Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The compounds which produce yellow precipitate with I_(2)//NaOH are- |
|
Answer» `ICH_(2)COCH_(2)CH_(3)` |
|
| 2. |
The compounds which gives the most stable carbonium ion on dehydration is : |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)OH` |
|
| 3. |
The compound(s) which generate (s) N_(2), gas upon thermal decomposition below 300^(@)C is (are) |
|
Answer» `NH_(4)NO_(3)` |
|
| 5. |
The compounds which exist as liquids are - |
|
Answer» `C_5H_(12)` |
|
| 6. |
The compounds which contains all the four 1^(@), 2^(@), 3^(@) and 4^(@) carbon atoms is |
|
Answer» 2,3-dimethylpentane  Thus all the FOUR TYPES of carbon atoms are PRESENT in this compounds. |
|
| 7. |
The compounds which can be purified by steam distillation. |
|
Answer» Nitrobenzene |
|
| 8. |
The compounds which are coloured only due to charge transfer are |
|
Answer» `KMnO_4` |
|
| 9. |
The compounds which are coloured only due to change transfer are |
|
Answer» `KMnO_(4)` |
|
| 10. |
The compounds used to fix a dye to the fabric is known as |
|
Answer» MORDANT |
|
| 11. |
The compounds used in Solvay process are |
|
Answer» `Na_2SO_4` |
|
| 12. |
The compounds used as refrigerant is are |
|
Answer» `CH_(2)F_(2)` |
|
| 13. |
The compounds used as refrigerant are |
|
Answer» `NH_(3)` |
|
| 14. |
The compounds used as refrant are: |
|
Answer» `NH_(3)` |
|
| 15. |
The compounds that will give an isomer of 2, 2-dimethyl propane on catalytic hydrogenation are (1)CH_3CH=undersetunderset(CH_3)|C-CH_3 (2)CH_3CH=CHCH_3 (3)CH_3CH=CHCH_3 (4)CH_3undersetunderset(CH_3)|C=undersetunderset(CH_3)|C-CH_3 |
|
Answer» 1 and 4 |
|
| 16. |
The compounds used as gaseous insulation in high voltage generators is |
|
Answer» `SeF_(4)` |
|
| 17. |
The compounds used as refrigent are |
|
Answer» `NH_(3)` |
|
| 18. |
The compounds that exhibit(s) geometrical isomerism is (are): |
|
Answer» `[Pt(en)Cl_(2)` |
|
| 19. |
The compounds that gives white precipitates with AgNO_(3). |
|
Answer» `[Co(NH_(3))_(6)]Cl_(3)` `[Fe(en)_(3)]Cl_(3)+3AgNO_(3)toAgCldarr[Fe(en)_(3)](NO_(3))_(3)` The COMPLEX is which `CL^(-)` ions present outside the []can give white ppt with `AgNO_(3)` solution. |
|
| 20. |
The compounds that doesnot produce nitrogen gas by thermal decomposition is ......... |
|
Answer» `Ba(N_(3))_(2)` |
|
| 21. |
The compounds shown below are |
|
Answer» LINKAGE ISOMERS |
|
| 22. |
The compounds (s) which generated (s) N_2 gas upon thermal decomposition below 300^(@)C is (are) |
|
Answer» `NH_(4)NO_(3)` `(b) (NH_(4))_(2) Cr_(2)O_(7) overset(Delta)to N_(2)+ Cr_(2)O_(3) + 4H_(2)O` `(c) Ba(N_(3))_(2) overset(Delta)to Ba+ 3N_(2)` (d) `Mg_(3)N_(2) ` (it does not decompose into `N_(2)`) |
|
| 23. |
The compounds soluble in water |
|
Answer» METHYL amine |
|
| 24. |
The compounds (s) that can be obtained by vapour phase nitration of propane is |
|
Answer» 2-Nitropropane |
|
| 25. |
The compounds of Zn, Cd and Hg are usually white. (ii) A dark blue precipitate is formed when NaOH solution is added to CuSO_(4) |
|
Answer» Solution : In the compounds of zinc metals, `M^(2+)` ions possess the penultimate d-orbitalsdoubly OCCUPIED, i.e. `(n-1)d^(10)` configuration. There is no d-d transition. Hence the compounds of zinc metals are colourless (i) NaOH reacts with `CuSo_(4)` when dark blue PRECIPITATE of `Cu(OH)_(2)` is FORMED. This precipitate of heating forms CuO which is BLACK in colour. Hence, the colour darkens on heating `CuSO_(4) + 2NaOH to Cu(OH)_(2) + Na_(2) SO_(4)` `Cu(OH)_(2) tounderset(Black)(CuO) + H_(2)O` |
