Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The central dogmaof molecular genetics statesthatthe geneticinformationflows from ………. . |
|
Answer» AMINO acids Protein DNA |
|
| 2. |
The central dogma of molecular genetics states that the genetic information flows from |
|
Answer»
|
|
| 3. |
The central carbon atom of a free radical contains |
|
Answer» 6 electrons |
|
| 4. |
The central atom does not assume sp^(2) hybridisation in |
|
Answer» `PCl_(3)` |
|
| 5. |
The celll reaction for the given cell is spontaneous if: Pt|Cl_(2)(P_(1) atm)| Cl (1M) | Cl_(2)(P_(2) atm)|Pt |
|
Answer» `P_(1) GT P_(2)` |
|
| 6. |
The cells having membrane bound nucleus are called: |
|
Answer» Eukaryotic |
|
| 7. |
The cell, Zn|Zn^(2+)(1M)||Cu^(2+)(1M)|Cu(E_(cell)^(o)=1.10V) was allowed to be completely discharged at 298K. The relative concentration of Zn^(2+) to Cu^(2+)[([Zn^(2+)])/([Cu^(2+)])] is |
|
Answer» Antilog (24.08) where, `Q=([ZN^(2+)])/([Cu^(2+)])` For COMPLETE discharge `E_(cell)=0` So, `E_(Cell)^(0)=(0.0591)/(2)"log"([Zn^(2+)])/([Cu^(2+)])` `implies|([Zn^(2+)])/([Cu^(2+)])|=10^(37.3)` |
|
| 8. |
The cell, Zn|Zn^(2+)(1M)||Cu^(2+)(1M)|Cu,(E_(cell)=1.10V), was allowed to be completely discharged at 298K. The realtive concentration of Zn^(2+) to Cu^(2+),((Zn^(2+)])/([Cu^(2+)])) is |
|
Answer» `9.65xx10^(4)` i.E., `1.10=(0.591)/(2)"log"([Zn^(2+)])/([Cu^(2+)])` or `([Zn^(2+)])/([Cu^(2+)])=37.3` or `([Zn^(2+)])/([Cu^(2+)])=10^(37.3)` |
|
| 9. |
The cell Zn | Zn^(2+)(1 M)||Cu^(2+)(1 M)|Cu E_("cell")^(@)=1.10 Vwas allowed to be copletely discharged at 298 K The relative concentration of (Zn^(2+))/(Cu^(2+)) is |
|
Answer» antilog (24.08) `ZN|Zn^(2+) (1M)||CU^(2+)(1 M)|Cu` `k_(eq)=[(Zn^(2+))]/[(Cu^(2+))]` `log k_("eq") =(E_("cell")^(@))/(0.591)=(2xxx1.1)/(0.591)=37.2` `k_("eq") =10^(37.2)` |
|
| 10. |
The cell , Zn//Zn^(2+) (1M) //Cu^(2+) (1M) // Cu (E_("Cell")^@ = 1.10V),was allowed to be completely discharged at 298 K. The relative concentration of Zn^(2+) to Cu^(2+) is |
|
Answer» 37.3 |
|
| 11. |
The cell Zn//Zn^(2+) (1 M) ||Cu^(2+)(1M)//Cu(E_("cell")^(0) = 1.10 V)was allowed to be completely discharged at 298 K. The relative concentration of Zn^(2+) to Cu^(2+) (([Zn^(2+)])/([Cu^(2+)])) is 10^(x). The value of x is : (take (2.303 RT)/(F) = 0.059 round off your answer up to one decimal) |
|
Answer» |
|
| 12. |
The cell voltage depends on _______. |
|
Answer» NATURE of the electrodes |
|
| 13. |
The cell voltage of a galvanic cell becomes zero after using for some tme because |
|
