Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. A basic mixture of 100mL of M//20 NaOH and 200mL of M//10 Ca(OH)_(2) is mixed with 200 mL of M//10 H_92)SO_94) and finally the whole mixture is diluted to 100mL then the pH of the resulting solution will be |
|
Answer» `12.0` |
|
| 2. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. How much volume of 10^(-2) M HCl should be added to 100mL of 10^(-2) M NaOH solution so that its pH changes by one unit ? |
| Answer» Answer :A | |
| 3. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. At temperature 150^(@)C, for pH =7, the nature of the solution will be |
|
Answer» BASIC |
|
| 4. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. 2g NaOH is added to 100mL of M//20 H_(2)SO_(4) solution and the resulting solution is obtained by additino of 900 mL of wter, then what will be the pH of the solution ? |
|
Answer» 12.6 |
|
| 5. |
The concentration of hydrogen ion is a measure of acidity or alkalinity of a solution. A convenient way of expressing the hydrogen ion concentration of a solution is, in terms of the pH scale devised by Sorenson's . The pH of a solutionis defined as the negative logarithm of active concentration of H^(+) ions to the base 10 i.e., pH =- log_(10)[HI^(+)] or [H^(+)]=10^(-pH) At 25^(@)C (i) if pH lt1 , then solution is acidic (ii) if pH=7, then solution is neutral (iii) if pH gt 7, then the solution is basic Total [H^(+)]and [OH^(-)] in a mixture of strong acids or bases are represented in terms normality, which is equal to final number of milliequivalents of H^(+) or OH^(-) present in millilitre of solution Since H_(2)O ionizes as H_(2)O hArrH^(+)+OH^(-) Ionic product of water , K_(w)=[H^(+)][OH^(-)] ...(i) Taking log on both sidesin equation (i), we find pH +pOH=pK_(w) For neutral solution, pH =pOH:. 2pH =pK_(w) :. pH =1.2pK_(w) Since dissociationof water is an endothermic process, so temperature willhave great effect on K_(w) as well as on the pH of solution i.e., the nature of the soluiton . K_(w) at different temperature are related as under. ln (K_(w2))/(K_(w1))=(DeltaH)/(R)[(1)/(T_(1))-(1)/(T_(2))] At T_(1)K=T_(2)K, K_(w_(2))=K_(w1) (i) pH mixture of monoprotic weak acid is calculated as under : pH of monoprotic weak acid i.e., CH_(3)COOH {:(,CH_(3)COOH,hArr,CH_(3)COO^(-),+,H^(+)),("at "t=0,C,,0,,0),("at "t=t,C-alpha,,C alpha,,Calpha):} :. K_(a)=(C alpha xx C alpha)/(C-Calpha)=(alpha^(2)C)/(1-alpha) When a le0.1 , 100 alpha =1-alpha=1 :. K_(a)=(alpha^(2)C)/(1) [When (1-alpha)=1] alpha=sqrt((K_(a))/(C)):. [H^(+)]=alphaC=sqrt(K_(a)xxC) (ii) pH of a mixture of monoprotic weak acids is calculated as follows [H^(+)]=sqrt(K_(a1)C_(1)+K_(a2)C_(2)). where K_(a1) and K_(a2) are dissociation constants of monoprotic