Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The cooking in refrigerator is due to: |
|
Answer» Reaction of the refrigerator GAS |
|
| 2. |
The -COOH group in a carboxylic acid can be replaced by .H. by heating the acid with: |
|
Answer» ZN with HCL |
|
| 3. |
The conversion reaction of phenol to salicylic acid is known as ……………………. . |
| Answer» SOLUTION :Kolbe.s SCHMITT REACTION | |
| 4. |
The conversion reaction of phenol of salicylic acid is known as …………………. . |
|
Answer» Schottan-Baumann REACTION |
|
| 5. |
The conversion reaction of Benzene to Benzaldehyde is known as .... |
|
Answer» ROSENMUND REDUCTION |
|
| 6. |
The conversion reaction of phenol into salieylaldehyde is known as ………………… . |
| Answer» SOLUTION :Riemer-Tiemann REACTION | |
| 7. |
The conversion of trypsinogen (A) into trypsin (B) is an autocatalytic reaction Ararr B where B catalyses the reaction. The rate equation is (-dx)/(dt) =K. x. y, where x and y are concentration of tripsinogen at time t. Integrate this equation for initial concentration of x_(0) and y_(0) for A and B. show that Kt=2.303/(x_(0)+y_(0))"log"(y.x_(0))/(x.y_(0)) |
|
Answer» |
|
| 8. |
The conversion of m-nitrophenol to resorcinol inivolves respectively: |
|
Answer» HYDROLYSIS , DIAZOTIZATION and REDUCTION |
|
| 9. |
The conversion of the molecules X to Y follows second order kinetics. If the concentration X is increased to three times, how will it affect the rate of formation of Y? |
|
Answer» Solution :For the reaction, `X to Y`, as it follows second order kinetics, the rate law equation will be Rate `=K[X]^(2)` If concentration of X is increased to three times, then Rate `=k[3X]^(2) "orRate"=9K[X]^(2)` Thus, the RTE of reaction will become 9 times. Hence, the rateof formation of Y will INCREASE 9 times. |
|
| 10. |
The conversion of the molecules X to Y follows second order kinetics. If the concentration X is increased to three times, how will it affect the rate of formation of Y ? |
|
Answer» Solution :For the reaction, `XtoY`, as it follows SECOND order KINETICS, the rate LAW equation will be Rate `=k[X]^(2)=ka^(2)""("if "[X]=a" mol L"^(-1))` If concentration of X is increased three times, now, `[X]=3"a mol L"^(-1)` `:."""Rate"=k(3" a")^(2)=9" ka"^(2)` Thus, the rate of reaction will become 9 times. Hence, the rate of FORMATION of Y will increase 9 times. |
|
| 11. |
The conversion of the molecules X to Y follows second order kinetics. If the concentration of X is increased to three times, how will it affect the rate of formation of Y ? |
| Answer» SOLUTION :9 TIMES | |
| 12. |
The conversion of primary aromatic amines into diazonium salts is known as ________. |
|
Answer» |
|
| 13. |
The conversion of pig iron to steel frequently requires the addition of: (I) oxygen or iron oxide(II) transition elements(III)inner transition elements(IV)silica |
|
Answer» I, II, III |
|
| 14. |
The conversion of ozone into oxygen is exothermic. under what conditions is ozone the most stable ? 2O_(3)(g) hArr 3O_(2)(g) |
|
Answer» At LOW T and low P `Deltang=o+ ve`. |
|
| 15. |
The conversion of PbS to Pb is : |
|
Answer» Dissociation |
|
| 16. |