Answer» All the FREE ELECTRONS are used up `THEREFORE` when current is DRAWN the conc. Of eec trolytic solution changes and hence the reduction potential. |
|
| 14. |
The cell reaction of the galvanic cell Cu_((s))|Cu_((aq))^(2+)||Hg_((aq))^(2+)|Hg_((l)) is |
|
Answer» `Hg+CU^(2+)toHg^(2+)+Cu` 
|
|
| 15. |
The cell reaction Zn(s) + Cu^(+2)rarr Zn^(+2) + Cu(s) is best represented as : |
|
Answer» `Cuabs(Cu^(+2))ABS(ZN^(+2))Zn` |
|
| 16. |
The cell that was used as the primary source of electrical energy on the appolo moon flights is |
|
Answer» VOLTAIC cell |
|
| 17. |
The cell reaction ,Zn + Cu^(2+)rarrZn^(2+) +Cu is best represented by : |
|
Answer» `CU//Cu^(2+)||ZN^(2+)//Zn` |
|
| 18. |
The cell reaction of the galvanic cell Cu(s)|Cu^(2+)(aq)||Hg^(2+)(aq)|Hg(l) is |
|
Answer» `Hg+CU^(2+)rarrHg^(2+)+cu` in theabove cell oxidatin of COPPER and redcution of mercury takes place its cell reactionis writtenas `Cu(s) +Hg^(2+) (aq) rarr Cu^(2+) (aq) +Hg` |
|
| 19. |
The cell reaction of a cell is Mg+Cu^(2+) to Mg^(2+)+Cu (Given E_(Mg^(2+)//Mg) = -2.37 V , E_(Cu^(2+)//Cu) = 0.337 V) The e.m.f of the cell will be _________. |
|
Answer» `-2.7 V` |
|
| 20. |
The cell reaction of the galvanic cell, Cu(s) // Cu^(2+)(aq) // Hg^(2+) (aq) // Hg (l)is |
|
Answer» `HG + CU^(2+) to Hg^(2+) + Cu` |
|
| 21. |
The cell reaction of the cell fuel cell used in the space program is |
|
Answer» `C(s)+O_(2)(g) rarr CO_(2)(g)` Liquid `H_(2)` is carried on board a s a propellant. The bolied-off `H_(2)` vapor that ordinarily WOULD be lost is used in a fuel cellto generate electrical power. Hydrogen (the fuel is supplied to the anode compartment. Oxygen (the oxidizer) is fed into the cathode compartment. The diffusion rates of the gases into the cell are carefully regulated for maximum efficiency. Oxygen is reduced at the cathode, which consists of porous carbon impregnated with a finely divided `Pt` o `Pd` CATALYST. Cathode `O_(2)(g)+2H_(2)O(l)+4e^(-)overset("Catalyzing") rarr 4OH^(-)(aq.)` The `OH^(-)` ions migrate through the electrolyte (an aqueous solution of a base) to the anode. The anode is also porous cabon containing a small amount of catalyst (`Pt, Ag`, or `CoO`). Here `H_(2)` is OXIDIZED to `H_(2)O`. Anode `H_(2)(g)+2OH^(-)(aq.) rarr 2H_(2)O(l)+2e^(-)` The net reaction obtained from the two half-reactions is `2H_(2)(g)+O_(2)(g) rarr 2H_(2)O(l)` The pure water produced by the cell is drunk by the astronauts. |
|
| 22. |
The cell reaction for the given cell is spontaneous if :Pt(H_2)|H^=(1M)||H^+(1M)|Pt(H_2) |
|
Answer» `P_1 GT P_2` |
|
| 23. |
