Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On heatingthe salt with NHO_(3) and ammonium molybdate , formationof yellow ppt indicates thepresence of (a)_____or (b) _____. |
|
Answer» |
|
| 2. |
On heating to 400-500^@C, relatively unstable hydrides and carbonates decompose. Which of the following will decompose when heated to 400-500^@C |
|
Answer» `II, III` |
|
| 3. |
When sodium metal heated in dry ammonia, the compound formed is: |
|
Answer» SODIUM nitrate |
|
| 4. |
on heating sodium as well as sulphur can be melted molten sodium and molten sulphur are used as: |
|
Answer» MEDIUM for EXTRACTING metals |
|
| 5. |
On heating sodium acetate with sodium hydroxide, the gas evolved will be |
|
Answer» Acetylene |
|
| 6. |
On heating salicylic acid with soda lime the productobtained is |
|
Answer» Benzene |
|
| 7. |
On heating propanamide with "SOCI"_(2) the productformed as : |
|
Answer» PROPANOL |
|
| 8. |
On heating potassium dichromate, it decomposes to give …..… and molecular oxygen. |
| Answer» SOLUTION :Chromium(III) oxide - `Cr_(2)O_(3)` | |
| 9. |
On heating potassium permanganate, one of the following compound is not obtained : |
|
Answer» `MnO_(2)` |
|
| 10. |
On heating, peroxides are _______ |
|
Answer» stable |
|
| 11. |
On heating ozone its volume |
|
Answer» DECREASES to half |
|
| 12. |
On heating orthoboric acid to red hot, the reidue is : |
|
Answer» Boron |
|
| 13. |
On heating NaCl + K_(2)Cr_(2)O_(7) +H_(2)SO_(4) conc., the gas comes out |
|
Answer» `CrO_(2)Cl_(2)` |
|
| 14. |
On heating magnesium boride with HCl a mixture of volatile. …………….. Are obtained. |
|
Answer» |
|
| 15. |
On heating MgCl_(2). 2H_(2)O at 350^(@)C in the presence of anhydrous MgC_(2), is formed |
| Answer» Answer :A | |
| 16. |
On heating, lead nitrate forms oxides of nitrogen and lead. The oxides formed are _____. |
|
Answer» `N_2O,PBO` |
|
| 17. |
On heating, lead nitrate forms oxides of nitrogen and lead. The oxides formed are: |
|
Answer» `N_2O,PbO` |
|
| 18. |
On heating lead (II) nitrate gives a brown gas "A". The gas "A" on cooling changes to colourless solid "B". Solid "B" on heating with no changes to a blue solid 'C'. Identify 'A', 'B' and 'C' and also write reactions involved and draw the structures of 'B' and 'C'. |
|
Answer» Solution :The gas A is `NO_2`. The reactions are explained as under : `2Pb(NO_3)_2 UNDERSET(673K) overset(Delta)to 2PbO + underset(" A(Brown colour)")(4NO_2) + O_2` `2NO_2 overset("On cooling ")to underset("(Colourless SOLID)")(N_2O_4)` `2NO+ underset(B)(N_2O_4) overset(250K)to underset("C (Brown colour solid )")(2N_2O_3)` STRUCTURE of B `(N_2O_4)` : 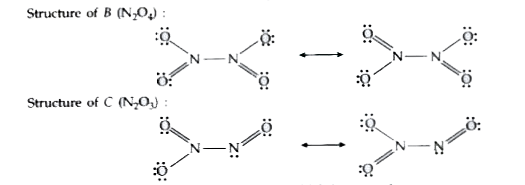
|
|
| 19. |
On heating lead nitrate forms oxides of nitrogen and lead. The oxides formed are....... |
|
Answer» `N_(2)O, PbO` |
|
| 20. |
On heating, lead (II) nitrate gives a brown gas 'A'. The gas 'A' on cooling changes to colourless solid 'B'. Solid 'B' on heating with NO changes to a blue solid 'C'. Identify 'A', 'B' and 'C' and also write reactions involved and draw the structures of 'B' and 'C'. |
|
Answer» Solution :(i) Since lead (II) NITRATE on heating gives a brown gas 'A', therefore, gas 'A' must be nitrogen dioxide `(NO_(2))`. `UNDERSET("Lead (II) nitrate")(2Pb(NO_(3))_(2)) overset(DELTA, 673 K)RARR 2 PbO + underset("Brown gas (A)")(4 NO_(2))+O_(2)` (ii) Thre brown gas 'A' on cooling dimerises to give a colourless solid 'B', therefore 'B' must be `N_(2)O_(4)` (dinitrogen tetroxide). `2 NO_(2) underset("On heating")overset("On cooling")(hArr) underset("Colourless solid (B)")(2N_(2)O_(4))` (iii) Since colourless solid 'B' on heating with NO, gives a blue solid 'C', therefore 'C' muct be dinitrogen trioxide. `2 NO + underset("Colourless (B)")(N_(2)O_(4)) rarr underset("Blue solid (C)")(2 N_(2)O_(3))` Thus, `A = NO_(2), B = N_(2)O_(4) and C = N_(2) O_(3)`. |