weak acids HA and HB acid C_(1) and C_(2) are their concentrations respectively. (ii) pH of polyproticweak acid, say H_(3)A-a triprotic weak acid having dissociation constants K_(a1).K_(a2) and K_(a3) where K_(a1) gt gt K_(a2) gt gt K_(a3). Then in that case , maximum [H^(+)] will be contributed from step I, and neglibily small from concentration of species producing from step III will be negligible with respect to steop II and similarly concentration of species producig from step II will be negligible with respect to step I. Based upon the above discussion, answer the following questions. On decreasing the temperature , the pH of the solution |
|
Answer» will INCREASE |
|
| 6. |
The concentration of hydrogen ion in water is |
| Answer» Solution :`[H^(+)][OH^(-)] = 10^(-14), (10^(-7))(10^(-7)) = 10^(-14)`. | |
| 7. |
The concentration of hydrogen ion [H^(+)] in 0.01 M HCl is |
|
Answer» `10^(12)` `Hcl H^(+) + CL^(-)`. |
|
| 8. |
The concentration of H_(2)SO_(4) in a botal labelled ''conc. Sulphuric acid'' is 18 M. The solution has a density of "1.84 g cm"^(-3). What is the mole fraction and weight percentage of H_(2)SO_(4) in this solution? |
|
Answer» `"Solvent "=1840-1764 = 76g` `"Mole FRACTION of "H_(2)SO_(4)=(18)/(76//18+18)=0.81" ,Weight "%=(1764)/(1840)xx1000=95.87.` |
|
| 9. |
The concentration of H_(2)SO_(4) in a bottle labelled ''conc. Sulphuric acid '' is 18 M. The solution has a density of 1.84gcm^(-3). What is the mole fraction and weight percentage of H_(2)SO_(4) in this solution? |
|
Answer» Solution :`"Conc. = 18 mol L"^(1)`. SOLUTE = 18 moles `=18xx98g=1764g`, Solution = 1000 `cm^(3)` = 1840 g. Solvent = `1840 - 1764 = 76 g` `"Mole FRACTION of "H_(2)SO_(4)=(18)/(76//18+18)=0.81",WEIGHT"%=(1764)/(1840)xx1000=95.87.` |
|
| 10. |
The concentration of H^(+) ion in a sample of soft drink is 3.8xx10^(-3)M. Its pH is : |
|
Answer» `3.42` `=-[log3.8+log10^(-3)]` `=-[(0.58)+(-3.0)]=2.42` |
|
| 11. |
The concentration of [H^(+)] and concentration of [OH^(-)] of a 0.1 aqueous solution of 2% ionised weak acid is [Ionic product of water = 1 xx 10^(-14)] |
|
Answer» `2 xx 10^(-3) M` and `5 xx10^(-12)M` |
|
| 12. |
The concentration of glucose (in g/litre) solution which is isotonic with a solution of urea containing 6 g per litre will be : |
|
Answer» Solution :For TWO non-electrolytic solution `C_(1) = C_(2)` `W_(1)/(M_(1) xx 1)=W_(2)/(M_(2) xx 1 ) ` `:. 6/(60 xx 1) = W_(2)/(180 xx 1 ) :. W_(2) = (180 xx 6)/60 =18` |
|
| 13. |
The concentration of [H^+] and concentration of [OH^(-) ] of a 0.1 aqueous solution of 2% ionised weak acid is ionic product of water = 1 xx10^(-14)] |
|
Answer» `2 xx 10^(-3)`M and` 5 xx 10^(-12)M` ( Asdegreeof dissociation=2 % = 0.02) hence ` [OH^-]= (10^(14))/(2 xx 10^(-3)) =5XX 10^(-12) M` |
