Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The cell constant of a conductivity cell is given by |
|
Answer» `lxxa` |
|
| 2. |
The cell content (Z) of the unit cell of a h.c.p. space lattice is |
|
Answer» 2 
|
|
| 3. |
The cell constant of a conductivity cell is ________. |
|
Answer» `L TIMES a` |
|
| 4. |
The cell constant of a conductivity cell_____ |
|
Answer» CHANGES with CHANGE of electrolyte |
|
| 5. |
The cell constant for an electrical conductivity cell having two electrodes of area A placed at a distance of l is expressed by |
|
Answer» `L//A` `K = (1)/(rho)and rho = R(A)/(l) thereforeK+ (1)/(R)(1/A) or K=Gl/A` where, G is conductance of the cell the quantity `(l/A)` is CALLED cell constant (R) |
|
| 6. |
The cell constant is the product of resistance and |
|
Answer» CONDUCTANCE |
|
| 7. |
The cell constant of a conductivity cell______ |
|
Answer» CHANGES with change of electrolyte A=cross SECTIONAL area of cell. * For each cell the VALUE of l and A is constant, which does not change, so the cell constant of cell does not change. |
|
| 8. |
The C Cl_4 and CHCl_3 can be distinguish by the action of : |
|
Answer» RNH_2+KOH (ALC.) |
|
| 9. |
The C=C bond length in ethylene is: |
|
Answer» 1.34 `OVERSET@A` |
|
| 10. |
The C-C bond length in ethane is: |
|
Answer» 1.20 `OVERSET@A` |
|
| 11. |
The cause of water pollution is due to :- |
|
Answer» Micro-organisms |
|
| 12. |
The cause of ozone deplection is due to release of chlorine atoms into the stratosphere. The chlorine atoms come from the breakdown of chloroflurocarbons by ultraviolet radiation. |
|
Answer» |
|
| 13. |
The cause of food spoilage |
|
Answer» PRESENCE of moisture |
|
| 14. |
The cause of Brownian movement is: |
|
Answer» HEAT CHANGES in the liquid state |
|
| 15. |
The cations present in a slightly acidic solution are Fe^(3+), Zn^(2+) and Cu^(2+). The reagent that when added in excess to this solution would be identified and separated Fe^(3+) in one step is |
|
Answer» 2 M HCl |
|
| 16. |
The cations present inA are |
|
Answer» `K^(o+)` and `Na^(o+)` 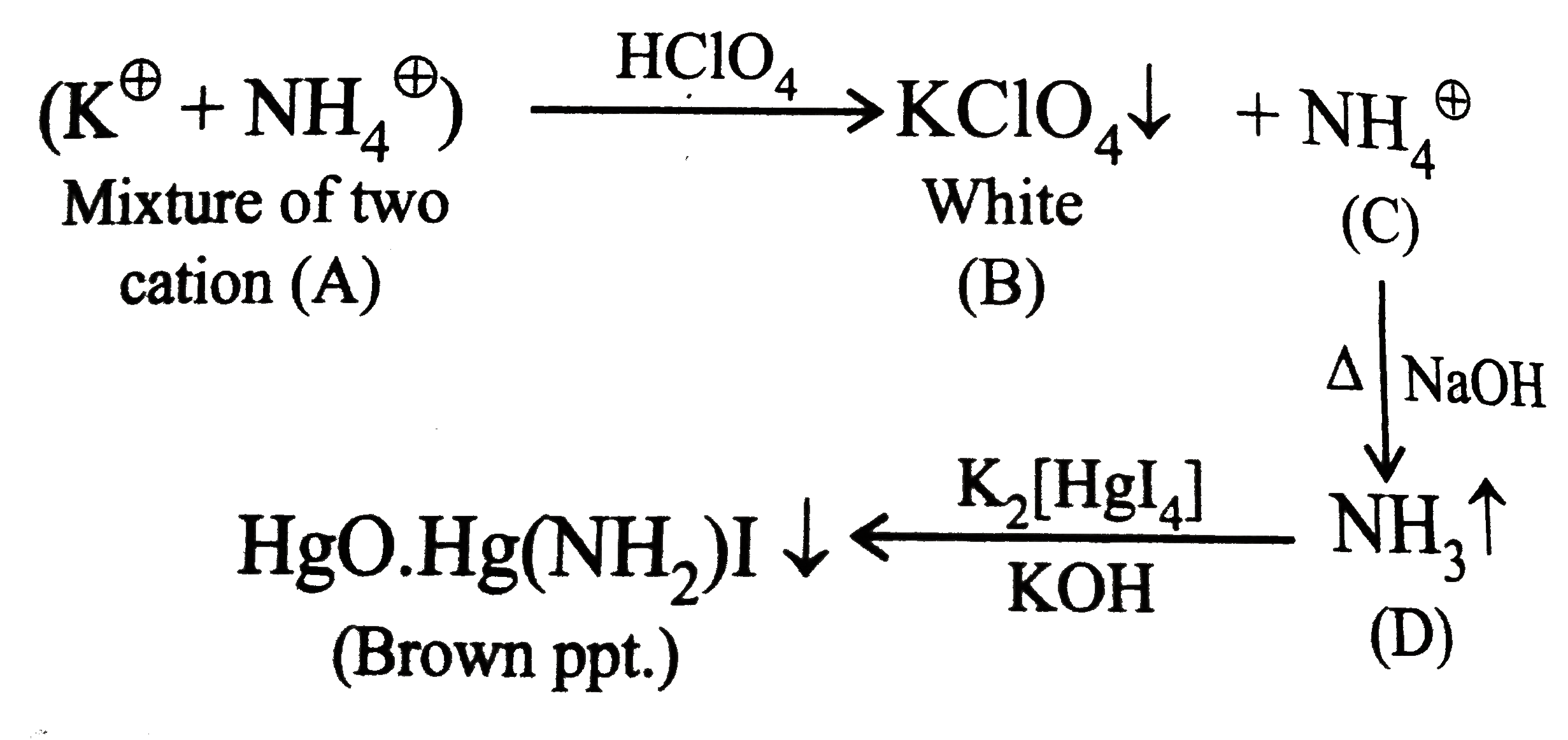
|
|
| 17. |
The cationic detergent that is used in hair conditioners is |
|
Answer» sodium DODECYLBENZENE sulphonate |
|
| 18. |
The cationic detergent that is used in hair conditioners is. |
|
Answer» SODIUM dodecy BENZENE Chloride |
|
| 19. |
