). Calculate its molar conductivity. |
| Answer» SOLUTION :`wedge_(m)=(kappaxx1000)/("MOLARITY")=(0.0248" S "cm^(-1)xx1000cm^(3)L^(-1))/(0.20" mol "L^(-1))=124" S "cm^(2)mol^(-1)` | |
| 25. |
The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm^(-1)?. Calculate its molar conductivity. |
|
Answer» Solution :Given that k = `0.025 S cm^(-1)` `C = 0.2 "moles" L^(-1) = 0.2` moles `(1000 cm^(3))^(-1) =(0.2 "moles")/(1000 cm^(3))` `Lambda_(m) = k/C = (0.025 S cm^(-1))/(0.2 "moles") XX 1000 cm^(3) = 125 S cm^(2) "mol"^(-1)` |
|
| 26. |
The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S cm^(-1). Calculate its molar conductivity. |
|
Answer» SOLUTION :* Molar conductivity `Lamda_(m)`: Molar conductivity `(Lamda_(m))=(kxx1000)/(c)` Where, conductivity `k=0.0248" S "cm^(-1)` `therefore Lamda_(m)=(kxx1000)/("Molarity")` `=((0.0248" S "cm^(-1))xx(1000cm^(3)DM^(-3)))/(0.20" mol "dm^(-3))(OR(m)L^(-1))/(OR" mol "L^(-1))` `=124.0S" "cm^(2)mol^(-1)` |
|
| 27. |
The conductivity of 0.1 M solution of AgNO_3 is 9.47 xx 10^(-3) S cm^(-1) at 291 K . The ionic conductivities of Ag^(+) and NO_(3)^(-) at the same temperature are 55.7 and 50.8 S cm^(2) "equiv"^(-1)respectively . Calculate the degree of dissociation of AgNO_3 in 0.1 M solution . |
|
Answer» `= 94.7 S CM^(2) "equiv"^(-1)` `Lambda^(oo) = lambda^(oo) (Ag^(+)) + lambda^(oo) (NO_(3)^(-)) = 106.5 S cm^(2) "equiv"^(-1)` `ALPHA = (94.7)/(106.5) = 0.089 or 89 %` |
|
| 28. |
The conductivity of 0.2 M solution of KCl at 298 K is 0.0248 S cm^(-1). Calculate its mlar conductivity. |
|
Answer» |
|
| 29. |
The conductivity of 0.20 M solution of KCI at 298 K is 0.0248 S cm^(-1).Calculate its molar conductivity. |
|
Answer» Solution :Applying the FOLLOWING relation and substituting the values, we get: `Lambda_(m) = (k xx 1000)/("MOLARITY") = (0.0248 S CM^(-1) xx 1000 cm^(3) L^(-1))/(0.20 mol L^(-1)) = 124 S cm^(2) mol^(-1)`. |
|
| 30. |
The conductivity of 0.1 M KCl solution is 1.29 S m^(-1). If the resistance of the cell filled with 0.1 M KCl is 100 Omega, Calculate the cell constant. |
|
Answer» Solution :The cell constant filled with electrolytic solution is GIVEN as , Cell constant = conductivity `xx` RESISTANCE. Cell constant `=1.29m^(-1)XX100 Omega` `=129m^(-1)=1.29cm^(-1)`. |
|
| 31. |
The conductivity of 0.01 mol L^(-1)KCl solution is 1.41xx10^(-3)" S "cm^(-1). What is the molar conductivity (S cm^(2) mol^(-1)) ? |
|
Answer» 14.1 `=(1000xx(1.41xx10^(-3)" S " cm^(-1)))/((0.01" MOL "cm^(-3)))=141" S "cm^(-2)mol^(-1)` |
|
| 32. |
The conductivity of 0.001028 M acetic acid is 4.95xx10^(-5)" S "cm^(-1). Calculate its dissociation constant if wedge^(@) for acetic acid is 390.5 S cm^(2)mol^(-1). |
|
Answer» Solution :For the given concentration of acetic acid solution, `wedge_(m)=(kappaxx1000)/(c)=(4.95xx10^(-5)S" "CM^(-1)xx1000cm^(3)L^(-1))/(0.001028" mol "L^(-1))=48.15" S "cm^(2)mol^(-1)` `alpha=(wedge_(m))/(wedge_(m)^(@))=(48.15" S "cm^(2)mol^(-1))/(390.5" S "cm^(2)mol^(-1))=0.1233` `{:(,CH_(3)COOH,HARR,CH_(3)COO^(-),+,H^(+)),("INITIAL conc.",c,,0,,0),("Equilibrium conc.",c-calpha=c(1-alpha),,calpha,,calpha):}` `K=(calpha.calpha)/(c(1-alpha))=(calpha^(2))/(1-alpha)=((0.001028" "molL^(-1))(0.1233)^(2))/(1-0.1233)=1.78xx10^(-5)mol" "L^(-1)` |
|