The cell reaction for the given cell is spontaneous if :Pt | Cl_2|CI^(-) (1M)||CI^(-) (1M)|Cl_2| Pt |
|
Answer» `P_1 GT P_2` |
|
| 24. |
The cellreactionfor agalvantcell isH_(2(g)) +C1_(2(g))to 2HC1_((aq)) Constructthe cell andalsocalculatestandardemf of cell E_(C1)^(@) = 1.36 V |
|
Answer» Solution :Cellfor the GIVENREACTION `Pt, H_(2(g))(1 ATM) | underset(1 M) (HC1_((aq))) | C1_(2(g))( 1 atm),Pt , E_(" cell ")^(@)= 1.36 V` |
|
| 25. |
The cell reaction for the given cell is :Pt(H_2)|pH=2||pH=3|Pt(H_2) |
|
Answer» SPONTANEOUS |
|
| 26. |
The cell, Pt|H_(2)" (1 atm)"||H^(+) (pH=x)| normal calomel electrode, has an emf of 0.67 volt as 25^(@)C. Calculate the pH of the solution. The oxidation potential of calomel electrode on the hydrogen scale is -0.28 volt. |
|
Answer» |
|
| 27. |
The cell reaction Cu+2Ag^(+)toCu^(+2)+Ag is best represented by |
|
Answer» `Cu_((s))|Cu_((AQ))^(+2)||Ag_((aq))^(+)|AG` |
|
| 28. |
The cell Pt, H_(2) (1atm) H^(+) (pH =x)| Normal calomel Electrode has an EMF of 0.67V at 25^(@)C .Calculate the pH of the solution. The oxidation potential of the calomel electrode on hydrogen scale is -0.28V. |
|
Answer» `0.67 =0.28 +(0.0591)/(1) XX x` `x = 6.61` |
|
| 29. |
The cell potential for the unbalanced chemical reaction: Hg_(2)^(2+)+NO_(3)^(-)+H_(3)O^(+) to Hg^(2+) +HNO_(2)+H_(2)O+e^(-) Is measured under standard conditions in the electrochemical sell shown in the diagram were Choose the correct statement for the given cell diagram |
|
Answer» Compartment (X) has less pH than compartment (Y) |
|
| 30. |
The cell potential of the galvanic cell Zn|Zn^(++)|Ag^(+)|Ag , E^(@)(Zn^(++)|Zn)=-0.76 V, E^(@)(Ag^(+)|Ag)=+0.80 V |
|
Answer» `+ve` |
|
| 31. |
The cell potential for the unbalanced chemical reaction: Hg_(2)^(2+)+NO_(3)^(-)+H_(3)O^(+) to Hg^(2+) +HNO_(2)+H_(2)O+e^(-) Is measured under standard conditions in the electrochemical sell shown in the diagram were Equilibrium constant for cell reaction is : |
|
Answer» `10^(3//2)` `log K_(c)=(2 XX 0.02)/(0.06) =2/3` `=10^(2/3)` |
|
| 32. |
The cell potential of the galvanic cell, Zn | Zn^(2+) ||Ag^(+) | Ag, whereE_(Zn^(2-)pin)^(@)= -0.76V,E_(Ag'//Ag)^(@) =+0.8V is |
| Answer» Answer :C | |
| 33. |
The cell membranes are mainly composed of : |
|
Answer» FATS |
|
| 34. |
The cell potential (E_("cell")) of a reaction is related as DeltaG = -nFE_("cell") , where DeltaG represents max. useful electrical work . n=no. of molesof electrons exchanged during the reactino of reversiblecell reaction d(DeltaG) = (DeltaV)dP-(DeltaS).dT at constant pressured(DeltaG) = - (DeltaS).dT :' At constant pressure DeltaG = DeltaG = DeltaH-TDeltaS.......(1) :. DeltaG = DeltaH + T((d(DeltaG))/(dT))_P ......... (B) ((dE_("cell"))/(dT))_(P) is known as temperature coefficient of the e.m.f of the cell When DeltaS increases, temperature coefficient of the emf of cell |
