Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On heating ammonium dichromate, the gas evolved is: |
|
Answer» Oxygen |
|
| 2. |
On heating ammonium dichromate and barium azide separately, we get |
|
Answer» `N_2` in both cases |
|
| 3. |
On heating acetamide in presence of P_(2)O_(5), which of the following is formed |
|
Answer» <P>Ammnium acetate |
|
| 4. |
On heating, a white amorphous inorganic compound becomes yellow and on cooling, turns white again. The salt may be |
|
Answer» `PbCO_(3)` |
|
| 5. |
on heating Al at 800^(@)C in air,Al_2 O_3 is formed. The reaction is : |
|
Answer» REDUCTION of aluminium |
|
| 6. |
On heating a sample of pure water: |
|
Answer» `[H^(+)]` INCREASES but `[OH^(-)]` decreases. |
|
| 7. |
On heating a mixture of potassium dichromate and sodium chloride with concentrated sulpuric acid in a dry test tube, the compound formed is |
|
Answer» chormiun chloride |
|
| 8. |
On heating a mixture of sodium benzoate and sodalime, the following is obtained |
|
Answer» Toluene 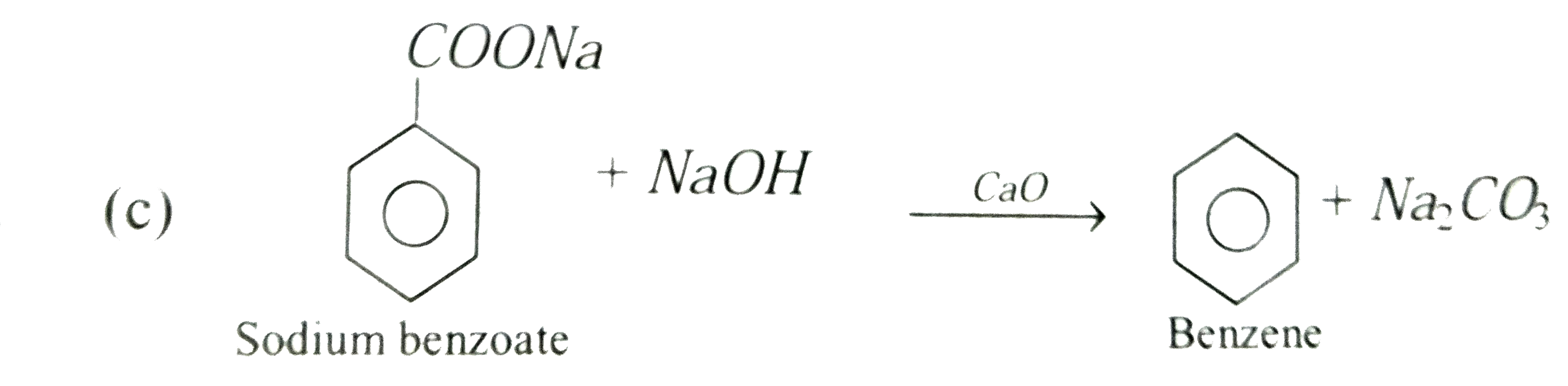
|
|
| 9. |
On heating a metal chloride with K_2Cr_2O_7 and conc. H_2SO_4, the gas evolved is : |
|
Answer» `O_2` |
|
| 10. |
On heating a crystal of KCl in potassium vapour, the crystal starts exhibiting a violet colour. What is this due to ? |
| Answer» SOLUTION :This is DUE to METAL EXCESS DEFECT. | |
| 11. |
On heating 2.701 g of mercuric oxide, 1.95 mL of dry oxygen at 37^@ C and 620 mm pressure were collected. The residue weighed 2.499 g. From this data calculate the molecular weight of oxygen. |
|
Answer» |
|
| 12. |
On heating 2,2-Dimethylcyclohexanol with concentrated sulphuric acid, the major alkene formed is …. |
|
Answer»
|
|
| 13. |
On heating 1.763 g of hydrated BaCl_2 to dryness, 1.505 g of anhydrous salt remained. Hence the formula of the hydrate is (Atomic weight of Ba = 137) |
|
Answer» `BaCl_2 . 1/2 H_2O` |
|
| 14. |
On gram of silver gets distributed between 10cm^(3) of molten zinc and 100 cm^(3) of molten lead at 800^(@)C. The percentage of silver still left in the lead layer is approximately |
|
Answer» 2 `("Conc. (%) of AG in molten Zn")/("Conc. (%) of Ag in molten PB")=K"(distribution coeff.)"` = 300 If at equilibrium, x g of Ag is LEFT in `100cm^(3)` of molten lead, Ag that goes into `10cm^(3)` ZINC `=(1-x)g` `therefore""(10(1-x))/(x)=300"or"10-10x=300x` `"or"310x=10"or"x=(1)/(31)g` Thus, out of 1 g of Ag, Ag present in Pb layer `=(1)/(31)g` `therefore"% of Ag left in the Pb layer "=(1)/(31)xx100~~3%` |
|
| 15. |
On going left to right in a period, in transition metals, their atomic volumes |
|
Answer» Decrease |
|
| 16. |
On going from right to left in a period in the periodic table the electronegativity of the elements |
|
Answer» INCREASES |
|
| 17. |
On fusion with KOH benzophenone undergoes ________, and gives potassium benzoate and __________. |
|
Answer» DISPROPORTIONATION reacion, toluene |
|
| 18. |
On exposure to air, ether forms an explosive compound ….. |
|
Answer» PEROXIDE |
|
| 19. |
On exposure to light electrical conductivity of selenium |
|
Answer» Increases |
|
| 20. |
On electrolysis of aq NaCl solution ,H_(2)(g), Cl_(2)(g)and NaOH are produced : 2Cl^(-)(aq)+2H_(2)Orarr2OH^(-)(aq)+H_(2)(g)+Cl_(2)(g) 62 % efficient 25 A electricity is passed through 20L 20 % NaCl solution . ( i ) Write the electrode reactions ( ii ) How much time will it take to produce 1 kgCl_(2)( iii ) What will be the molarity at that time with respect to OH^(-)? ( suppose no volume change occurs during electrolysis ) |
