Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct difference between first- and second -order reactions is that- |
|
Answer» a first ORDER REACTION can be catalysed , a second order reaction cannot be catalysed |
|
| 2. |
The correct difference between first and second order reaction is that |
|
Answer» The rate of a FIRST order reaction does not DEPEND on reactant CONCENTRATION , the rate of a second order reaction does depend on reactant concentrations `(t_(1//2)) 2^(nd)` order `prop (1)/([A]_(0))` |
|
| 3. |
The correct difference between first and second order reaction is that………. |
|
Answer» the rate of a first-ORDER reaction does depend on reactant concentration ,the rate of a SECOND-order reaction does not depend on reactant concentrations First life order reaction `(t_(1)/(2))=(0.693)/(K)` Half life time of a Second order reaction `(t_(1)/(2)),t_((1)/(2))prop(1)/([A_(o)])` |
|
| 4. |
The correct descending order of the heat liberated(in kJ) during the neutralisation of the acids CH_3COOH (W), HF (X), HCOOH(Y) and HCN (Z) under identical conditions is (Given K of CH_3COOH=1.8xx10^(-5) , HCOOH = 1.8xx10^(-4) , HCN =4.9xx10^(-10) and HF=3.2xx10^(-4) ) |
|
Answer» `Y gt X gt Z gt W` Stronger the ACID, higher is the heat liberated during the neutralisation as lesser energy is used for ionisation. `therefore`The CORRECT order of heat liberated is `X gt Y gt W gt Z` |
|
| 5. |
The correct description of bonds in C_2H_2 Molecule is: |
|
Answer» `sp^3d` |
|
| 6. |
The correct decreasing order ofoxidation number ofoxygen in compounds BaO_2, O_3, KO_2 and OF_2 is : |
|
Answer» `BaO_2 GT KO_2 gt O_3 gt OF_2` |
|
| 7. |
The correct decreasingorder of SN^(1) reactivity of the folliwng is: I. PhCH_(2)X. II. CH_(2) = CHCH_(2)X III. Me_(2)CHX, IV. C_(2)H_(5)X |
|
Answer» `(I) GT (II) gt (III) gt (IV)` BENZYL gt ALLYL `gt 2^(@) gt 1^(@)` halide. |
|
| 8. |