|
| 21. |
On heating lead (II) nitrate gives a brown gas "A". The gas "A" on cooling changes to colourless solid "B". Solid "B" on heating with NO changes to a blue solid «C. Identify 'A', 'B' and 'C and also write reactions involved and draw the structures of 'B' and 'C\ |
|
Answer» SOLUTION :`2PB(NO_(3))_(2) overset(Delta)UNDERSET(673 K) to 2PbO + underset(A)(4NO_(2)) + O_(2)` `2NO_(2)(g) overset("COOLING")underset("Heating")  |
|
| 22. |
Onheatinglead (II)nitrategives a brown gas 'A' . Thegas 'A' on coolingchanges to colourisess solid 'B'. Solid 'B' on heatingwith NO changeto a bluesolid 'C'. Identify 'A' ,'B' and 'C' and alsowritereactions involvedand draw the structures of 'B' and 'C'. |
|
Answer» Solution :`2Pb(NO_(3))_(2) underset(673K)overset(Delta)to 2PbO + 4NO_(2) (A)`(brown)+ `O_(2)` `2NO_(2)underset("Heating")overset("On cooling")hArr N_(2)O_(4)` (B) (brown) + `O_(2)` `2NO_(2) + N_(2)O_(4) overset(Delta 250K)to underset("(C)")2N_(2)O_(3)` (bluesolid) STRUCTUREOF `N_(2)O` :  Structure of `N_(2)O_(3)` : 
|
|
| 23. |
On heating in following compound an allyl group will migrate to which possition. |
|
Answer» |
|
| 24. |
On heating K_(2)Cr_(2)O_(7) with NaCl and conc. H_(2)SO_(4), the gas liberated is |
|
Answer» `CrO_(2)Cl_(2)` |
|
| 25. |
On heating K_(4)[Fe(CN)_(6)] with conc. H_(2)SO_(4) gives the gas |
|
Answer» `SO_(2)` |
|
| 26. |
On heating in excess supply of air, K, Rb and Cs form superoxides rather than oxides and peroxides? |
| Answer» SOLUTION :`K^(+)`,`Rb^(+)` and `Cs^(+)` are larger cations which stabilize`O_(2)^(-)` (superoxide) the larger ANION. Thus when `MO_(2)` is formed, it is stabilized hence FORMATION is preferred to OXIDES and PEROXIDES. | |
| 27. |
On heating in air, 0.12 g of metal gave 0.20 g of its oxide. The carbonate and nitrate of the metal were found to contain 28.5% and 16.2% of the metal respectively. Calculate by applying law of constnat proportions, the masses of oxide of the metal that will be obtained by heating 10 g each of the carbonate and the nitrate. |
|
Answer» Solution :Mass of oxygen in metal oxide =(0.20-0.12)=0.08g So, the ratio of masses of metal and oxygen in metal oxide 100gof metal CARBONATE contain metal =28.5g 10 G of metal carbonate contain metal `=(28.5)/(100)xx10=2.85g` If x g of oxygen combine with 2.85 g of metal, the ratio of masses of the metal and oxygen should be 3:2 as the LAW of constant proportions is true. `(2.85)/(x)=(3)/(2)` or `x=1.9g` Mass of metal oxide obtained from 10G metal carbonate =Mass of metal +Mass of oxygen `=(2.85+1.9)=4.75g` 100g of metal NITRATE contain metal =16.2g 10g of metal nitrate contain metal `=(16.2)/(100)xx10=1.62g` If y g oxygem combine with 1.62 g of metal, the ratio of masses of the metal and oxygen should be 3:2. `(1.62)/(y)=(3)/(2)` or `y=1.08g` Mass of metal oxide obtained from 10g metal nitrate =Mass of metal+Mass of oxygen. =(1.62+1.08)=2.70g |
|
| 28. |
On heating glycerol with conc. H_(2)SO_(4) a compound is obtained which has a bad odour. The compound is : |
|
Answer» Glycerol sulphate |
|
| 29. |
On heating glycerol with .KHSO_4, compound is obtained which has bad odour. The compound is : |
|
Answer» Acrolein |
|
| 30. |
On heating glucose with Fehling solution, we get a precipitate whose colour is |
|
Answer» Orange |
|
| 32. |
On heating ethyl bromide with an alloy of lead-sodium, we get : |
|
Answer» ETHYL LEAD BROMIDE |
|
| 33. |
On heating ethanol with exess of concentrated H_(2)SO_(4) " to " 170^(@)C, the product obtained is |
|
Answer» ethyne `CH_(3)CH_(2)OH + H_(2)SO_(4) rarr underset(underset(C_(2)H_(4) + H_(2)SO_(4))(darr 170^(@)C))(CH_(3) CH_(2)HSO_(4)) + H_(2)O` |
|
| 34. |
On heating ethanol with excess of conc. H_2SO_4 at 443 K, product obtained is |
|
Answer» ethene |
|
| 35. |
