Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The condition of vitamin deficiency is known as |
|
Answer» Vitaminosis |
|
| 2. |
The condition for spontaneity of a process is |
|
Answer» <P>lowering of entropy at constant temperature and PRESSURE |
|
| 3. |
The condition of spontaneity of process is |
|
Answer» LOWERING of entropy at constant TEMPERATURE and pressure |
|
| 4. |
The condition for minimum change in pH for a buffer solution is |
|
Answer» Isoelectronic species are ADDED |
|
| 5. |
The condition for a reaction to occur spontaneously is |
|
Answer» `DELTA H`must be negative ` therefore `For a SPONTANEOUS reaction, `(Delta H - T Delta S)`should be negative. |
|
| 6. |
The condition for a compound to be precipitated is |
|
Answer» IONIC product= SOLUBILITY product |
|
| 7. |
The condensation reaction of esters with nitriles containing alpha- hydrogen is known as………………… |
|
Answer» |
|
| 8. |
The condensation polymer among the following is |
|
Answer» Protein |
|
| 10. |
The condensation of two amino acids, glycine and (pm) alanine yields total products x. Find value of x ? |
|
Answer» (1)Gly-Gly (2)Gly-ALA(+) (3)Gly-Ala(-) (4)Ala(+)-Gly (5)Ala(-)-Gly (6)Ala(+)-Ala(+) (7)Ala(-)-Ala(-) (8)Ala(+)-Ala(-) (9)Ala(-)-Ala(+) |
|
| 11. |
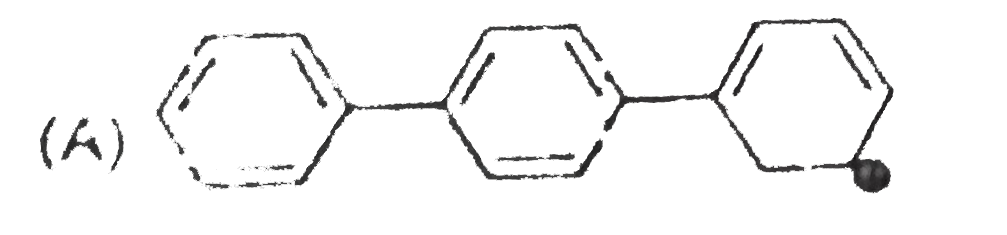
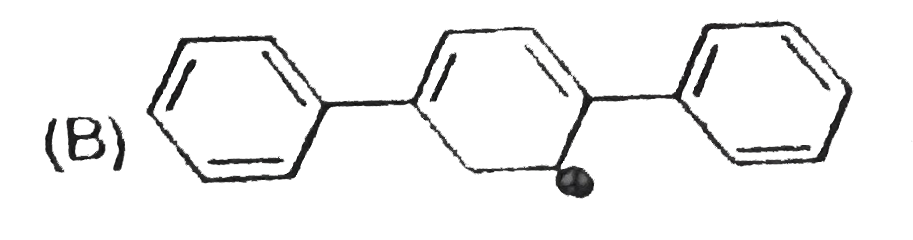
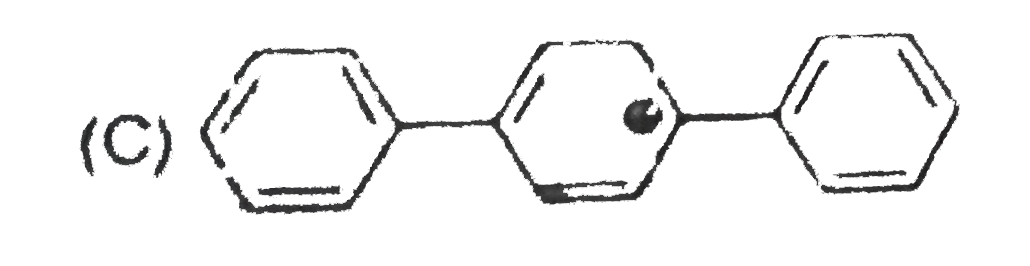
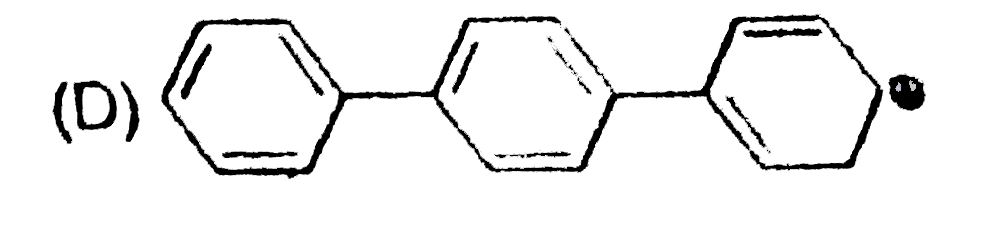
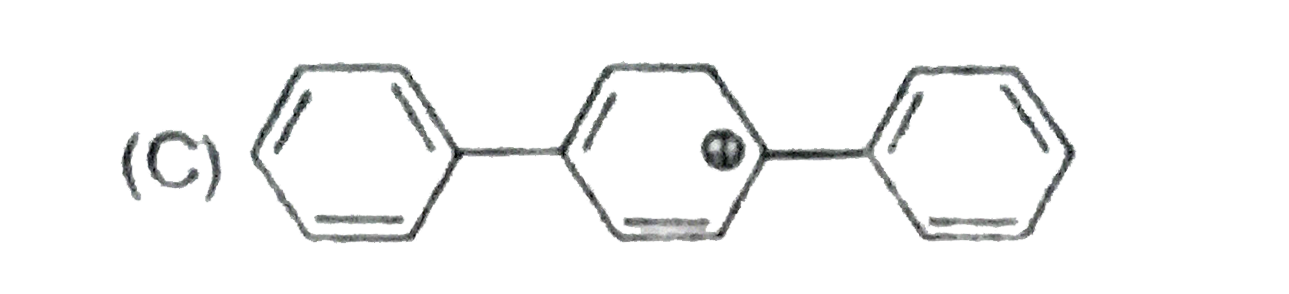
The concept of resonance explains various properties of compounds.The molecules with conjugated system of pi bonds, are stabilized by resonace and have low heat of hydrogenation.Hyperconjugative stabilization also decreases heat f hydrogenation.In aromic rings a functional group with a lone pair of electron exerts +m effect. Some functional groups like -NO, -NC, -CH=CH_2 can function both as electron releasing (+m,+R) or electron withdrawing (-m,-R) groups.More extended conjugation provides more stabilization. The most stable carbocation is : |
|
Answer»

|
|
| 12. |
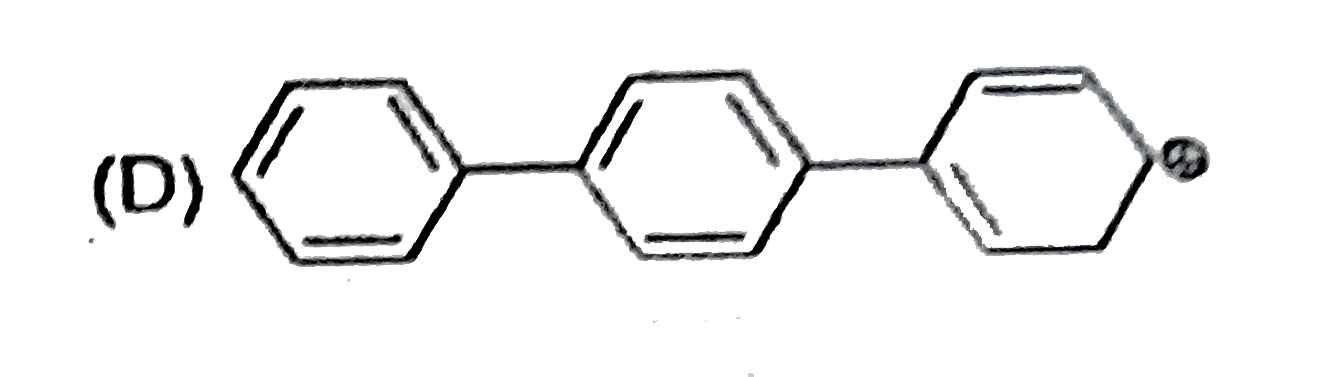
