Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The cornpressibility factor of a van der Waals gas at the critical point is equal to |
|
Answer» 0 |
|
| 2. |
The cornpound X, in the reaction, Xoverset(CH_(3)Mgl)toYoverset("Hydrolysis")toMg(OH)I+CH_(3)COOH is: |
|
Answer» HCHO |
|
| 3. |
The corect order of decreasing length of the bond as indicated by the arrow in the following structures is |
|
Answer» `I gt II gt III` C = C bond length is 1.33 Å C - C bond length is 1.54 Å 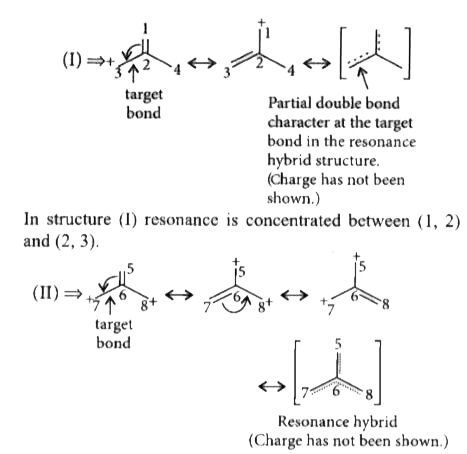 Here, resonance is concentrated in between (5, 6), (6, 7) and (6, 8). So, partial double bond CHARACTER is lesser in between (6 & 7) than that of (2 & 3) in (I). 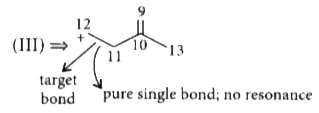 So, the order of single bond character is , 11 - 12 `gt` 6 - 7 `gt` 2 - 3 THUS , bondlength order will be `III gt II gt I`. |
|
| 4. |
The copounds which is not coloured is |
|
Answer» `K_(4)Fe(CN)_(6)` So, its ELECTRONIC configuration is `4d^(10)4s^(0)` All the 4d orbitals are fully filled. hence, there will not be d-d transition resulting in colour. So, it is colourless. |
|
| 5. |
The core of a non-luminous Bunsen burner flame is observed to be yellow in colour. This is because of : |
|
Answer» Contamination from the METAL of the BURNER |
|
| 6. |
The coordination polyhedra of [Ni(CO)_(4)] is ………… |
|
Answer» Octahedral |
|
| 7. |
The coordination polyhedral of [Ni(CO)_(4)] is …………… . |
|
Answer» |
|
| 8. |
The coordination polyhedra of K_(3)[Fe(CN)_(6)] is ………… |
|
Answer» SQUARE planar |
|
| 9. |
The coordination number X (---) of each ion in KBr is changed to Y (---) by |
|
Answer» X=6 to Y= 8 APPLYING high temperature |
|
| 10. |
The coordination numbers of oxygen and silicon in SiO_(4)respectively |
|
Answer» 1,2 |
|
| 11. |
The coordination number of nickel (II) ion is 4. NiCl_(2)+underset(("excess"))(KCN)tounderset(("cyano complex"))(X) X+underset(("excess"))(conc. HCl)tounderset(("chloro complex"))(Y) The IUPAC names for the complexes X and Y are respectively. |
|
Answer» potassium tetracyanidonickle (II) , potassium tetrachloridonickle (II) `K_(2)[Ni(CN)_(4)]+4HCl to underset("potassium tetrachloridonickelate"(II)) (K_(2)[Ni(Cl)_(4)])+4HCN` |
|
| 12. |
The coordination number of Pt in [Pt(NO_(2))(H_(2)O)(NH_(3))_(2)] Br is……. . |
|
Answer» |
|
| 13. |
The coordination number of zinc sulphide is ............ ……………………. |
|
Answer» 3 |
|
| 14. |
The coordination number of Ni^(2+) is 4 NiCl_(2)+KCN ("excess")rarr A("cyano complex") A + conc. HCl ("excess")rarr B ("chloro complex") The hybridisation of A and B are |
|
Answer» `DSP^(2), SP^(3)` |
|
| 15. |
The coordination number of Ni^(2+) is 4 NiCl_(2)+KCN ("excess")rarr A("cyano complex") A + conc. HCl ("excess")rarr B ("chloro complex") Predict the magnetic nature of A and B |
|
Answer» Both are diamagnetic 
|
|
| 16. |
The coordination number of Ni^(2+) 4 NiCl_(2) + KCN (excess) to A (cyano complex) NiCl_(2) + Conc. HCI (excess) to B (chloro complex)QThe hybridization of A and B are |
|
Answer» `DSP^(2)SP^(3)` |
|
| 17. |
The coordination number of Ni^(2+) 4 NiCl_(2) + KCN (excess) to A (cyano complex) NiCl_(2) + Conc. HCI (excess) to B (chloro complex)QPredict the magnetic nature of A and B |
|
Answer» Both are DIAMAGNETIC |
|
| 18. |
The coordination number of Ni^(2+) is 4 NiCl_(2)+KCN ("excess")rarr A("cyano complex") A + conc. HCl ("excess")rarr B ("chloro complex") The IUPAC names of A and B are |
|
Answer» POTASSIUM tetracyanonickeltate(II), potassium tetrachloronickeltate(II) `K_(2)[Ni(CN)_(4)]+4HCL ("excess")RARR underset("tetrachloronickelate(II)")underset("Potassium")(K_(2)[NiCl_(4)])+HCN` |
|
| 19. |
The coordination number of Ni^(2+) 4 NiCl_(2) + KCN (excess) to A (cyano complex) NiCl_(2) + Conc. HCI (excess) to B (chloro complex)QThe IUPAC name of A and B are |
|
Answer» POTASSIUM tetracyanonickelate (IL), potassium tetrachloronickelate (II) |
|
| 20. |
The coordination number of metal M in the complen [M(en)(C_(2)O_(4))ClBr]^(-) is |
|
Answer» |
|
| 21. |
The coordination number of h.c.p. or c.c.p. structure is 12, that is, one atom touches 12 other atoms. The 12 atoms lie as |
|
Answer» 8 ATOMS are on the same plane, 2 above and 2 below the plane |
|
| 23. |