The cation which is expected to act as ligand is: |
|
Answer» `Pb^(4+)` |
|
| 20. |
The cation which forms a yellow precipitate with potassium chromate in acetic acid is: |
|
Answer» `NH_4^+` |
|
| 22. |
The cation leaves its normal position in the crystal and moves to some interstitial position, the defect in the crystal is known as ...................................................... |
|
Answer» SCHOTTKY DEFECT |
|
| 23. |
The cation gives +ve microcosmic salt (or) phsophate bead test Cu^(+2), Mg^(+2), Fe^(+2), Cr^(+3), Co^(+2), Zn^(+2), Al^(+3), Mg^(+2), K^(+), Ca^(+2), Ba^(+2) |
|
Answer» `Cu^(+2), Mg^(+2), Fe^(+2), CR^(+3), Co^(+2), Zn^(+2), AL^(+3), Mg^(+2), K^(+), Ca^(+2), Ba^(+2)` |
|
| 24. |
The cathodic reaction of a Zn - MnO_2dry cell is represented as2 MnO_2(s) + Zn^(2+) + 2e = Zn MnO_4(s) .If there is 8.0 g MnO_2present in the cathodic chamber, how long will the cell function to supply 4 xx 10^(-3)ampere of current? |
|
Answer» Solution :`(4 XX 10^(-3) xx t(s))/(96500)= 8/87 , E_(MnO_2)= 87` 25.67 days |
|
| 25. |
The cation-anion bond have the largest amount of covalent character for: |
|
Answer» NaBr |
|
| 26. |
The cathodic reaction in electrolysis of dilute sulphuric acid with platinum electrode is : |
|
Answer» OXIDATION |
|
| 27. |
The cathodic reaction in electrolysis of dilute H_(2)SO_(4) with Pt electrode is |
|
Answer» REDUCTION |
|
| 28. |
The cathode reaction : MnO_2 + NH_4^(+) + e^(-)to MnO(OH) + NH_3is applicable in |
|
Answer» VOLTAIC cell |
|
| 29. |
The cathode in Leclanche cell is _______. |
|
Answer» Zinc container |
|
| 30. |
The catenation tendency of C, Si and Ge is the order GeltSiltC. The bond energies (in Kjmol^(-1)) of C-C, Si-Si and Ge-Ge bonds, respectively are |
|
Answer» `167, 180, 248` |
|
| 31. |
The cathode in agalvanic cell and electrolytic cell is |
|
Answer» negatively CHARGED in both CASES |
|
| 32. |
The catenation power of a 'N' in its compound where 'N' shows fractional oxidation state is 'X' . |
| Answer» Solution :In `N_3H` - Nitrogen exihibits fractional OXIDATION state and CATENATION POWER is x = 3 `OVERSET(Theta)N=overset(o+)N=N-H` | |
| 34. |
The catalytic decomposition of formic acid may take place in two ways: (I) HCOOH rarr H_(2)O+CO (II) HCOOH rarr H_(2)+CO_(2) The rate constant and activation energy for reaction (I) are 2.79xx10^(-3) min^(-1) at 237^(@)C and 12.0 kcal mol^(-1) respectively. (II) are 1.52xx10^(-4) min^(-1) at 237^(@)C and 24.5 kcal mol^(-1) respectively. Find out the temperature at which equimolar quantities of H_(2)O, CO, CO_(2) and H_(2) are formed. (R=2 cal K^(-1) mol^(-1)) |