The concept of resonance explains various properties of compounds.The molecules with conjugated system of pi bonds, are stabilized by resonace and have low heat of hydrogenation.Hyperconjugative stabilization also decreases heat f hydrogenation.In aromic rings a functional group with a lone pair of electron exerts +m effect. Some functional groups like -NO, -NC, -CH=CH_2 can function both as electron releasing (+m,+R) or electron withdrawing (-m,-R) groups.More extended conjugation provides more stabilization. The correct heat of hydrogenation order is (p)1,3-Pentadiene , (q)1,3-Butadiene , (r)2,3-Dimethyl-1,3-butadiene , (s)Propadiene |
|
Answer» pgtqgtrgts 
|
|
| 13. |
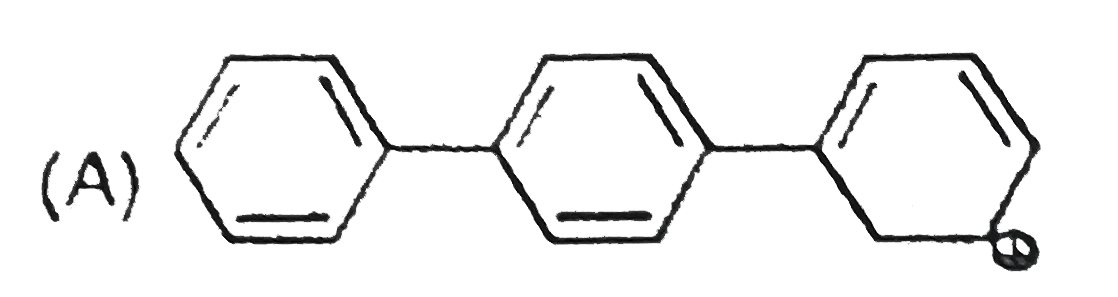
The concept of resonance explains various properties of compounds.The molecule with conjugated system of pi bonds, are stabilized by resonance and have low heat of hydrogenation .Hyperconjugative stabilization also decreases heat of hydrogenation. In aromatic rings a functional group with a lone pair of electron exerts +m effect.Some functional groups like -NO, -NC, -CH=CH_2 can function both as electrons releasing (+m,+R) or electron withdrawing (-m,-R) groups.More extended conjugation provides more stabilization. The most stable resonating structure of following compound is |
|
Answer»
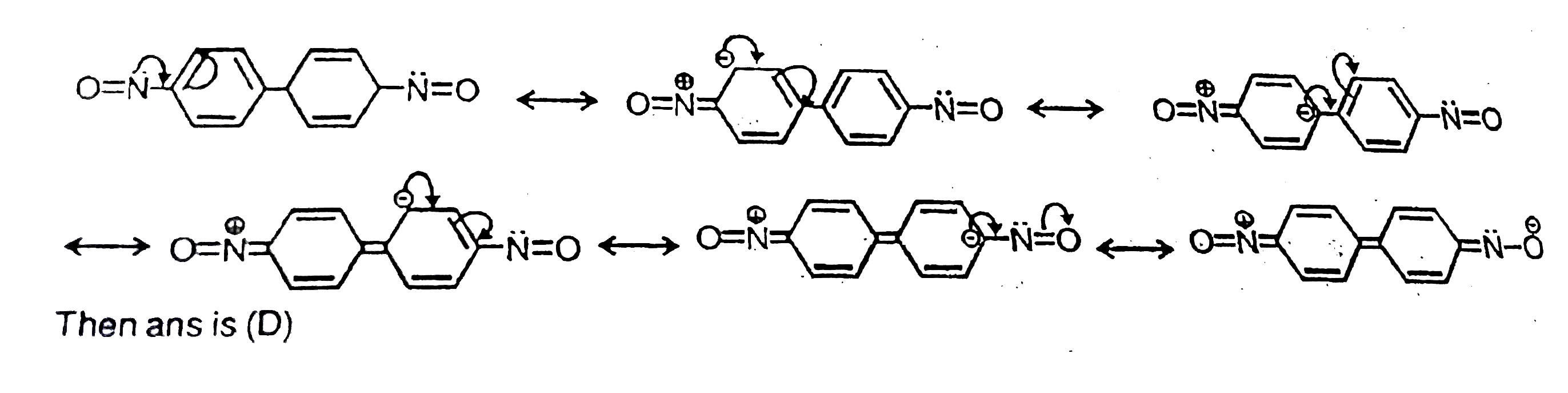
|
|
| 14. |
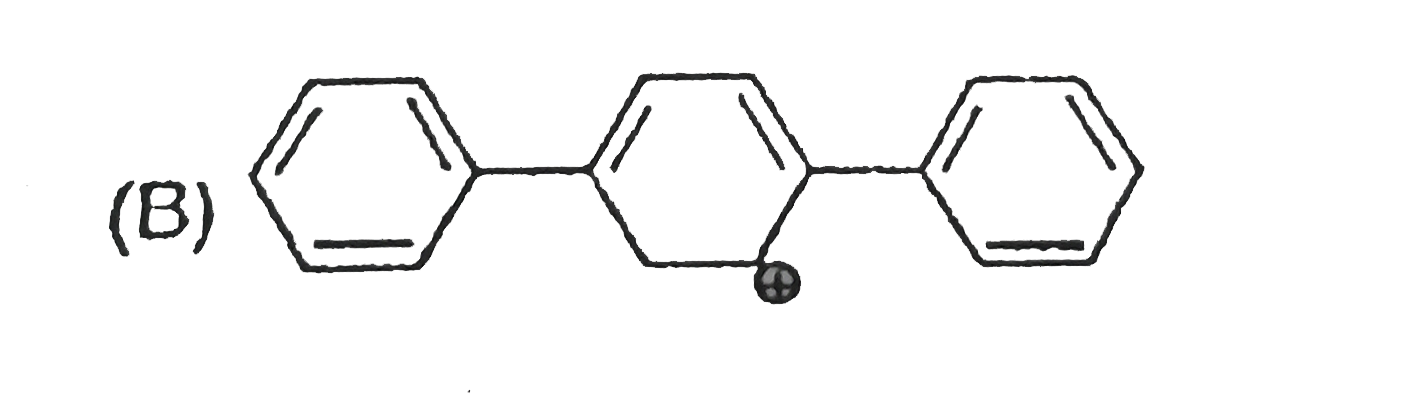
The concept of resonance explains various properties of compounds.The molecule with conjugated system of pi bonds, are stabilized by resonance and have low heat of hydrogenation .Hyperconjugative stabilization also decreases heat of hydrogenation. In aromatic rings a functional group with a lone pair of electron exerts +m effect.Some functional groups like -NO, -NC, -CH=CH_2 can function both as electrons releasing (+m,+R) or electron withdrawing (-m,-R) groups.More extended conjugation provides more stabilization. The most stable carbocation is |
|
Answer»
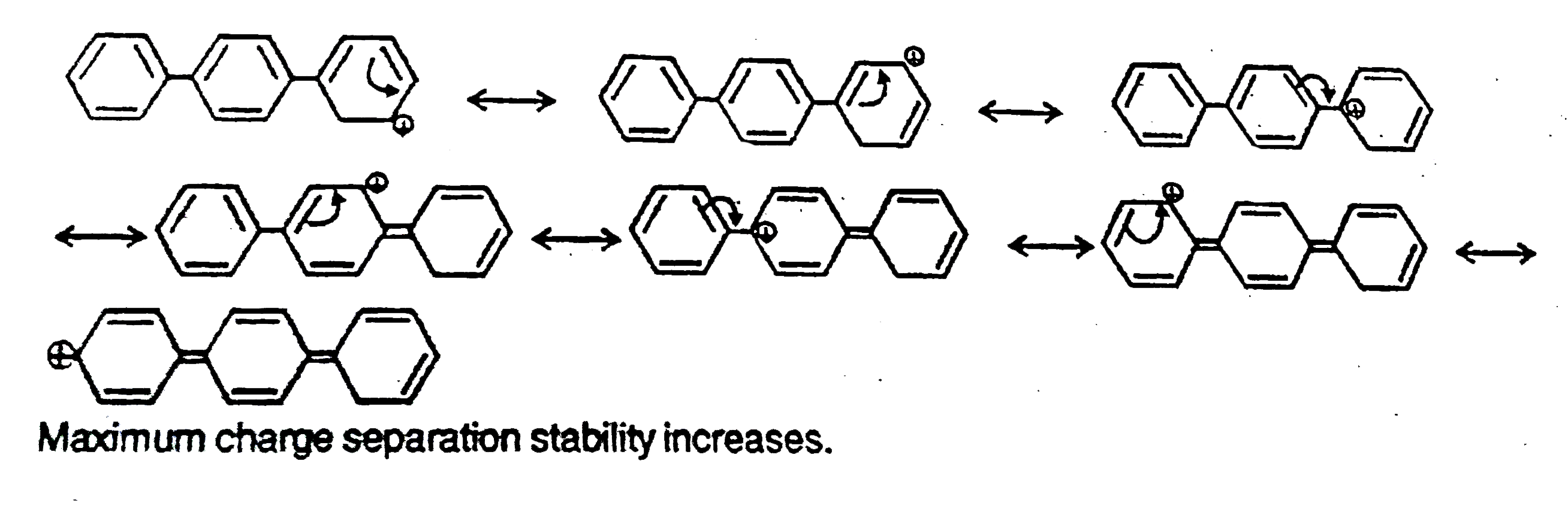
|
|
| 15. |