|
| 14. |
The concentration of glucose (in g/litre)solution which is isotonic with a solution of urea containing 6 g per litre will be: |
|
Answer» 6 |
|
| 15. |
The concentration of fluoride, lead, nitrate and iron in a water sample from an underground lake was found to be 1000 ppb, 40 ppb, 100 ppm and 0.2 ppm, respectively. This water is unsuitable for drinking due to high concentration of |
|
Answer» Lead |
|
| 16. |
The concentration of electrolyte required to coagulate a given amount of As_(2)S_(3) solution is minimum in case of |
|
Answer» POTASSIUM SULPHATE |
|
| 17. |
The concentration of C_(4)H_(9)Cl (n-butyl chloride) at different times are given. Calculate the average rate for the hydrolysis of n-butyl chloride. C_(4)H_(9)Cl + H_(2)O to C_(4)H_(9)OH + HCl |
Answer» SOLUTION :Average rate of reaction in the interval `t_(1)` to `t_(2)` = `({[C_(4)H_(9)Cl]_(t_(2))-[C_(4)H_(9)Cl]_(t_(1))})/(t_(2)-t_(1))` 
|
|
| 18. |
The concentration of electrolyte required to coagulate a given amount of As_(2)S_(3) sol is minimum in the case of |
|
Answer» magnesium nitrate `Al^(+3) GT Ba^(+2) gt K^(+)` |
|
| 19. |
The concentration of an organic compound in chloroform is 6.15" g per "100" mL" of the solution. A portion of this solution in a 5 cm polarimeter tube causes an observed rotation of -1.2^(@). What is the specific rotation of the compound ? |
|
Answer» Solution :Specific ROTATION `=(100xxalpha)/(lxxc)=(100xx(-1.2))/(0.5xx6.15)=-39^(@)(L=5" cm "=0.5" dm")` `ALPHA`= angle of rotation l = length of the tube in dm c = concentration of the solution in `g//100` ML |
|
| 20. |
The concentration of an organic compound in chloroform is 6.15 g per 100 mL of solution . A portion of this solution in a 5 cm polarimeter tube causes an observed rotation of -1.2^(@) . What is the specific rotation of the compound |
|
Answer» `+ 12^(@)` |
|
| 21. |
The concentration of an aqueous solution of 0.01 M CH_(3)OH solution is very nearly equal to which of the following |
|
Answer» `0.01% CH_(3)OH` |
|
| 22. |
The concentration of an aqueous solution of glucose is 10% W/V. How much solution is required to dissolve 1 mole glucose ? |
|
Answer» 18 litre x = 180 mL = 1.8 litre. |
|
| 23. |
The concentration of Ag^(+) ion in a given saturated solution of AgCl at 25^(@)C is 1.06xx10^(-5)g ion per litre. Thus, the solubility product of AgCl is : |
|
Answer» `0.353xx10^(-10)` `[Ag^(+)]=1.06xx10^(-5)` and `[Cl^(-)]=1.06xx10^(-5)` `K_(sp)=(1.06xx10^(-5))xx(1.06xx10^(-5))` `=1.12xx10^(-10)` |
|
| 24. |
The concentration of a solution is expressed in terms of mole fraction, molarity, molality andin____ |
| Answer» SOLUTION : PERCENTAGES | |
| 25. |