| 33. |
The conductivity of 0.01 M NaCl solution is 0.00147" ohm"^(-1)cm^(-1). What happens to the conductivity if extra 100 mL is added to the above solution. |
|
Answer» REMAINS same |
|
| 34. |
The conductivity of 0.001028 mol L^(-1)acetic acid is 4.95 xx 10^(-5) S cm^(-1). Calculate its dissociation constant if Lambda_(m)^(0) for acetic is 390.5 S cm^(2) mol^(-1) ? |
|
Answer» 14. 1 |
|
| 35. |
The conductivity of 0.001M acetic acid is 4 times 10^-8 S//cm. Calculate the dissociation constant of acetic acid, if molar conductivity at infinite dilution for acetic acid is 390 S cm^2//mol. |
|
Answer» SOLUTION :`A_m=(K TIMES 1000)/m` `=(4 times 10^-5 times 1000)/0.001` `alpha=A_m/A_m^0=40/390=0.102` |
|
| 36. |
The conductivity measurement of a coordination compound of cobalt (III) shows that it dissociates into 3 ions in solution. The compound is |
|
Answer» hexaamminecobalt (III) chloride `[Co(NH_(3))_(5)CL]Cl_(2)hArr ubrace([Co(NH_(3))_(5)Cl]^(2+)+2CL^(-))_(3"ions")` |
|
| 37. |
The conduction of electricity occurs by a direct low of electrons under the influence of applied potential are known as |
|
Answer» ELECTRO conductor |
|
| 38. |
The conductivity, K, of an electrolytic solution depends upon the concentration of electrolyte, nature of the solvent and _________ |
| Answer» SOLUTION :TEMPERATURE | |
| 39. |
The conductivity of 0.001 M acetic acid is 5 xx 10^(-5) S cm^(-1) and ^^""^(0) is 390.5 S cm^(2) "mol" ^(-1) then the calculated value of dissociation constant of acetic acid would be |
|
Answer» `81.78 XX 10^(-4)` |
|
| 40. |
The conductance of a 0.0015M aqueous solution of a weak monobasic acid was determined by using a conductivity acid was determined by using a cunductivity cell consisting of platinised pt electrodes. The distance between the electrodes is 120cm with an area of croos section of 1 cm^(2). The conductance of this solution was found to be 5xx10^(-7) S. the pH of the solution is 4. The value of limiting moalr conductivity (A_(m)^(@)) of this weak monobasic acid in aqueous solution is Zxx10^(2)S cm^(-1) "mol"^(-1). the value of Z is |
|
Answer» |
|
| 41. |
The conductance of a salt solution (AB) measured by two parallel electrodes of area 100 cm^(2) separated by 10 cm was found to be 0.0001 Omega^(-1). If volume enclosed between two electrode contains 0.1 mole of salt, and the molar conductivity (S cm^(2) mol^(-1)) of salt at same concentration is 1.0 xx 10^(-x), x is |
|
Answer» ` M = (N)/(V) = (0.1)/( 100 xx 100) xx 1000 = 0.1M , ^^_M = (K xx 1000)/(M) = (10^(-5) xx 1000)/(0.1) = 0.1 = 1 xx 10^(-x) = 1 xx 10^(-1)` x=1 |
|
| 42. |
The conductance of an emulsion increase on adding common slat, What type of emulsion is this ? |
| Answer» SOLUTION :Oil-in-water TYPE. | |
| 43. |
The conductance of solution of an electrolyte is equal to that of its specific conductance. The cell constant of the conductivity cell is equal to |
|
Answer» Resistance |
|
| 44. |
The conductance of a 0.0015 M aqueous solution of a weak monobasic acid was determined by using a conductivity cell consisting of Pt electrodes. The distance between the electrodes is 120 cm with an area of cross-section of 1 cm^(2). The conductance of this solution was found to be 5xx10^(-7)S. the pH of the solution is 4. the value of the limiting molar conductivity (wedge_(m)^(@)) of the weak monobasic acid is aqueous solution is Zxx10^(2).S" "cm^(2)mol^(-1). the value of Z is |