The concept of resonance explains various properties of compounds.The molecule with conjugated system of pi bonds, are stabilized by resonance and have low heat of hydrogenation .Hyperconjugative stabilization also decreases heat of hydrogenation. In aromatic rings a functional group with a lone pair of electron exerts +m effect.Some functional groups like -NO, -NC, -CH=CH_2 can function both as electrons releasing (+m,+R) or electron withdrawing (-m,-R) groups.More extended conjugation provides more stabilization. The correct heat of hydrogenation order is (p)1,3-Pentadiene , (q)1,3-Butadiene , (r)2,3-Dimethyl-1,3-butadiene , (s)Propadiene |
|
Answer» pgtqgtrgts 
|
|
| 17. |
The concentraton of hydrochloric acid in agiven solutioon is 10^(-8)M what is the vlaue of pH for this solution? |
|
Answer» salts have ORDERED packing arrrangements called lattices |
|
| 18. |
The concept of critical temperature for a gas was given by : |
|
Answer» Andrew |
|
| 19. |
The concentrations of N_(2)O_(5) decomposingin firstorder kinetics after 800 s is1.45 mol L^(-1) andafter1600 s is 0.88 mol L^(-1). Calculate the rateconstant. |
|
Answer» |
|
| 20. |
The concentration term independent of temperature is molarity/molality. |
| Answer» SOLUTION :MOLALITY | |
| 21. |
The concentration R in the reaction RtoP was measured as a function of time and the following data is obtained. The order of reaction is |
|
Answer» `(d[R])/(dt) = (1-0.75)/(0.05-0) = 0.25/0.05 =5` `(d[R])/(dt) = (0.75-0.40)/(0.12-0.05) = 0.35/0.07=5` |
|
| 22. |
The concentrationfo R in the reaction RtoP was measured as a function of time and the following data is obtained: [R] (molar)""1.0""0.75""0.40""0.10 t(min)""0.0""0.05""0.12""0.18 The order of the reactionis |
|
Answer» r=K = constant so ZERO ORDER |
|
| 23. |
The concentration of which ion is to be decreased, when NH_(3) solution is added |
|
Answer» `OH^(-)` |
|
| 24. |
The concentration of the reactant A in the reaction AtoB at different times are given below: "Concentration "(M)"""Time(seconds)" 0.069""0 0.052""17 0.035""34 0.018""51 The rate constant of the reaction according to the correct order of reaction is |