|
Answer» Constant ` IMPLIES -DeltaS = ((d(DeltaG))/(dT))_P - DeltaS = ((d(-nFE_("cell")^(0)))/(dt))_(P)- DeltaS = -NF((dE_("cell"))/(dt))_(P)` `((dE_("cell"))/(dt))_P = (DeltaS)/(nF) implies If DeltaS uarr ` then `((dE_("cell"))/(dt))_(P)uarr` |
|
| 35. |
The cell potential E_("cell") is ______ |
|
Answer» `E_("cell")^(@)=E_("CATHODE")^(@)-E_("anode")^(@)` |
|
| 36. |
The cell membranes are mainlycomposed of ………. . |
|
Answer» carbohydrates |
|
| 37. |
The cell membranes are following statements is not true |
|
Answer» Buna-S is a copolymer of BUTADIENE and styrene |
|
| 38. |
The cell in which the following reactions occurs : 2Fe_((aq))^(3+)+2l_((aq))^(-) to 2Fe_((aq))^(2+)+I_(2(S)) has E_(cell)^(Theta)=0.236V at 298 K. calculate the standard Gibbs energy and the equilibrium constant of the cell reaction. |
Answer» Solution :(a) Cell reaction and calculation of `Delta_(R)G^(Theta)`:  Where, n=2 MOL, `F=96487" C "mol^(-1)`, `E_(cell)^(Theta)=0.236V` `THEREFORE Delta_(r)G^(Theta)=-nFE_(cell)^(Theta)` `=-(2mol)(96487" C "mol^(-1))(0.236V)` `=-45542(V)` `=-45542J` =-45.542 kJ `~~-45.54kJ` (b) Calculation of equilibrium constant `K_(c)`: `therefore Delta_(r)G^(Theta)=-2.303RT" log "K_(c)` `therefore -45542=-2.303xx8.314xx298xxlog" "K_(c)` `therefore log" "K_(c)=(-45542)/(-2.303xx8.314xx298)` `therefore log" "K_(c)=+7.9816` `thereforeK_(c)="Antilog"(7.9816)` `=9.586xx10^(7)` `~~9.6xx10^(7)` |
|
| 39. |
The cell in which the followingreactionoccurs : 2Fe_(aq)^(3+)+ 2I_(aq)^(-) to2Fe_(aq)^(2+) + I_(2(s)) "has"E_("cell")^(o) = 0.236 V "at" 298 K The equilibriumconstnat of the cell reaction is |
|
Answer» `6.69xx 10^(-7)` |
|
| 40. |
The cell in which the following reaction occurs: 2Fe^(3+)(aq)+2I^(-)(aq)to2Fe^(2+)(aq)+I_(2)(s) has E_(cell)^(@)=0.236V at 298K. Calculate the standard Gibbs energy and the equilibrium constant of the cell reaction. |
|
Answer» Solution :`2Fe^(3+)+2e^(-)to2Fe^(2+)` or `2I^(-)toI_(2)+2e^(-)` `Delta_(r)G^(@)=nFE_(cell)^(@)=-2xx96500xx0.236J=-45.55kJ" "MOL^(-1)` `Delta_(r)G^(@)=-2.303RT" LOG "K_(c)` or `"log "K_(c)=(Delta_(r)G^(@))/(2.303RT)=(-45.55" kJ "mol^(-1))/(2.303xx8.314xx10^(-3)" kJ "K^(-1)xx298K)=7.983` `thereforeK_(c)=`ANTILOG `(7.983)=9.616xx10^7` |
|
| 41. |