| Answer» SOLUTION :`Q=Ixxtxx0.62=2718309.86""THEREFORE t=48.715h.` | |
| 21. |
On electrolysis, which of the following does not give out hydrogen? |
|
Answer» Acidic water using Pt ELECTRODES |
|
| 22. |
On electrolysis of sodium succinate, the alkene obtained is ______ and nature of solution after electrolysis is _______ |
|
Answer» `OVERSET(CH_(2))overset(||)(CH_(2))` and ACIDIC |
|
| 23. |
On electrolysis of an aqueous solution NaCl, why H_(2) and not Na is liberated at the cathode? |
| Answer» Solution :This is because `H^(+)` ions produced from ionization of WATER have LOWER discharge potential that `Na^(+)` ions produced from ionization of NACL or REDUCTION potential of water is greater than that of sodium ions. | |
| 24. |
On electrolysis of 500 mL of an aqueous solution of NaCl, NaOH is produced, the normality of which is found to be N/2. The number of faradays used is |
|
Answer» 0.25 |
|
| 25. |
On electrolysis, 1 mole of aluminium will be deposited from its molten salt by |
|
Answer» 3 MOLES of electrons |
|
| 26. |
On electrolysis , 1 mole of aluminium will be deposited from its molten salt by : |
|
Answer» 1 MOLE of electrons |
|
| 27. |
On electrolysing a solution of dilute H_2SO_4 between platinum electrodes, the gas evolved at the anode and cathode are respectively is : |
|
Answer» `SO_2 and O_2` |
|
| 28. |
On doubind P and V with constant temperaturee the eq1ulibrium constant will |
|
Answer» Remain constant Pressure will never affect the VALUE of K. It may result in the shifting or equlibrium but not the equlibrium constnt value. Tepmperatue always changes K because on adding heat to a system in state of equlibrium will always alter the FORWARD reaction rate differently than the reverse reaction rate. Also by doubling the volume, the concentrations of both reactants and product evently become half. Therefore overall there is no change in equlibrium constant value (K). |
|
| 29. |
On electrolysing a sample of acidified water, 22.4 ml of hydrogen was obtained. The volume ofoxygen in ml obtained is |
|
Answer» 22.4 |
|
| 30. |
ON disuphonatioon followed by fusion with NaOH an dacidificant, an aromaticcompoundC_(8) H_(10) (A) gives two isomeric comnpounds(B) and (C)(molecularformulaC_(8) H_(10) O_(2). Oxidationof estersof (B) and (C) followed by hydrolysis givestwo isomers comnpounds (D) and (E) (C_(7) H_(6) O_(4)) which may bedecarboxylated to formC_(6) H_(6) O_(2) (F). Identigycompounds (A) to (F). |
Answer» SOLUTION :`4 D.U` and `C:H = 1:1` in `(A)` SUGGEST benzene ring with either TWO `(Me)` groups in the ring or one `(Et)` group in the side chain.  Formation of `(F)` suggests that only one `(-COOH)` is decarboxylated becuase from the above REACTION sequenceit is clear that `(F)` contains two `(OH)` groups. Therefore. `(A)` contains `(Et)` group. Reactions: 
|
|
| 31. |
On doping Ge metal with a little of In, one gets: |
|
Answer» `rho-type` semiconductor |
|
| 32. |
On doping Ge metal with a little of gallium one gets |
|
Answer» <P>p - TYPE SEMI conductor |
|
| 33. |
When phenol is distilled with Zn dust it gives |
| Answer» Answer :A | |
| 34. |