The concentration of a solution contaning 23g of phosphorous in 315g CS_(2) is 2.34 mol kg^(-1). The formula of phesphorous is (at mass of P=31) |
| Answer» ANSWER :C | |
| 26. |
The concentration of a reactant in a solution falls (i) from 0.2 to 0.1 M in 2 hrs (ii) from 0.2 to 0.05 M in 4 hrs. The order of hydrolysis of the reactant is |
|
Answer» zero |
|
| 27. |
The concentration of a reactant X decreases from 0.1 M to 0.025 M in 40 minutes. If the reaction follows I order kinetics, the rate of the reaction when the concentration of X is 0.01 M will be |
|
Answer» `1.73xx10^(-4)"M min"^(-1)` `=(2.303)/(40)log 20 = 0.075` Rate of the reaction when the concentration of X is 0.01 M will be `0.075xx0.01=7.5xx10^(-4)M min^(-1)`. Note : No OPTION is correct in the GIVEN question. |
|
| 28. |
theconcentrationof areactantin a solutionfalls(i)from0.2 to 0.1 M in2 hrs,(ii )from0.2to 0.05 m in4 hrs, theorderof thehydrolysisof thereactant is |
|
Answer» zero |
|
| 29. |
The concentrationof a reactantdecreasesfrom0.2M to 0.1M in10minutes. Therateof thereactionis ______. |
|
Answer» `0.01 M` `0.01` mol `dm^(-3) "min"^(-1)`. |
|
| 30. |
The concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes . The average rate of reaction using time in seconds is : |
|
Answer» `0.4xx10^(-3) "mol L"^(-1) s^(-1)` `=0.4 xx10^(-3) "mol L"^(-1) min^(-1)` `=0.4xx10^(-3) "mol L"L^(-1) min^(-1) `min /60 s `=0.66 xx10^(-4) "mol L"^(-1) s^(-1)` |
|
| 31. |
The concentration of a 100 ml solution containing xg of Na_(2)CO_(3) (molecular wt=106) is YM.The value of X and Y are respectively |
|
Answer» 2.12 , 0.05 |
|
| 32. |
The concentration in g//L of a solution of cane sugar (Molecular weight = 342) which is isotonic with a solution containing 6 g of urea (Molecular weight = 60) per litre is |
|
Answer» `3.42` `=(w_(1))/(342xx1)=(6)/(60xx1)=(342xx6)/(60)=34.2`. |
|
| 33. |
The concentraion of free HN_3 in a 0.01 M solution of KN_3 if K_a=1.9xx10^(-5) is : |
|
Answer» 2.3 mol/mL |
|
| 34. |
The concentration in gms per litre of a solution of cane sugar ( M =342) which is isotonic wih a solution containing 6 gms of urea ( M = 60 ) per litreis |
|
Answer» 3.42 `m_(2) = m_(1)` `(g_(2))/( M_(2)) =(g_(1))/( M_(1))` `( X )/( 342) =( 6)/( 60)` |
|
| 35. |
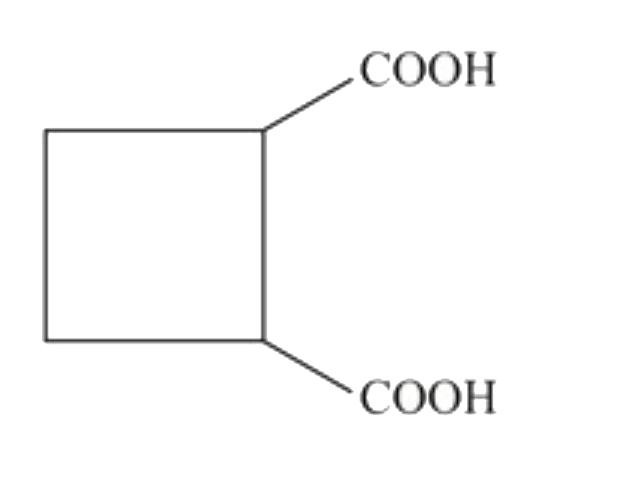
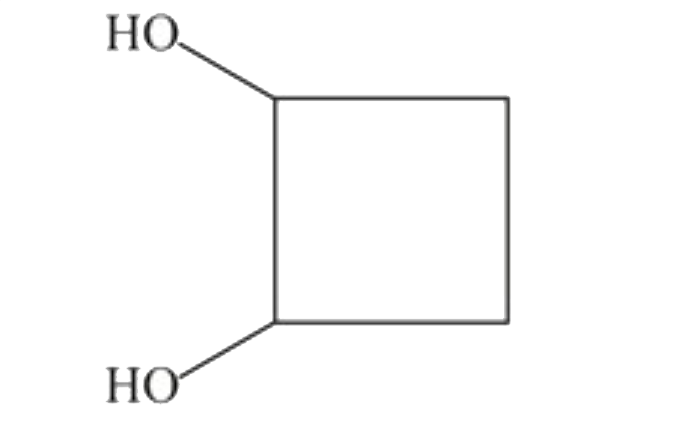
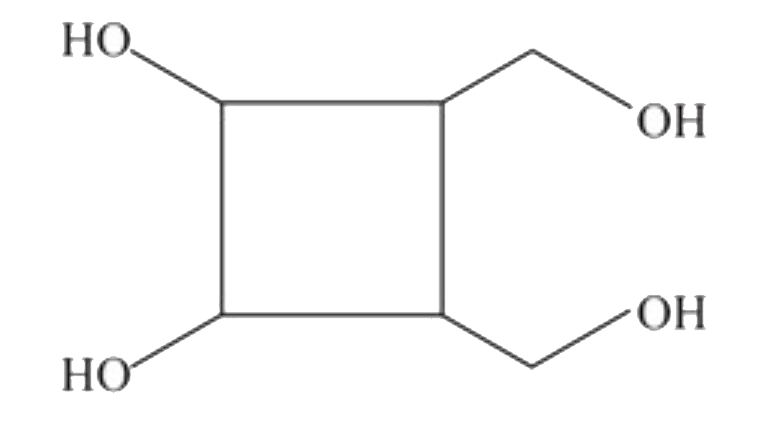
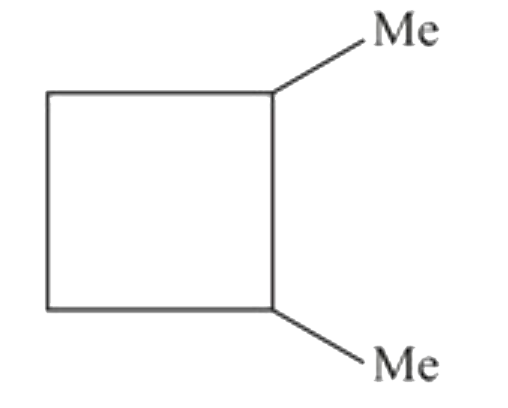
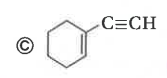
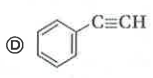
The compund 'A' is |
|
Answer»
|
|
| 36. |
The compression factor (compressibility factor) for one mole of a van der Waals' gas at 0^(@)C and 100 atmosphere pressure is found to be 0.5. Assuming that the volume of a gas molecule is negligible, calculate the van der Waal's constant 'a'. |