The conversion of one element into another by aritificial means is termed ………………………... |
|
Answer» |
|
| 17. |
The conversion of molecules X to Y follows second order kinetics.If concentration of X is increased to three times how will it affect the rate of formation of Y? |
|
Answer» Solution :Given reaction X `to` Y Order of reaction =2 `therefore Rate r_(1)=-K[x]^(2)` Now,the CONCENTRATION of X increase to THREE time then new concentration is =3X mol `L^(-1)` `therefore New rate r_(2)=k[3X]^(2)=9K [x]^(2)` `therefore (r_(2))/(r_(1))=(9k[X]^(2))/(k[X]^(2))=9` So ,new rate is nine times. `therefore` so,it will effect nine times in the rate of formation of Y. |
|
| 18. |
The conversionof nitroaklane toprimaryamineis carried out by |
|
Answer» REDUCTION |
|
| 19. |
The conversion of molecules x to y follows second order kinetics. It concentration of x is increased to three times how will it affect the rate of formation of y ? For the reaction x rarr y as it follows second order kinetics wherefore the rate of formation of y ? |
|
Answer» SOLUTION :Rate `=k[x]^(2)=KA^(2)` `[x]="a mol"^(-1)` If the concentration of x is incross three time, then `(x)=3"a mol L"^(-1)` Rate `= R(3A)^(2)=9 ka^(2)` Hence, the rate of formation will increase by 9 times. |
|
| 20. |
The conversion of molecules X to Y follows second order kinetics . If concentration of X is increased to three times how will it affect the rate of formation of Y ? |
|
Answer» Solution :For the REACTION , `X rarrY` as it follows second order kinetics , therefore the rate law EQUATION will be Rate `=k[X]^2=ka^2` if`[X]= "a mol"^(-1)` If the CONCENTRATION of X is increased three times , then `[X] = 3 " a mol L"^(-1)` `:.` Rate `=k(3A)^2=9ka^2` Thus, the rate of the reaction will become 9 times. Hence the rate of FORMATION of Y will increase by 9 times. |
|
| 21. |
The conversion of molecules X to Y follows second order kinetics. IF concentration of X is increased to three times how will it affect the rate of formation of Y. |
| Answer» Solution :`r_(1) = K[X]^(2), " "r_(2) = k[3X]^(2), ""r_(2) = 9K[X]^(2)` RATE increases by 9 TIMES. | |
| 22. |
The conversion of molecules of A to B follows second order kineitcs. Doubling the concentration of A will increase the rate of formation of B by a factor of? |
|
Answer» |
|
| 23. |
The conversion of molecules of X to Y follows second order kinetics. If concentration of X is increases to three how will it affect the rate of formation of Y? |
|
Answer» SOLUTION :Rate (r)=`K[A]^2`,If [A] is increased by 4 times,r=`k[4A]^2=16k[A]^2` `therefore`Rate of reaction (i.e.,formation of B)increased by 16 times |
|
| 24. |
The conversion of molecule A to B follows second order kinetics. Indicate the order and molecularity of the reaction given below:C_12H_22O_11+H_2OoversetHtoC_6H_12O_6+C_6H_12O_6 |
|
Answer» Solution :This is Pseudo order REACTION `THEREFORE`Order =1,Molecularity=2 |
|
| 25. |
The conversion of maltose to glucose is possible by the enzyme : |
|
Answer» zymase |
|
| 26. |
The conversion of m-nitrophenol to resorcinol involves respectively |
|
Answer» Hydrolysis,diazotization and REDUCTION 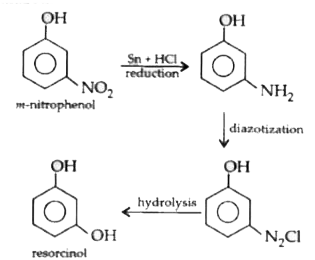 . .
