Saved Bookmarks
| 1. |
The corect order of decreasing length of the bond as indicated by the arrow in the following structures is |
|
Answer» `I gt II gt III` C = C bond length is 1.33 Å C - C bond length is 1.54 Å 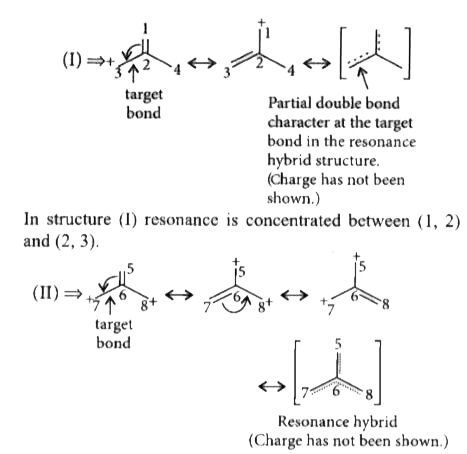 Here, resonance is concentrated in between (5, 6), (6, 7) and (6, 8). So, partial double bond CHARACTER is lesser in between (6 & 7) than that of (2 & 3) in (I). 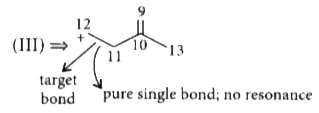 So, the order of single bond character is , 11 - 12 `gt` 6 - 7 `gt` 2 - 3 THUS , bondlength order will be `III gt II gt I`. |
|
Discussion
No Comment Found
Related InterviewSolutions
- Which of the following compounds is not cleaved by HI even at 525 K ?
- To a 25 mL H_(2)O_(2) solution excess of an acidified solution of potassium iodide was added. The iodine liberated required 20 " mL of " 0.3 N sodium thiosulphate solution Calculate the volume strength of H_(2)O_(2) solution.
- The suggested mechanism of a reaction is : (a) A+BhArrD("fast) "(b)A+Drarr2C("slow")Write the balanced equation of the reaction if its experimentally deduced rate equation is , rate k=[A]^(2)[B] Find the intermediate formed during the course of the reaction . Does the predicted rate law from the mechanism match the experimental rate law ?
- Which of these changes with time for a first-order reaction A Rate of reaction B . Rate constant C . Half-life
- What is the hybridisation of central atom in the product obtained along with hydrofluoric acid when complete hydrolysis of Xenon Hexa Fluoride takes place ?
- Which of the following amino acid forms sulphide bond in polypeptide
- Which of following pair is Diastereomers:
- What is the major product of the following reaction CH_3C-=C-CH_2-CH_3overset("1 mole of " Cl_2)to
- Which polymer is used in petrol tank linings ?
- Which of the following carbohydrates are branched polymer of glucose ?