|
Answer» `0.001 M^(-2)s^(-1)` |
|
| 25. |
The concentration of the aqueous solutions of each of the following salts is 0.01 (M) KF,CH_3N^(o+)H_3C^(o+)l,KCl,CH_3COOH If K_a(HF)=7.0xx10^(-4),K_b(CH_3NH_2)=5xx10^(-4) and K_a(CH_3COOH)=1.8xx10^(-5) then increasing order of the pH values of the solutions- |
|
Answer» `KClltKFltCH_3N^(o+)H_3Cl^(o+)ltCH_3COOH` |
|
| 26. |
The concentration of sulphide ion in 0.1 M HCI solution saturated with hydrogen sulphide is 1.0 xx10^(-19) M. If 10 mL of this solution is added to 5 ml of 0.04 M solution of FeSO_4, MnCl_2, ZnCl_2 and CdCl_2, in which solutions precipitation will take place? (Given K_(sp)for FeS = 6,3 xx10^(-18) , MnS = 2.5 xx10^(-13), ZnS = 1.6 xx10^(-24) and CdS = 8.0 xx10^(-27).) |
|
Answer» `FeSO_4` `[s^(2-)]=1.0 xx10^(-19) xx (10)/(15 ) =6.67 xx 10^(-20)` `[Fe^(2+)] = [Mn^(2+)] = [Zn^(2+)] = [Cd^(2+)]` `=(5)/( 15) xx 0.04 = 1.33 xx 10^(-2)M` ` therefore ` Ionicproduct FOREACH of thesewill be ` =[M^(2+)] [S^(2-)]` `=(1.33 xx 10^(-2) ) ( 6.67 xx 10^(-20)) = 8.87 xx 10^(-22)` asthisvalueis greater than the solubilityproductof ` ZnS ` andCdstherefore`,ZnCI_2`and `CdCI_2`solutionswillbe precipitatedwhereasFeSand MnSis lesserthansolubilityproductthereforeprecipitationdoes nottakeplacein thiscase . |
|
| 27. |
The concentration of potassium ions inside a biological cell is least twenty time higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is M_((s))|M^(+)(aq,0.05" molar")||M^(+)(aq,1" molar")|M_((s)) For the above electrolytic cell the magnitude of the cell potential |E_(cell)|=70mV. Q. For the above cell |
|
Answer» `E_(cell) LT 0, DELTAG gt 0` =a POSITIVE value =70mV Hence `DeltaG lt 0`. [GIVEN] |
|
| 28. |
The concentration of potassium ions inside a biological cell is least twenty time higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is M_((s))|M^(+)(aq,0.05" molar")||M^(+)(aq,1" molar")|M_((s)) For the above electrolytic cell the magnitude of the cell potential |E_(cell)|=70mV. Q. if the 0.05 molar solution of M^(+) is replaced by a 0.0025 molar M^(+) solution,then the magnitude of the cell potential would be |