The cell in which the following reaction occurs 2Fe^(3+)(aq)+2I^(-)(aq)to2Fe^(2+)(aq)+I_(2)(s) has E_(cell)^(@)=0.236V at 298K. Calculate the standard gibbs energy of the cell reaction. (Given: 1F=96,500" C "mol^(-1)) |
|
Answer» |
|
| 42. |
The cell in which the following reaction occurs 2Fe^(3+)(aq)+2I^(-)(aq)to2Fe^(2+)(aq)++I_(2)(s) has E_(cell)^(0)=0.236V at 298 K. Calculate the standard Gibbs energy and the equilibrium constant of the cell reaction. |
|
Answer» Solution :`n=2` `DeltaG^(@)=-nFE_("CELL")^(@)=-2xx96500xx0.236J=-45.55kJ//mol` `DeltaG^(@)=-2.303"RT LOG K"_(c)` `logK_(c)=(DeltaG^(@))/(-2.303RT)=(45.55xx10^(3))/(2.303xx8.314xx298)=7.983` `K_(c)="antilog (7.982)"=9.616xx10^(7)` |
|
| 43. |
The cell in which electrical energy is used to bring about chemical change is known as ________ |
|
Answer» ELECTROLYTIC cell |
|
| 44. |
The cell in which of the following reaction occurs: 2Fe^(3+)(aq)+2I^(-)(aq) to 2Fe^(2+)(aq)+I_(2)(s)" has "E_("cell")^(0)=0.236V" at "298K Calculate the standard Gibbs energy and the equilibrium constant of the cell reaction. |
|
Answer» Solution :Data: `E_("cell")^(0)=0.236V, T=298K, N=2, F=96500, R=8.314J//K MOL` FORMULA 1 . `DeltaG^(0)=-n E_("cell")^(0)F` `=-2 xx 0.236 xx 96500 =-45.55kJ` Formula 2. `DeltaG^(0)=-2.303 RRT log K_(C)` `-45.55=-2.303 xx 8.314 xx 298 xx log K_(C)` `log k_(c)=(4550)/(2.303 xx 8.314 xx 298)` `log k_(c)=7.983` `k_(C)="Antilog "7.983=9.62 xx 10^(7)` |
|
| 45. |
The cell for which the cell reaction is H_2 + Cu^(2+) to 2H^(+) + Cu is represented as |
|
Answer» `Cu//Cu^(2+)//H^(+)//H_(2)` |
|
| 46. |
The cell EMF is independent of the concentration of the species of the cell in |
|
Answer» `Feoverset(c-)(|)FEO(s)|KOH(aq)|NiO(s)|Ni_(2)O_(3)(s) overset(o+)(|)Ni` 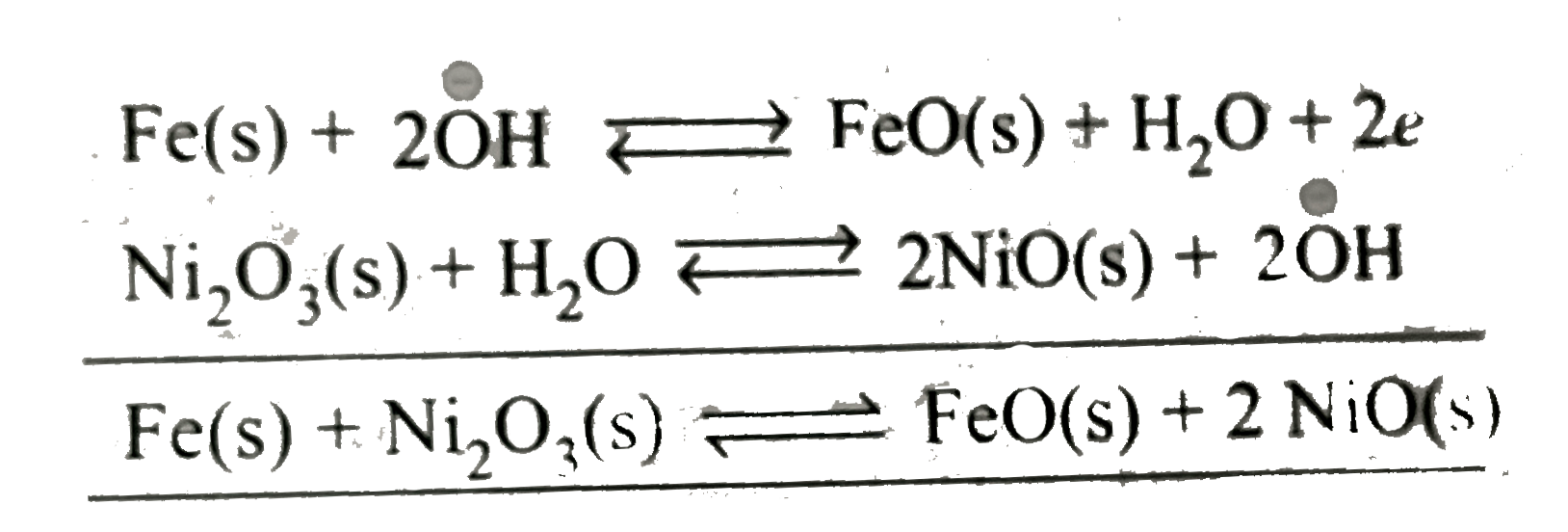 HENCE, `EMF` is independent of `overset(c-)(O)H` ions. |
|
| 47. |
The cell emf depends on |
|
Answer» Temperature |
|
| 48. |
The cell emf in a cell with cell reaction Cr_2O_7^(2-) + 14H^(+) + 6Fe^(+2) to 2Cr^(+3) + 6Fe^(+3) + 7H_2O could be increased above the standard emf by |
|
Answer» Increasing `[Cr^(+3)]` |
|
| 49. |
The cell emf is independent of concentration of the species of the cell in: |
|
Answer» `Fe|FeO(s)|KOH(aq)|NiO(s)|Ni_(2)O_(3)|Ni` |
|
| 50. |
The cell edge of a fcc crystal is 100 pm and its density is 10.0 g cm^(-3) . The number of atoms in 100 g of this crystal is: |
|
Answer» `1 xx 10^(25)` `= 10^(-24) cm^(3)` Density `= 10.0 G cm^(-3)` Mass of unit cell `= 10^(-24) xx 10` `= 10 ^(-23) g ` No. of unit cells in 100 g `= ( 100)/( 10^(-23))= 10^(25)` Since the lattice is fcc and each unit cell has 4 atoms PER unit cell. No. of atoms `= 4 xx 10^(25)` |
|