The correct decreasing order of preference of functional groups during the IUPAC nomenclature of polyfunctional compounds is: |
|
Answer» ` -COOH,-SO_(3)H,-CONH_(2),-CHO` |
|
| 9. |
The CORRECT decreasing order of energy releasedwhen an electron is added to neutral gaseous halogens is _________. |
|
Answer» `F GT CL BR gt I` |
|
| 10. |
The correct decreasing order of calorific values is |
|
Answer» `C_(2)H_(2)gtC_(4)H_(10)gtC_(3)H_(8)gtC_(2)H_(4)` |
|
| 11. |
The correct decreasing order of basic strength of the following species is_____ H_(2)O,NH_(3),OH^(-),NH_(2)^(-) |
|
Answer» `NH_(2)^(-) GT OH^(-) gt NH_(3) gt H_(2)O` |
|
| 12. |
The correct decreasing order of basic strength of the following species is ….. H_(2)O, NH_(3), OH^(-),NH_(2)^(-) |
|
Answer» `NH_(2)^(-)GT OH^(-) gt NH_3gtH_2 O` Hence, the order of basic strength is `NH_(2)^(-)gtOH^(-)gtNH_3gtH_2 O` |
|
| 13. |
The correct decreasing order of basic strength of the following species is: H_(2) O, NH_3 , OH^(-) ,NH_(2)^(-) |
|
Answer» `NH_(2)^(-)gt OH^(-) gt NH_3gtH_2 O` Hence, the order of basic STRENGTH is `NH_(2)^(-)gtOH^(-)gtNH_3gtH_2 O` |
|
| 14. |
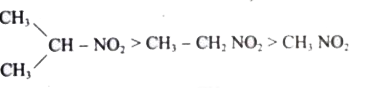
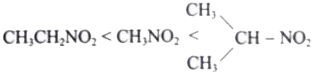
The correct decreasing order of acidity of nitro alkane is ……………….. . |
|
Answer»
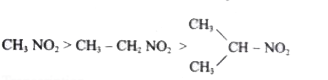
|
|
| 15. |
The correct decreasing order of acidic character of following phenols is |
|
Answer» (`AAK_MCP_36_NEET_CHE_E36_019_A01`) |
|
| 16. |
The correct decreasing order of 1^st ionizationenergy of 13-group elements is |
|
Answer» `BgtAlgtGagtIngtTl` |
|
| 17. |
The correct corresponding order of names of four aldoses with configuration given below respectively, is |
|
Answer» L-erythrose, L- threose, D- erythrose, D-threose `HOCH_(2) (CHOH)_(2) CH_(2) OH` It contains two different ASYMMETRIC `C` ATOMS RESULTING into two PAIRS of enantiomers (four optical isomess): 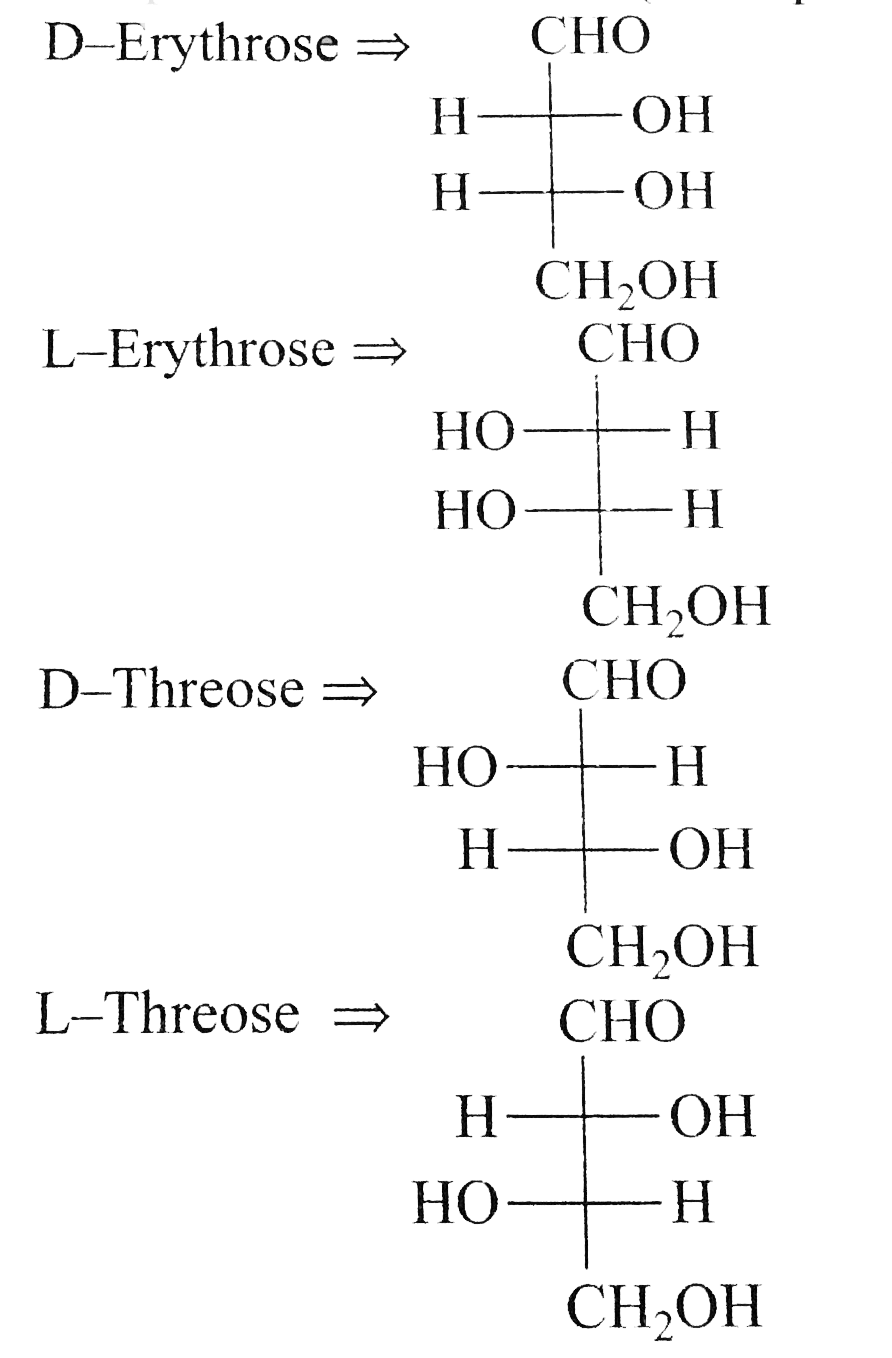