|
Answer» 35 mV `=(2.303RT)/(F)log(0.05)^(2)=2xx70mV=140mV`. |
|
| 29. |
The concentration of R in the reaction R to P was measured as a funcation of time and the following data is obtained: |
|
Answer» |
|
| 30. |
The concentration of pure solid and liquid phase is not inculded in the expression of equilibrium constant because: |
|
Answer» SOLID and liquid CONC. are independent of their quarries. |
|
| 31. |
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such concentration cell involving a metal M is : M_((s)) |M_((aq , 0.05(" molar"))^(+) ||M_((aq, 1 " molar"))^(+) | M_(s) For the above electrolytic cell the magnitude of the cell potential |E_("cell")|=70mV. If the 0.05 molar solution of M^(+) is replaced by a 0.0025 molar M^(+) solution, then the magnitudethe cell potential would be |
|
Answer» `35mV ` `((2.303RT)/(nF)) = ((70)/(1.301)), " But " E_2 = E^0 - ((2.303 RT)/(nF)) XX log(2.5 xx 10^(-4))` `E_2 = 0 - ((70)/(1.303)) xx (-2.602) , E_2 = 140 mV ` |
|
| 32. |
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such concentration cell involving a metal M is : M_((s)) |M_((aq , 0.05(" molar"))^(+) ||M_((aq, 1 " molar"))^(+) | M_(s) For the above electrolytic cell the magnitude of the cell potential |E_("cell")|=70mV. For the above cell |
|
Answer» `E_("cell") lt 0 , DELTAG GT 0` |
|
| 33. |
The concentration ofpotassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple mode for such a concentration cell involving a metal M is : M_((s))|M_((aq.))^(@) 0.05"molar"||M_((aq.))^(@) 1"molar"|M_((s)) For the above electrolytic cell the magnitude of the cell potential |E_(cell) | = 70mV If the 0.05 molar solution of M^(+) is replaced by 0.0025 molar M^(+) solution, then the magnitude of the cell potential would be : |
|
Answer» `35 mV` `E_(cell) = (0.059)/(1)"log"_(10)([1.0])/(2.5 xx 10^(-3))` `= 0.152 V` or `APPROX 140 mV` |
|
| 34. |
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in serveral processes such as transmission of nerve impules and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is : M_((s))|M^(+)(aq, 0.05" molar")||M^(+)(aq, 1" molar")|M_((s)) For the above electroltic cell, the magnitude of the cell potential |E_(cell)|=70 m V. For the above cell : |
