Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The conversionof 1^(0)amine to 3^(0)amine . Theprocess involved maybe called . |
|
Answer» reduction |
|
| 2. |
The conversion of 2,3-dibromobutane to 2-butene with zinc and alcohol is : |
|
Answer» REDOX reaction |
|
| 4. |
The continuum in an atomic spectrum is due to |
|
Answer» INSTABILITY in EXCITED state |
|
| 5. |
The contribution of particle at the edge centre to a particular unit cell is |
|
Answer» `1/2` |
|
| 6. |
The continuous etherification process will give _________. |
|
Answer» only simpleethers |
|
| 7. |
The contribution of particle at the edge centre to a particular unit cell is, |
|
Answer» `(1)/(2)` |
|
| 8. |
Name the monomers of Nylon-6,6. |
|
Answer» BENZOIC acid and ETHYLAMINE |
|
| 9. |
The constant boiling mixture of two miscible liquids of a definite mole fraction is called ………………. . |
| Answer» SOLUTION :AZEOTROPE | |
| 10. |
The constant 'a' in van der Waals' equation is maximum in |
| Answer» ANSWER :D | |
| 11. |
The consituent of the powerful explosive RDX is formed during the nitration of |
|
Answer» toluene |
|
| 12. |
The consitutent particles in carborundum |
|
Answer» atoms |
|
| 13. |
The consequences of global warming may be |
|
Answer» increase in AVERAGE temperature of the earth |
|
| 14. |
The connect kinetic rate equation for the addition-elimination mechanism of nucleophilic aromatic substitution |
|
Answer» RATE = k [ARYL halide][nucleophile] 
|
|
| 15. |
The conjugate base of serotonin (used as tranquilisers) is given as follows: How many basic groups present in following compound? |
|
Answer» |
|
| 16. |
The connect difference between first and second-order reactions is that |
|
Answer» a first - ORDER reaction can be catalyzed, a second-order reaction cannot be catalyzed |
|
| 17. |
The conjugate base of NH_(2)^(-) is |
|
Answer» `NH_(3)` Conjugate acid, base pair. |
|
| 19. |
The conjugate base of HSO_(3)^(-) is |
|
Answer» `H_(2)SO_(3)` `underset("ACID")(HSO_(3)^(-)) hArr underset("conjugate base")(SO_(3)^(2-))+H^(+)` |
|
| 23. |
The conjugate base of H_2SO_4 in the following reaction is: H_2SO_4 + H_2O harr H_3O^+ + HSO_3^-4 |
|
Answer» `H_2O` |
|
| 25. |
The conjugate base of H_3O^+is _____. |
|
Answer» |
|
| 26. |
The conjugate base of H_(2)PO_(4)^(-) is |
|
Answer» `H_(3)PO_(4)` |
|
| 27. |
The conjugatebaseof (CH_(3))_(2)NH_(2)^(+) is |
|
Answer» `(CH_(3))_(2)` |
|
| 28. |
The conjugate base of (CH_(3))_(3)NH^(+) is : |
|
Answer» `(CH_(3))_(3)N` |
|
| 29. |
The conjugate base of (CH_(3))_(2)NH_(2)^(+) is |
|
Answer» `CH_(3))_(3)` N |
|
| 31. |
The conjugate base in the following reaction H_(2)SO_(4)+H_(2)O rarr H_(3)O^(+) + HSO_(4)^(-) |
|
Answer» `H_(2)O` 
|
|
| 32. |
The conjugate base of [Al(H_(2)O)_(3) (OH)_(3) is |
|
Answer» `[A(H_(2)O)_(3)(OH)_(2)]^(+)` |
|
| 33. |
The conjugate base of [Al (H_2O)_3(OH)_3] is : |
|
Answer» `[Al(H_2O)_3 (OH)_2]^+` |
|
| 34. |
The conjugate acid of S_(2)O_(8)^(2-) is |
|
Answer» `H_(2)S_(2)O_(8)` |
|
| 35. |
The conjugate acid of NH_(2)^(-) is |
|
Answer» `NH_(3)` |
|
| 36. |
The conjugate acid of NH_(2)^(-) is : |
|
Answer» `NH_(3)` |
|
| 37. |
The conjugate acid of NH_2^- IS : |
|
Answer» `NH_3` |
|
| 39. |
The conjugate acid of HPO_(4)^(2-) is |
|
Answer» `H_(2)PO_(4)^(-)` |
|
| 40. |
The conjugate acid of HPO_(3)^(2-) is |
|
Answer» `H_(3)PO_(4)` |
|
| 41. |
The conjugate acid of HCO_3^- is _____. |
|
Answer» |
|
| 42. |
The conjugate acid of H_(2)PO_(4)^(-) is |
|
Answer» `H_(3)PO_(4)` |
|
| 45. |
The configuration of the given compound is |
|
Answer» E |
|
| 46. |
The conformations of n-butane, commonly known as eclipsed, gauche and anti-conformations can be interconverted by |
|
Answer» rotation AROUND C-H bond of a methyl group 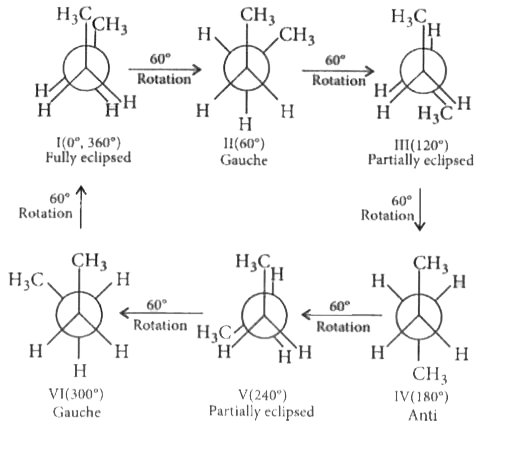
|
|
| 47. |
The configuration of the following is …… |
|
Answer» 1S, 2S 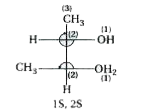 CHANGE R to S since H i.e., atom of lowest PRIORITY is on horizontal axis. |
|
| 48. |
The configuration of eight compounds, a through h are shown below, using various kind of stereo representations. To answer the question given below. Write (a through h ) indicating your choice. |
|
Answer» |
|
| 49. |
the configuration of an element 'X' is 4s^1 3d^10. The wrong statement regarding the element 'X' is |
|
Answer» it forms COMPLEXES |
|
| 50. |
The conductivity of strong electrolyte is ……….. . |
|
Answer» INCREASE on dilutin slightly |
|