|
|
| 18. |
The correct decreasing order for the acidic strength of oxides of nitrogen is |
|
Answer» `N_(2)O_(5)gtN_(2)O_(3)gtN_(2)O_(4)gtNOgtN_(2)O` |
|
| 19. |
The correctcorrespondingorderof namesof four aldoeswithconfigurationgivenbelowRespectively is , |
|
Answer» L- ERYTHROSE, L - THREOSE , L- Erythrose , D- Threose |
|
| 20. |
The correct corresponding order names of four aldoses with configuration given below. {:(""CHO),("|"),(H-C-OH),("|"),(H-C-OH),("|"),(""CH_(2)OH):}""{:(""CHO),("|"),(" "HO-C-OH),("|"),(""H-C-OH),("|"),(""CH_(2)OH):} {:(""CHO),("|"),(" "OH-C-OH),("|"),(" "OH-C-OH),("|"),(""CH_(2)OH):}""{:(""CHO),("|"),(""H-C-OH),("|"),(" "OH-C-OH),("|"),(""CH_(2)OH):} respectively, is : |
|
Answer» L-erythrose, L-threose, D-erythrose, D-threose |
|
| 21. |
The correct combination of names for isomeric alcohols with molecular formula C_(4)H_(10)O is/are |
|
Answer» tert-butanol and 2-methylpropan-2-ol |
|
| 22. |
The correct compounds statement is /are : |
|
Answer» `X is CH_3-oversetoverset(O)(||)C-Oph` 
|
|
| 23. |
The correct configuration of the compound is …… |
|
Answer» (2R, 3R) |
|
| 24. |
The correct combination is |
|
Answer» II,III,R |
|
| 25. |
The correct combination of IV, |
|
Answer» <P>(IV) (III) (P) |
|
| 26. |
The correct combination is |
|
Answer» I,III,S |
|
| 27. |
The correct combination in which the reaction proceeds through radical intermediate is/are : |
|
Answer» <P>(I)(i)(S) 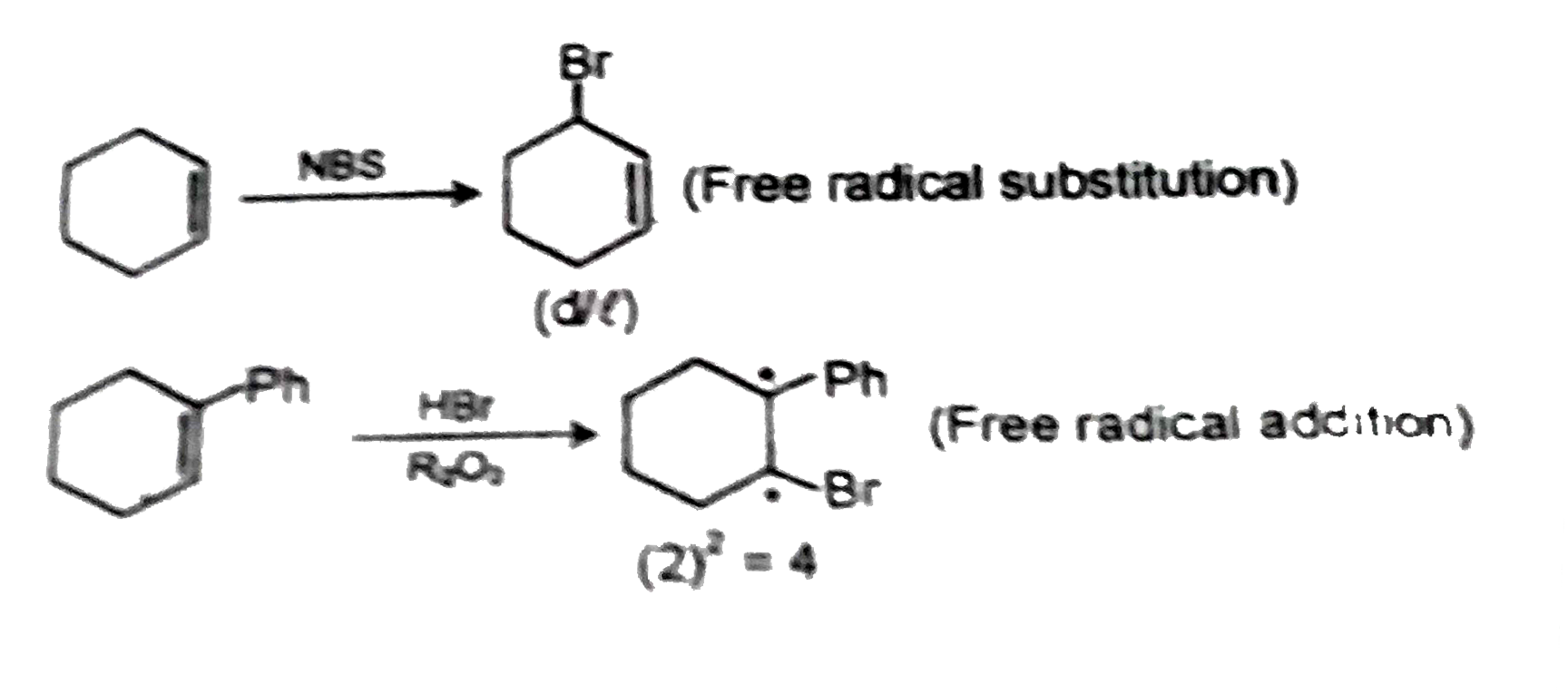
|
|
| 28. |
The CORRECT combination is : |
|
Answer» <P>(I) (II) (P) |
|
| 29. |
The CORRECT combination is : |
| Answer» Answer :C | |
| 30. |
The correct code for stability, of oxidation states for given cations is: (i) Pb^(2+) gt Pb^(4+) ,Tl^(+) lt Tl^(3+) (ii) Bi^(3+) lt Sb^(3+) , Sn^(3+) lt Sn^(4+) (iii) Pb^(3+) gt Pb^(4+),Bi^(3+) gt Bi^(3+) (iv) Tl^(3+) lt ln^(3+), Sn^(2+) gt Sn^(4+) (v) Sn^(2+) lt Pb^(2+) , Sn^(4+) gt Pb^(4+) (vi) Sn^(2+) lt Pb^(2+), Sn^(4+) lt Pb^(4+) |