|
Answer» `E_(cell) lt 0, DeltaG gt 0` `M_((s))+M_((aq 1M))^(+) to M_((aq. 0.05 M))^(+)+M_((s))` According to Nernst equation, `E_(cell)=E_(cell)^(@)-(0.059)/(n)"LOG"([M^(+)]_((0.05" M")))/([M^(+)]_([1M]))` `=E_(cell)^(@)-(0.059)/(1)log(0.05)" log "(5xx10^(-2))` `=-0.059[-2+"log "5]=-0.059[-2+0.6990]` =+ve `:. E_(cell)=+ve" or " E_(cell) gt 0` and `DeltaG=-nFE_(cell.)` `DeltaG=-nFE_(cell.)=-ve` THUS `E_(cell) gt 0, DeltaG lt 0`. |
|
| 35. |
The concentration of potassium ions inside a biological cell is at least twenty times higher than the outside. The resulting potential difference across the cell is important in serveral processes such as transmission of nerve impules and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is : M_((s))|M^(+)(aq, 0.05" molar")||M^(+)(aq, 1" molar")|M_((s)) For the above electroltic cell, the magnitude of the cell potential |E_(cell)|=70 m V. If the 0.05 molar solution of M^(+) is replaced by a 0.0025 molar M^(+) solution, then the magnitude of the cell potential would be : |
|
Answer» Solution :(c ) `E_(cell(2))-E_(cell)^(@)-(0.059)/(1)"log"((0.0025)/(1))` `=-0.059 log (0.0025)` `E_(cell(1))=70 mV` `implies (E_(cell(2)))/(E_(cell(1)))=(log(0.0025))/(log(0.05))=(-2.6)/(-1.3)~~2` `implies E_(cell(2))=70xx2=140 mV`. |
|
| 36. |
The concentration of potassium ions inside a bilological cell is at least twenty times higher than the outisde. The resulting potential difference across the cell is important in several process such as transmission of nerve impuses and maintaning the ion balance. A simple model for such a concentration cell involving a metal M is M(s)|M^(+)(aq,0.05" molar")||M^(+)(aq,1" molar")|M(s) For the above electrolytic cell, the magnitude of the cell potential |E_(cell)|=70mV. Q. For the above cell |
|
Answer» `E_(cell)lt0,DELTAGGT0` `E_(cell)=(2.303RT)/(nF)"log"(C_(2))/(C_(1))` `=(2.303RT)/(F)"log"(1)/(0.05)=+ve,` i.E., `E_(cell)gt0`. For the reaction to be spontaneous, `DeltaG lt 0`. |