|
Answer» Solution :CONC `C=0.0015M`, conductance, `G=5xx10^(-7)S` `l=120cm,a=1cm^(2)` `wedge_(m)^(c)=(Kxx1000)/("Molarity")=(6xx10^(-5)xx1000)/(0.0015)` `=40" S "cm^(2)mol^(-1)` `{:(HA,HARR,H^(+),+,A^(-)),(C,,0,,0),(C-Calpha,,Calpha,,Calpha):}` `[H^(+)]=Calpha=10^(-4)M""(becausepH=4)` `theeforealpha=(10^(-4))/(C)=(10^(-4))/(0.0015)` `alpha=(wedge_(m)^(c))/(wedge_(m)^(@)) therefore(10^(-4))/(0.0015)=(40)/(wedge_(m)^(@))` or `wedge_(m)^(@)=(40xx0.0015)/(10^(-4))=6xx10^(2)" S "cm^(2)mol^(-1)` |
|
| 45. |
The conductance of a 0.0015 M aqueous solution of a weak monobasic acid was determined by using a conductivity cell consisting of Pt electrodes. The distance between the electrodes is 120 cm with an area of cross section of 1 cm^(2). The conductance of this solution was found to be 5 xx 10^(-7)S. The pH of the solution is 4. The value of limiting molar conductivity (Lambda^(@)) of this monobasic acid in aqueous solution is Z xx 10^(2)S cm^(-1) mol^(-1). The value of Z is ......... . |
|
Answer» |
|
| 46. |
The conductance of a 0.0015 M aqueous solution of a weak monobasic acid was determined by using a conductivity cell consisting of platinized Pt electrodes. The distance between the electrodes is 120 cm with an area of cross section of 1 cm^(2). The conductance of this solution was found to be 5xx10^(-7)S. the pH of the solution is 4. The value of limiting molar conductivity (wedge_(m)^(0)) of this weak monobasic acid is aqueous solution is Zxx10^(2)S" "cm^(-1)mol^(-1). the value of Z is:- |
|
Answer» Solution :`C=0.0015M""l=120cm` `G=5xx10^(-7)s""a=1cm^(2)` `G=KXX(a)/(l)` `5xx10^(-7)=Kxx(1)/(120)` `K=6xx10^(-5)S" "cm^-1` `(^^)_(m)^(c)=(Kxx1000)/(M)=(6xx10^(-5)xx1000)/(0.0015)` `pH=4implies[H^(+)]=10^(-4)=calpha=0.0015alpha` `alpha=(10^(-4))/(0.0015)""alpha=((^^)_(m)^(c))/((^^)_(m)^(0))impleis(10^(-4))/(0.0015)=((6xx10^(-5)xx1000)/(0.0015))/((^^)_(m)^(0))` `(^^)_(m)^(0)=6xx10^(2)s" "cm^(2)"mole"-1` |
|
| 47. |
The conductance of a 0.0015 M aqueous solution of a week monobasic acid was determind by using a conductivity cell consisting of plantnized Pt electrodes. The distance between the electrods is 120 cm with an area of cross section was found to be5xx10^-7S. The pH of the solution is 4. The value of limiting molar conductivity(Lambda_m^0) of the weak monobasic acid is aqueous solution is Zxx10^2S cm^(-1). The value of Z is |
|
Answer» |
|
| 48. |
The conductance of 0.0015 M aqueous solution of a weak monobasic acid was determined by using a conductivity cellconsisting of platinized Pt electrodes. The distance between the electrodes is 120 cm with an area of cross section of 1 cm^(2). The conductance of this solution was found to be 5xx10^(-7)" S ". The pH of the solution is 4. The value of limiting molar conductivity Lambda_(m)^(0) ofweak monobasic acid in aqueous solution is Zxx10^(2)" S "cm^(-1). The value of Z is |
|
Answer» SOLUTION :z=6 For solution, CONSULT SOLVED EXAMPLE 3.68 |
|
| 49. |
The conditions that are necessary in the preparation of aryl halides from arenes |
|
Answer» LOW temperature |
|