|
Answer» V and VI |
|
| 31. |
The correct combination for a titration in which conductance at equivalent point is lower than initial |
|
Answer» (I)(II)(Q) |
|
| 32. |
The correct combination from the following given statements about chemisorption I) It is unilayered adsorption II) It is irreversible and takes place slowly III) It occurs rapidly The correct combination is |
|
Answer» Both I and II are CORRECT |
|
| 33. |
The CORRECT combination among the following is: |
|
Answer» <P> (IV) (III) (P) |
|
| 34. |
The correct charge on an co-ordination number of 'Fe' in K_(3)[Fe(CN)_(6)] is |
|
Answer» `+2, 4` O.N. of K=+1 O.N. of Fe = x O.N. of CN=`-1` Sum=O `K_(3)Fe(CN)_(6)` `(3 XX 1) + (x) + 6(-1)=0` `x=+3` (b) `6CN^(-)` ions are attached to Fe and hence C.N. of Fe=6 |
|
| 35. |
The correct order of boiling points of the given hydrides of 16^(th) group elements is |
|
Answer» `H_(2) O GT H_(2) S gt H _(2) Se gt H _(2) TE` |
|
| 36. |
The correct basicity order of thefollowing lanthanoid ions is |
|
Answer» `La^(3+) gt Lu^(3+) gt Ce^(3+) gt Eu^(3+)` For DETAILS, consult section 11. |
|
| 37. |
The correct bhasicity order of the folloiwing lanthanide ions is |
|
Answer» `LA^(3+) gt LU^(3+) gt CE^(3+) gt Eu^(3+)` |
|
| 38. |
Explain, why. alkyl halides have higher boiling points than the corresponding hydrocarbons? |
|
Answer» ALKYNE GT ALKANE gt ALKENE |
|
| 39. |
The correct ascending order of adsorption of the following gases on the same mass of charcoal at the same temperature and pressure is |
|
Answer» `CH_(4)lt H_(2)SO_(2)` `{:(H_(2)lt " "CH_(4)lt " "SO_(2)), (33.2 " "190.6 ""430.3):}` or ease of liquefaction is `H_(2)lt CH_(4) lt SO_(2).` Hence, ascending ORDER of adsorption is `H_(2) lt CH_(4)lt SO_(2).` |
|
| 40. |