|
| 37. |
The concentration of NaOH solution is 10^(-8)M . Find out the (OH^(-)) concentration |
|
Answer» `10^(-8)` Total `[OH^(-)] = 10^(-8) + 10^(-7) = 1.1 XX 10^(-7)` pOH = 7 - log 1.1 PH = 14-7 + log 1.1 `pH = 7+ log 1.1 gt 7`. |
|
| 38. |
The concentration of K^(+) inside a biological cell is at least 20 times higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is: M(s)|M^(+)(aq,0.05M)||M^(+)(aq,1M)|M(s) For the above galvanic cell the magnitude of the cell potential |E_("cell")|=70mV. If the 0.05 M solution of M^(+) is replaced by 0.0025 M M^(+) solution, then the magnitude of the cell potential would be |
|
Answer» 35 mV |
|
| 39. |
The concentration of KI and KCl in certain solution containing both is 0.001M each. If 20 mL of this solution is added to 20 ml of a saturated of AgI in water ? What will happen |
|
Answer» AgCl will be precipitated |
|
| 40. |
The concentration of Kl and KCl in a certain solution containing both is 0.001 M each . If 20 mL of this solution is added to 20 mL of a saturated solution of Agl in water . What will happen ? K_(sp) ofAgCl = 10 ^(-10) , K_(sq)ofAgI = 10^(-16) |
|
Answer» AGL will be PRECIPITATED |
|
| 41. |
The concentration of KI and KCl in centain solution containing both is 0.001 M each. If 20 ml of this solution is added to 20 ml of a saturated solution of AgI in water . What will happen ? |
|
Answer» AgCl will be precipitated |
|
| 42. |
The concentration of K^(+) inside a biological cell is at least 20 times higher than the outside. The resulting potential difference across the cell is important in several processes such as transmission of nerve impulses and maintaining the ion balance. A simple model for such a concentration cell involving a metal M is: M(s)|M^(+)(aq,0.05M)||M^(+)(aq,1M)|M(s) For the above galvanic cell the magnitude of the cell potential |E_("cell")|=70mV. For the above cell |
|
Answer» `E_("cell")lt0,DELTAGGT0` |
|
| 43. |
the concentration of hydroxide ion in a water sample is found to be 2.5 times 10^-6 M. Identify the nature of the solution. |
|