On dissolving sugar in water at room temperature, solution feels cool to touch. Under which of the following cases dissolution of sugar will be most rapid? |
|
Answer» sugar crystals in cold water |
|
| 35. |
On dissolving sugar in water at room temperature solution feels cool to touch. Under which of the following cases dissolution of sugar will be most rapid? |
|
Answer» Sugar crystals in cold water |
|
| 36. |
On dissolving NaCl in water there occurs: |
|
Answer» BECOMES zero |
|
| 37. |
On dissolving moderate amount of sodium metal is liquid NH_(3) at low temperature, which one of the following does not occur |
|
Answer» Blue coloured solution is obtained |
|
| 38. |
On dissolving moderate amount of sodium metal in liquid ammonia at low temperature, which one of the following does not occur ? |
|
Answer» Blue COLOURED solution is obtained |
|
| 39. |
On dissolving 3.24 g of sulphur in 40 g of sulphur in 40 g of benzene, boiling point of solution was higher than that of benzeneby 0.81 K. K_(b) value for benzene is "103.0 g mol"^(-1), calculate the molal elevation constant for benzene. |
|
Answer» |
|
| 40. |
On dissolving 3.24 g of sulphur in 40 g of benzene, the boiling point of the solution was higher than sulphur? (K_(b) for benzene = 2.53 K kg mol^(-1) , atomic mass of sulphur = 32 g mol^(-1)). |
|
Answer» Solution :The given VALUES are: `W_(B) = 3.24 g`, `W_(A) = 40 g` `DeltaT_(b) = 0.81 K`, `K_(b) = 2.53 KG mol^(-1)` Using formula, `Mw_(B) = (K_(b) xx 1000 xx W_(B)) /(DeltaT_(b) xx W_(A))` On substituting all the values, we get `:. Mw_(B) = (2.53 xx 1000xx 3.24) / (0.81 xx 40) = 253` Let the molecular formula of sulphur = `S_(x)` Atomic mass of sulphur = `32` Molecular mass = `32 xx x` `:. 32X = 253` `x= (253)/(32) = 7.91 ~~ 8` `:.` Molecular formula of sulphur `= S_(8)` |
|
| 41. |
On dissolving 3.24 g of sulphur in 40 g of benzene, boliling point of the solution was highter then that of benzene by 0.81 K. The molal elevation constant (K_(b)) for benzeneis 2.53 K kg mol^(-1). "What is the molecular formula of sulphur "("Atomic mass of" S=32 g mol^(-1))? |
|
Answer» Solution :Calculation of molecular mas of sulphur `(S_(N))` `M_(B)=(K_(b)xxW_(b))/(DeltaT_(b)xxW_(A))` Mass of sulphur `(W_(b))=3.24g` `" Mass of benzene"(W_(A))=40g=0.04 kg` Molal elevetion constant `(K_(b))=2.53 K kg mol^(-1)` Elevation in BOILING point `(Delta T_(b))=0.81 K` `M_(B)=((2.53 K kg mol^(-1))XX(3.24 g))/((0.81 K)xx(0.04 kg))=253g mol^(-1)` Calcultion of molecular formula of sulphur. Gram atomic mass of sulphur =32 g `mol^(-1)` In `S_(n),nxx(32g mol^(-1))=253 g mol^(-1)` `n=((253 g mol^(-1)))/((32g mol^(-1)))=7.9~~8` Molecular formula of sulphur is `S_(8)`. |
|
| 42. |
On dissolving 3.24 g of sulphur in 40 g of benzene, boiling point ofsolution was higher than that of benzene by 0.81K (K_(b) = 2.53 K kg mol^(-1)). What is molecular formula of sulphur ? (Atomic mass s = 32 gmol^(-1)) |
|
Answer» SOLUTION :`M_(B) = (K_(b) xx W_(b) xx 1000)/(Delta T_(b) xx W_(A)) = (2.53 xx 3.24 xx 10^(3))/(0.81 xx 40)` `= 253 g mol^(-1)` Let the molecular formula of sulphur = `S_(x)` Atomic MASS of sulphur = 32 Molecular mass = `32 xx x` 32 x = 253 `x = 7.91 = 8` Molecular formula of sulphur = `S_(S)` |
|
| 43. |
On dissolving 2.0g of metal in sulphuric acid. 4.51g of the metal sulphate was formed. The specific heat of the metal is 0.057 cal g^(-1). What is the valency of the metal and exact atomic mass? |