|
Answer» Solution :`Z=(PV)/(RT) =0.5` `therefore (100xxV)/(0.082xx273)=0.5` `therefore V=0.112` litre Now, USING van der Waals. equation `[P+(a)/(V^(2))][V]=RT` or `[P+(a)/(V^(2))]=(RT)/(V)" "(therefore` b is negligible) `therefore [100+(a)/((0.112)^(2))]=(0.082xx273)/(0.112)` `=199.88` `therefore (a)/((0.112)^(2))=99.88` `therefore a=1.253" litre"^(2)" mole"^(-2)` atm. |
|
| 37. |
The compressibility of a gas is less than unity at S.T.P. Therefore : |
|
Answer» `V_m` GT 22.4 LITRES |
|
| 38. |
The compressibility factor (Z) of one mole of a va der Waals gas of negligible a value is |
|
Answer» 1 `(P+a/(V^(2)))(V-b)=RT` If a is negligible then `a/(V^(2))~~0` `:.P(V-b)=RTimpliesPV-PB=RT` `IMPLIES(PV)/(RT)-(Pb)/(Rt)=1impliesZ=1+(Pb)/(RT)` (Z, COMPRESSIBILITY factor `=(PV)/(RT)`) |
|
| 39. |
The compressibility factor for N_(2) at - 50^(@)C and 800 atmp pressure is 1.95 and at 100^(@)C and 200 atmp, it is 1.10. A certain mass of nitrogen occupied one litre at - 50^(@)C and 800 atmp. Calculate the volume occupied by the same quantity of N_(2) at 100^(@)C and 200 atmp. |
|
Answer» |
|
| 40. |
The compressibility factor of an ideal gas is : |
|
Answer» 0 |
|
| 41. |
The compressibility factor of gases is less than unity at STP. Therefore, |
|
Answer» `vm GT 22.4 ` LITRES |
|
| 42. |
The compressibility factor of a gas is defined as Z =(PV)/Nrt. The compressibility factor of an ideal gas is : |
|
Answer» zero |
|
| 43. |
The compressibility factor for definite amount of vander walls gas at 0^(@)C and 100 atm is found to be 0.5. Assuming the volume of gas molecules negligible the vander Waal's constant 'a' for gas in "litre"^(2)" mol"^(-2) atm is : |
|
Answer» 1.256 `(P+(n^(2)a)/(V^(2))(V-nb)=nRT)` `RARR (P+(n^(2)a)/(V^(2))(V) =nRT` (b is negligible) `therefore PV^(2) -nRTV +n^(2)a=0` `V =(nRT pm SQRT(n^(2)R^(2)T^(2)-4n^(2)aP))/(2P)` Since, V is constant at given P and T THUS discriminant is zero `therefore n^(2)R^(2)T^(2)=4n^(2) aP` `therefore |
|
| 44. |
The compressibility factor for H_2 and He is usally : |
|
Answer» GT 1 |
|
| 45. |
Thecompressedgasaviailableincookinggascylinders isa mixtureof : |
|
Answer» `C_(6) H_(6) +C_(6) H_(5) CH_(3) ` |
|
| 46. |
The compoung givenbelow is not antipyretic |
|
Answer» CHLORAMPHENICOL |
|
| 47. |
The compounds with maximum and least ionic characters among the following are: |
|
Answer» `LICL and RBCL` |
|
| 48. |
The compounds with highest ionic character in metal halides, highest stability in halogen acids and highest acidic strength in halogen acids respectively are : |
|
Answer» MI, HCl, HF |
|
| 49. |
The compounds with high heat of formation are less stable because |
|
Answer» HIGH temperature is required to SYNTHESISE them |
|