|
|
| 27. |
The conversion of liquid hydrocarbon into a mixture of gaseous compounds by heat alone is known as: |
|
Answer» Hydrolysis |
|
| 28. |
The conversion of glucose into glycogen in liver is called: |
|
Answer» Glycogenolysis |
|
| 29. |
The conversion of ethyl methyl ketoxime to N-methylpropanamide represents the example of which of the following type of the following type of organic reaction |
|
Answer» ELIMINATION |
|
| 30. |
The conversion of Ethyl acetate to propyl acetate by the action of propyl alcohol is named as |
|
Answer» Esterification |
|
| 31. |
The conversion of d-form of an optically active compound into l-form of the same or of different compound or vice versa is known as : |
|
Answer» Resolution |
|
| 32. |
The conversion of-COOH group to-NH_2 group can be made by: |
|
Answer» WURTZ reaction |
|
| 33. |
The conversion of CH_3OH to CH_3COOH can be brought in by: |
|
Answer» `K_2Cr2O7 |
|
| 34. |
The conversion of Benzene to Benzaldehyde by the action of carbon monoxide and HCl is known as....... |
| Answer» SOLUTION :Gattermann-koch REACTION | |
| 35. |
The conversion of Benzene diaxonium chloride is Biphenyl is called …………….. Reaction. |
|
Answer» |
|
| 36. |
The conversion of benzene diazonium chloride to benzene by H_(3)PO_(2) proceeds through …………………. Mechanism. |
|
Answer» |
|
| 37. |
The conversion of an amide to an amine with one carbon atom less by the action of alkaline hydrohalite is known as Hofmann bromamide degradation In this reaction, RCONBr is formed from which the reaction has derived its name. Hofmann degradation is an intra molecular reaction. What are the constituent amines formed when the mixture of (i) and (ii) undergoes Hofmann bromamide degradation ? |
|
Answer»
|
|
| 38. |
The conversion of an amide to an amine with one carbon atom less by the action of alkaline hydrohalite is known as Hofmann bromamide degradation In this reaction, RCONBr is formed from which the reaction has derived its name. Hofmann degradation is an intra molecular reaction. Which is the rate determining step in Hofman bromamide degradiation ? |
|
Answer» Formation of (i) |
|
| 39. |
The conversion of atomic hydrogen info ordinary hydrogen is : |
|
Answer» EXOTHERMIC CHANGE |
|
| 40. |
The conversion of an amide to an amine with one carbon atom less by the action of alkaline hydrohalite is known as Hofmann bromamide degradation In this reaction, RCONBr is formed from which the reaction has derived its name. Hofmann degradation is an intra molecular reaction. How can be conversion (i) to (ii) be brought about ? |
|
Answer» `KBR` |
|
| 41. |
The conversion of an amide into an amine with one carbon atom less by the action of alkaline hydrohalite is known as Hofmann bromamide rearrangement: RCONH_(2) overset(Br_(2)//KOH)toR-NH_(2) The most important feature of the reaction is the arrangement of N–bromamide anion to isocyanate : Predict the product in the following reaction |
|
Answer» I and II |
|
| 42. |
The conversion of acetyl chloride to acetaldehyde by the action of Pd//BaSO_4is called |
|
Answer» PERKIN's REACTION |
|
| 43. |
The conversion of an alkyl halide into an alcohol by aqueous NaOH is classified as |
|
Answer» a DEHYDROHALOGENATION reaction |
|
| 44. |
The conversion of acetamide to methylamine by the action of caustic alkali and Bromine is known as .......... |
| Answer» SOLUTION :HOFF mann.s DEGRADATION | |
| 45. |
The conversion of A to B follows second order kinetics. Doubling the concentration of A will increases the rate of formation of B by a factor of |
|
Answer» Solution :`"Rate"_(1) = K[A]^(2)` `"Rate"_(2) = K[2A]^(2) = 4K[A]^(2)` `RARR ("Rate"_(2))/("Rate"_(1)) = 4 rArr "Rate"_(2) = 4 XX "Rate"_(1)` |
|
| 46. |
The conversion of A to B follows second order kinetics. Doubling the conc. of A will increase the rate of formation of B by factor |
|
Answer» 4 |
|
| 47. |
The conversion of a precipitate into colloid is called ................. |
|
Answer» COAGULATION |
|
| 48. |
The conversion of a carboxylic acid to alpha-bromocarboxylic acid using red phosphorus and bromine is a |
|
Answer» CANNIZZARO's reaction |
|
| 49. |
The conversion of a carboxylic acidto alpha - bromo- carboxylic acid using red phosphorus and bromine is a: |
|
Answer» CANNIZZARO 's reaction |
|
| 50. |
The conversion of 2,3,-dibromobutane to 2-butene with Zn and alcohol is |
|
Answer» redox reaction Since TWO BROMINE atoms are lost from different carbon atom, the reaction is known as `beta`-elimination. |
|