|
| 26. |
The compounds [PtCl_(2)(NH_(3))_(4))]Br_(2) and [PtBr_(2)(NH_(3))_(4)]Cl_(2) constitutes a pair of |
|
Answer» COORDINATION isomers |
|
| 27. |
The compounds P and Q were separately subjected to nitration using HNO_(3)//H_(2)SO_(4) mixture. The major product formed in each case respectively is |
|
Answer»
 `-OH` is ctivating and o-, p-directing, whereas `-COOH` is deactivating and m - directing.  Since `-OCH_(3)` is more activating, the SUBSTITUTION OCCURS at a position ortho to it. |
|
| 28. |
The compounds obtained by the substitution of hydrogen by halogen in alkane series is …….. |
|
Answer» Aryl halides |
|
| 29. |
The compounds HCHO and CH_(3) CHO on intermolecular aldol condensation gives . |
|
Answer» `HO - CH_(2) - CH_(2) - CHO` |
|
| 30. |
The compounds having a common difference of CH_(2) in their molecular formulae are known as : |
|
Answer» ISOMERS |
|
| 31. |
The compounds formed when excess of Cl_(2), reacts with NH_3 are |
|
Answer» `NCl_(3)+N_(2)` |
|
| 32. |
The compounds given are: Which of the following statements is/are correct ? |
|
Answer» Compounds (A) and(B) are (I) and (II), respectively. |
|
| 33. |
The compounds formed on heating chlorobenzene with chloral in the presence of concentrated sulphuric acid, is |
|
Answer» freon 
|
|
| 34. |
The compounds formed by the acidby the acid hydrolysis of ethyl acetate are |
|
Answer» formic acid and propanol |
|
| 35. |
The compounds [Cr(H_(2)O)_(6)]Cl_(3),[Cr(H_(2)O)_(5)Cl]Cl_(2)H_(2)O and [Cr(H_(2)O)_(4)Cl_(2)]Cl*2H_(2)O exhibits: |
|
Answer» LINKAGE isomerism |
|
| 36. |
The compounds [Cr(H_(2)O)_(6)]Cl_(3) and : [Cr(H_(2)O)_(5)Cl]Cl_(2). H_(2)O are example of |
|
Answer» Linkage ISOMERISM |
|
| 37. |
The compounds [Co(SO_(4))(NH_(3))_(5)]Br and [Co(SO_(4))(NH_(3))_(5)]Cl represent |
|
Answer» LINKAGE ISOMERISM |
|
| 38. |
The compounds [CO(SO_(4)NH_(3))_(5) Br] and [[CO(SO_(4)NH_(3))_(5) ]Cl represent. |
|
Answer» LINKAGE isomerism |
|
| 39. |
The compounds [Co(SO_(4))(NH_(3))_(5)]Br and [Co(SO_(4))(NH_(3)) _(5)]Cl represent |
|
Answer» LINKAGE isomerism. |
|
| 40. |
The compounds [Co(NO_(2))(NH_(3))_(5)]Cl_(2) and [Co(ONO)(NH_(3))_(5)]Cl_(2) are examples of : |
|
Answer» Geometrical isomers |
|
| 41. |
The compounds [Co(NO_2)(NH_3)_5]Cl_2 and [Co(ONO)(NH_3)_5]Cl_2 are examples of: |
|
Answer» GEOMETRICAL isomers |
|
| 42. |
The compounds CH_(3)-O-C_(3)H_(7) and C_(2)H_(5)-O-C_(2)H_(5)exhibit |
|
Answer» OPTICAL isomerism |
|
| 43. |
The compounds CH_(3)CH = CHCH_(3) and CH_(3)CH_(2)CH = CH_(2) |
|
Answer» Are tautomers |
|
| 44. |
The compounds CH_3NH_2 and CH_3CH_2. NH_2 are : |
|
Answer» Isomers |
|
| 45. |
The compounds CH_(3)COCH_(3)andCH_(2)=CH(OH)CH_(3) represent : |
|
Answer» FUNCTIONAL isomerism |
|
| 46. |
The compounds CH_3-O-C_3H_7 and C_2H_5-O-C_2H_5 exhibit |
|
Answer» metamerism |
|
| 47. |
The compound(s) behaving both oxidizing as well as reducing agent is/are |
|
Answer» `H_(2)O_(2)` |
|
| 48. |
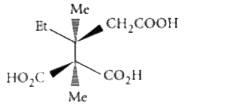
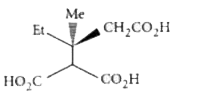
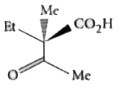
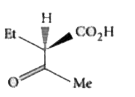
The compound(s), capable of producing achiral compound on heating at 100^(@)C "is" // "are" |
|
Answer»
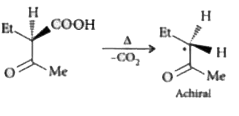
|
|
| 50. |
The compounds 'A' undergoes Koble's reaction in the presence of CO_(2) and NaOH to give compound 'B' . Compound 'B' when treated with acetic anhydride in acidic medium , gives acetylsalicylic acid i.e. aspirin. Identify the compound 'A' and 'B'. |
|
Answer» SALICYLADEHYDE, SALICYLIC ACID |
|