|
Answer» |
|
| 35. |
The catalytic decomposition of hydrogen peroxide is a ................. order reaction. |
|
Answer» zero |
|
| 36. |
The catalytic behaviour of transition metals and their compounds is ascribed mainly due to |
|
Answer» their MAGNETIC behaviour |
|
| 37. |
The catalytic activity of transition metals and their compounds is mainly due to |
|
Answer» their magnetic behaviour `2V_(2)O_(4)+O_(2) to 2V_(2)O_(5)` |
|
| 38. |
The catalyticactivityy of transitionmetal isdue to |
|
Answer» THEFORMATIONOF a VARIETYOF OXIDATION STATE |
|
| 39. |
Thecatalyticbehaviourof transitionmetalsand theircompounds isascribedmainlydueto |
|
Answer» theirmagneticbehaviour |
|
| 40. |
The catalytic activity of transition metals and their compounds is ascribed mainly to |
|
Answer» their ability to ADOPT variable oxidation STATES |
|
| 41. |
Catalytic activity of transition elements and their compounds is due to their |
|
Answer» their magnetic behaviour |
|
| 42. |
The catalysts which increase the rate of reaction are called……….. Catalysts. |
| Answer» SOLUTION :POSITIVE | |
| 43. |
The catalyst used to increase the dissociation of H_(2)O_(3) is |
|
Answer» ACETANILIDE |
|
| 44. |
The catalyst used in the preparation of high density polyethylene is |
|
Answer» `N_2` |
|
| 45. |
The catalyst used in the preparation of red from yellow P is : |
|
Answer» `I_2` |
|
| 46. |
The catalyst used in the preparation of an alkyl chloride by the action of dry HCl on an alcohol is |
|
Answer» anhydrous `AlCl_(3)` Note : Tertiary alcohols are very reactive and hence they react READILY with conc. HCl even in the absence of `ZnCl_(2)`. |
|
| 47. |
The catalyst used in the manufacture of sulphuric acid by contact process is: |
|
Answer» Platinished asbosios |
|
| 48. |
The catalyst used in the manufacturer of HNO_3 by Ostwald.s process is: |
|
Answer» PLATINUM black |
|
| 49. |
The catalyst used in the manufacture of polythene by Zeigler method is: |
|
Answer» TITANIUM TETRACHLORIDE and TRIPHENYL aluminum |
|
| 50. |
The catalyst used in the manufacture of polyethane by Zeigler method is : |
|
Answer» TITANIUM TETRACHLORIDE and TRIPHENYL aluminium |
|