Answer» SOLUTION :The concentration of `OH^-` ION in a water sample is found to be `2.5 times 10^-6 M` `pOH=-log_10[OH^-]` `pOH=-log_10[2.5 times 10^-4]` `=-log_10[2.5]-log_10[10^-6]` `=-0.3979-(-6)` `=-0.3979+6` `pOH=5.6` pH=8.4, shows the nature of the solution is BASIC We know that, `pH=pOH=14` pH+5.6=14 pH=14-5.6 pH=8.4 |
|
| 44. |
The concentration of hydroxide ion in a water sample is found to be 2.5xx10^(-6)M. Identify the nature of the solution. |
|
Answer» SOLUTION :`[OH^-]=2.5xx10^(-6)M` `pOH=-LOG[OH^-]` `=-log(2.5xx10^(-6))` `pOH=5.6021` `PH+pOH=14` `PH=14-5.6021` `=9.3979` The solution is BASIC. |
|
| 45. |
The concentration of hydronium (H_(3)O^(+)) ion in water is |
|
Answer» ZERO `pOH + PH = 14, 7 + 7 = 14, ""[H^(+)][OH] = 10^(-14)` `10^(-7) xx 10^(-7) , [OH^(-)] = 10^(-7)` gm ion/l. |
|
| 46. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. 100mLM//10HA (K_(a1) =8 xx 10^(-5)) and 200 mL of HB (K_(a2)=10^(-5)) acid mixture is diluted to one litre, then what will be the pH of the resulting solution ? |
|
Answer» 3 |
|
| 47. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. What will be the value of [H^(+)] of a 5 xx 10^(-6)M CH_(3)COOH solution (K_(a)=1.8 xx 10^(-5)) ? |
|
Answer» `sqrt(1.8 xx 10^(-5)xx10^(-6))` |
|
| 48. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. What is the sulphide ion concentration of a dilute solution that has been saturated with 0.1 M of H_(2)S, If pH of the solution is 3 ? (K_(a1)= 1.0 xx 10^(-7) , K_(a2) =1.3 xx 10^(-13)) |
|
Answer» `1.25 XX 10^(-17)` |
|
| 49. |
The concentration of hydrogen ions ina sample of soft drink is 3.8 times 10^-3m. What is the pH value? Whether the soft drink is acidic (or) basic? |
|
Answer» SOLUTION :`PH=-log_10[H_3O^+]` `=-log_10[3.8 times 10^-3]` `=-LOG 3.8 +3` `=3-0.5798=2.4202` `pH=2.42` When `pH lt 7`, the soft drink is acidic. |
|
| 50. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. What will be the [S^(2-)] and pH for 0.1 M H_(2)S solution respectively ? (Given that for H_(2)S, K_(a1)=1.0 xx 10^(-7), K_(a2)=1.3 xx 10^(-13)) |
|
Answer» `1.1 xx 10^(-11),5` |
|