The coordination number of CsCl is ....................................... |
|
Answer» 3 |
|
| 25. |
The coordination number of [Cr(en)_(3)][CrF_(6)] and oxidation state of Cr are…….. . |
|
Answer» |
|
| 27. |
The coordination number of Ca^2+ ion in fluorite crystal is: |
|
Answer» 2 |
|
| 28. |
The coordination number of Ca^(2+) ion in fluorite crystal is: |
|
Answer» 2 |
|
| 29. |
The coordination number of a metal in coordination compound is |
|
Answer» Same as PRIMARY valency |
|
| 30. |
The coordination number of a metal crystallizing in a hexagonal close packed structure is |
|
Answer» 4 |
|
| 31. |
The coordination number of a metal crystallising in a hexagonal close-packed structure is |
|
Answer» 12 |
|
| 32. |
The coordination number of a metal crystallising in a hexagonal close-packed structure is : |
|
Answer» 12 |
|
| 33. |
The coordination number of a metal crystallising a hcp structure is |
| Answer» ANSWER :A | |
| 34. |
The coordination number of a centrla metal atom in a complex is determined by : |
|
Answer» The number of only anionic ligands BONDED to the metal ion |
|
| 35. |
The coordination number of a central metal atom in a complex is determined by the number of _________. |
|
Answer» ligands around a metal ions is BONDED by both SIGMA and pi-bonds |
|
| 36. |
The coordination number of a body-centred atom in cubic structure is |
|
Answer» 4 |
|
| 37. |
The coordination number, EAN of the central metal atom and geometry of the complex ion obtained by adding CuSO_4 to excess of aqueous KCN are respectively. |
|
Answer» `4, 35 , sp^2 d ` `2Cu(CN)_2 to Cu_2 (CN)_2 + (CN)_2` `Cu_2 (CN)_2 + 6KCN to K_3[Cu(CN)_4],` `O.N. ` of Cu= +1 `Cu^(2+) (29) =(AR)3d^(10)` 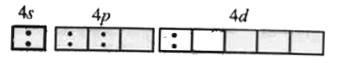
|
|
| 38. |
The coordination number and the oxidation state of element E in the complex [E(en)_(2)(C_(2)O_(4))]NO_(2) (where en is ethylene diamine) are, respectively : |
|
Answer» 4 and 2 |
|
| 39. |
The coordination number and oxidation number of 'x' in the following, Compound [x(NH_(3))_(5)(SO_(4))]Cl will be |
| Answer» Solution :Both `NH_(3)` and `SO_(4)^(2-)` are monodentate LIGANDS so, co-ordination no. of X is 6. No. of X is +3. | |
| 40. |
The coordination number and oxidation number of M in the compound [M(SO_4 )(NH_3)_5] will be |
|
Answer» 6 and 3 |
|
| 41. |
The coordination compounds are of great importance. These compounds are widely present in the mineral, plant and animal worlds and are known to play many important functions in the area ofanalytical chemistry, metallurgy, biological systems, industry and medicine. In which of the following processes, co-ordination complex formation/usage is done for extraction of metals ? |
|
Answer» Mc ARTUR FORREST CYANIDE process |
|
| 42. |
The coordination compounds are of great importance. These compounds are widely present in the mineral, plant and animal worlds and are known to play many important functions in the area ofanalytical chemistry, metallurgy, biological systems, industry and medicine. Which of the following reagents can be used for qualitative and quantitative analysis (based on colour reactions) as well as for estimation of hardness of water ? |
|
Answer» EDTA |
|
| 43. |
The coordination compound of which one of the following compositions will produce two equivalents of AgCl on reaction with aqueous silver nitrate solution ? |
|
Answer» `CoCl_(3).3 NH_(3)` |
|
| 44. |
The coordination isomerism is exhibited by : |
|
Answer» `[CR(NH_(3))_(6)][CO(CN)_(6)]` |
|
| 45. |
The cooling in refrigerator is due to: |
|
Answer» The WORK of compressor |
|
| 46. |
The cooling in refrigerator is due to |
|
Answer» reaction of the refrigerator gas |
|
| 47. |
The cooling caused by the adiabatic expansion of a compressed gas below its inversion temperature (T_i) without doing external work is called : |
|
Answer» Joule-Thomson effect |
|
| 48. |
The coolant usually contains a solution of antifreeze prepared by mixing equal volumes of ethylene glycol, C_2 H_4 (OH)_2and water. The density of ethylene glycol is 1.113 g/mL. Calculate the f.p. of the mixture. K_f for H_2Ois 1.86. |
| Answer» SOLUTION :`-33.4^@C` | |
| 49. |
The coolant used in the reaction core can be |
|
Answer» Heavy water |
|