|
Answer» Solution :Equivalent MASS of `SO_(4)^(2-)` radical `=("Ionic mass")/("Valency")=(96)/(2)=48` Mass of metal SULPHATE =4.51 g ltbr. Mass of metal =2.0g Mass of sulphate radical =(4.51-2.0)=2.51g 2.51 g of sulphate combine with 2.0g of metal. So, 48 g of sulphate will combine with `(2)/(2.51)=48=38.24g` metal Equivalent mass of metal =38.24 ACCORDING to Dulong and PETIT's law, Approximate atomic mass `=(6.4)/("specific HEAT")=(6.4)/(0.057)=112.5` `"Valency"=("Approximately atomic mass")/("Equivalent mass")` `=(112.5)/(38.24)=2.9=3` Exact atomic mass `=38.24xx3=114.72`. |
|
| 44. |
On dissolving 1 mole of each of the following acids in 2 litre water, the which does not give a solution of strength 1 N is |
|
Answer» HCl `H_(2)PO_(4)^(-)to H^(+)+HPO_(4)^(2-)` `HPO_(4)^(2-)to H^(+)+PO_(4)^(3-)` Phosphoric acid does not give 1N strength. |
|
| 45. |
On dissolving 19,5 g of CH_2 FCOOHin 500 g of water, a depression of 1^@Cin freczing pointof water is observed. Calculate the van't Hoff factor and dissociation constant of fluoro acetic acid. Given, K_f = 1.86 K kg "mol"^(-1) |
|
Answer» Solution :Use the equation : `M_2 = (K_f xx w_2 xx 1000)/(Delta T_fxx w_1)` where `M_2`= observed molecular mass of the solute, `K_f` = molal depression constant, `w_2` = mass of the solute, `w_1 `= mass of the solvent, `Delta T_f` = Depression in FREEZING point Substituting the values in the above equation, we have `M_2 = (1.86 xx 19.5 xx 1000)/(1 xx 500) = 72.54` No. of moles of the ACID n = 19.5/76 = 0.2566 van.t HOFF factor, i=Normal MOLAR mass / Abnormal (observed) molar mass ` = (78)/(72.54) = 1.0753` DISSOCIATION constant `underset(C(1 - alpha))(CH_2FCOOH) iff underset(C alpha)(CH_2FCOO^(-)) + underset(C alpha)(H^(+))` `alpha = i - 1 = 1.0753` `K_a = ([CH_2FCOO^(-) ][H^+])/([CH_2FCOOH]) = (C alpha.C alpha)/(C(1 - alpha)) = (C alpha^2)/(1 - alpha) = C alpha^2` (neglecting a compared to 1) ` C = (19.5 xx 1000)/(78 xx 500) = 0.5 M` `K_a = C alpha^2 = 0.5 xx (0.0753)^2 = 2.83 xx 10^(-3)` |
|
| 46. |
On dissolving 0.25 g of a non-volatile substance in 30 mL of benzene (density 0.8 g mL^(-1)), its freezing point decreases by 0.40^(@)C. Calculate the molecular mass of the non-volatile substance. K_(f) for benzene is 5.12 Km^(-1) . |
|
Answer» `K_(f)=5.12" K kg MOL"^(-1), M_(B)=?` `M_(B)=(K_(f)xxW_(B))/(DeltaT_(f)xxW_(A))=((5.12" K kg mol"^(-1))xx(0.25 g))/((0.40 K)xx(0.024 kg ))=133.33" g mol"^(-1)`. |
|
| 47. |
On dissolution of Xenates [HXeO_4]^-in alkaline solution perxenate and Xe are obtained(R) Xenates show disproportionation in alkaline solution |
|
Answer» Both (A) and (R) are TRUE and (R) is the correct EXPLANATION of (A) |
|
| 48. |
On dilution of 0.1 M of Na_(2)SO_(4), what will happen to its (a) Conductance (C) (b) Conductivity kappa (c) Molar conductance wedge_(m) (d) Equivalent conductance wedge |
| Answer» SOLUTION :Conductivity, MOLAR CONDUCTANCE and equivalent conductance increases with dilution whereas Conductance (C) DECREASES. | |
| 49. |
On decreasing intermoleculer distance below, the optimum distnace (where potential energy is minimum), there is sleep increase in potential energy due to: |
|
Answer» Increase in force of ATTRACTION between ELECTRONS and nucleus |
|
| 50. |
On cracking petrol, we get |
|
Answer» `CH_4` |
|