The correct arrangement of following acids of phosphorus in the increasing order of oxidation state of phosphorous is |
|
Answer» HYPOPHOSPHORIC acid LT orthophosphorous acid lt pyrophosphoric acid |
|
| 41. |
The correct arrangement for the ions in the increasing order of their radii is |
|
Answer» `NA^(+), Cl^(-), Ca^(2+)`  The species `Ca^(2+), K^(+)` and `S^(2-)` are isoelectronic. For isoelectronic ions, the radius decreases as the nuclear charge increases. Hence, the increasing order of their RADII is `Ca^(2+) LT K^(+) S^(2-)` |
|
| 42. |
The correct arrangement of the species in the decreasing order of the bond length between carbonand oxygen in them is |
|
Answer» `CO, CO_(2), HCO_(3)^(-), CO_(3)^(2-)` |
|
| 43. |
The correct answer upto required number of significant figures of 0.083 x 10.1 is : |
|
Answer» 0.8383 |
|
| 44. |
The correct answer upto proper number of significant digits : (2.50 x 10^(-2) km) + (3.7 xx 10^2 cm)is : |
|
Answer» 6.20 KM `(25.0 m) + (3.7 m) = 28.7 m ` |
|
| 45. |
The correct answer upto appropriate number of significant figures for 50.0 x 0.0160 + 19 is: |
|
Answer» SOLUTION :`50.0 XX 0.0160= 0.800` `0.800 + 19 = 19.8 " or" 20` |
|
| 46. |
The correct answer of 126/8.0 is (upto proper number of significant figures) : |
|
Answer» 15.75 |
|
| 47. |
The correct acidity order of the following is |
|
Answer» IIIgtIVgtIIgtI 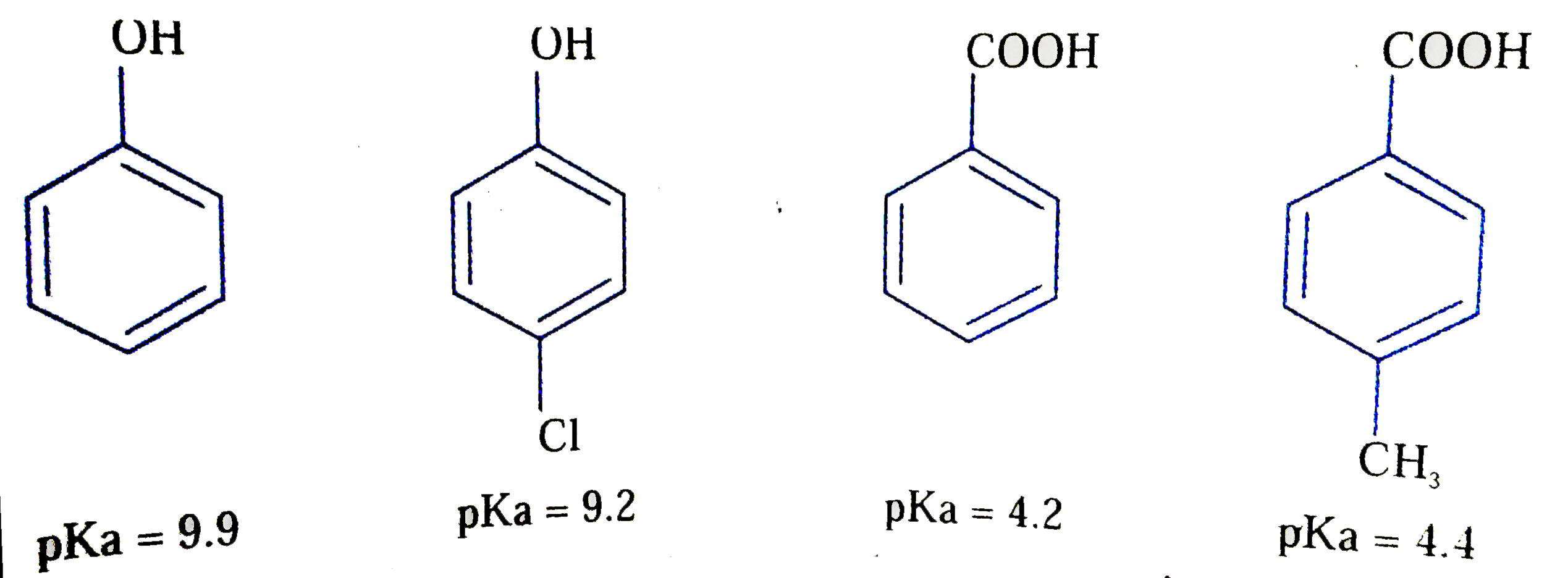 Carboxylic acids are more ACIDIC than phenols (exception as pictric acid which is a PHENOL) Electron withdrawing groups exerts acid -strengthening effect while Electron supplying group EXERT acid -WEAKENING effect. The -I effect of Cl of DOMINATING over its +R effect. |
|
| 48. |
The correct acidic strength order of the following compounds is (a)CH_3COOH (b)H_2CO_5 ©C_2H_5OH (d)C_6H_5OH (e)H_2O (f) C_2H_2 |
|
Answer» ` agt B GT d gt e gt C gt f` |
|
| 49. |
The correct –I effect order is:- |
|
Answer» `-NH_(2) lt -OH lt -F` |
|