On heating compound (A) gives a gas (B) which is a constituent of air. This gas when treated with 3 moles of hydrogen (H_2) in the presence of a catalyst gives another gas (C) which is basic in nature. Gas C on further oxidation in moist condition gives a compound (D) which is a part of acid rain. Identify compounds (A) to (D) and also give necessary equations of all the steps involved. |
|
Answer» Solution :(A): `NH_(4)NO_(2), ` (B) : `N_(2)` ( c) : `NH_(3)`, (D) : `HNO_(3)` (i) `underset(A)(NH_(4)NO_(2)) overset(Delta)to underset(2H_(2)O)` (ii) `N_(2)(g) + 3H_(2)(g) overset(Fe)to 2NH_(3)(g)` (iii) `4NH_(3)(g) + 5O_(2)(g) to 4NO(g) + 6H_(2)O(l)` (iv) `2NO(g) + O_(2)(g) to 2NO_(2)(g)` (v) `3NO_(2)(g) + H_(2)O(l) to underset(D)(2HNO_(3)) + NO(g)` |
|
| 36. |
On heating diethyl ether with conc. HI, 2 moles of which of the following is formed ? |
|
Answer» Ethanol |
|
| 37. |
On heating copper nitrate strongly______is finally obtained. |
|
Answer» COPPER |
|
| 38. |
On heating compound (A) gives a gas (B) which is a constituent of air. This gas when treated with 3 mol of hydrogen (H_(2)) in the presence of a catalyst gives another gas (C) which is basic in nature. Gas C on further oxidation in moist condition gives a compound (D) which is a part of acid rain. Identify compounds (A) to (D) and also give necessary equations of all the steps involved. |
|
Answer» Solution :(i) Since gas 'B' on heating with 3 moles of HYDROGEN in presence of a catalyst, gives a gas 'C' which is basic in nature, therefore, 'C' must be ammonia `(NH_(3))` and gas (B) must be `N_(2)` `underset((B))(N_(2))+3H_(2) overset(Delta, "Catalyst")rarr underset("Ammonia (C)")(2 NH_(3))` (ii) Since gas 'B' (i.e., `N_(2)`) is obtained by heating compound (A), therefore, compound (A) must be EITHER ammonium NITRITE or ammonium dichromate `underset((A))(NH_(4))NO_(2) overset(Delta)rarr underset((B))(N_(2))+2H_(2)O or underset((A))((NH_(4))_(2))Cr_(2)O_(7) overset(Delta)rarr underset((B))(N_(2))+Cr_(2)O_(3) + 4 H_(2) O` (iii) Since gas 'C', (i.e., `NH_(3)`) on further oxidation in moist conditions gives compound (D) which is a part of acid rain, therefore, compound (D) must be nitric acid `(HNO_(3))`. `{:(underset((C))(4NH_(3))+5O_(2) overset("Oxidation")rarr 4 NO + 6 H_(2)O),(2 NO + O_(2) rarr 2 NO_(2)"," " "3 NO_(2) + H_(2)O rarr underset((D))(2HNO_(3))+NO):}` Thus, `A = NH_(4)NO_(2) or (NH_(4))_(2) Cr_(2) O_(7), B = N_(2), C = NH_(3) and D = HNO_(3)`. |
|
| 39. |
On heating compound (A) gives a gas (B) which is a constituent of air. This gas when treated with 3 mol of hydrogen (H_2) in the presence of a catalyst gives another gas (C) which is basic in nature. Gas C on further oxidation in moist condition gives a compound (D) which is a part of acid rain. Identify compounds (A) to (D) and also give necessary equations of all the steps involved. |
|
Answer» Solution :Compound (A) to (D) are as FOLLOWS : (A) = `NH_4NO_2` (B) `=N_2` (C) `=NH_3` (D)`=HNO_3` The reactions are GIVEN as under : (i) `NH_4NO_2 OVERSET(DELTA)to N_2 + 2H_2O ` (ii) `N_2 + 3H_2 to 2NH_3` (iii) `4NH_3 + 5O_2 to 4NO + 6H_2O ` `2NO + O_2 to 2NO_2 ` `3NO_2 + H_2O to 2HNO_3 + NO` |
|
| 40. |
On heating colourless solid A, gas B (liquid at room temp.) and gas C are formed . A is decomposed by NaOH on heating to form gas D giving white fumes with HCl. Gas C occupies 800ml//g at N.T.P. Hence A is - |
|
Answer» `NH_(4)NO_(3)` |
|
| 41. |
On heating, chloride acid decomposes to |
|
Answer» `HCIO_(4), CI_(2), O_(2)` and `H_(2)O` |
|
| 42. |
On heating chloroform with silver powder , which of the following is formed ? |
| Answer» Answer :3 | |
| 43. |
On heating calcium acetate and calcium formate , the product formed is |
|
Answer» `CH_(3)COCH_(3)` |
|
| 44. |
On heating aniline with fuming sulphuric acid at 180^(@)C, the compound formed will be |
|
Answer» ANILINE disulphate 
|
|
| 45. |
On heating aniline with alcoholic CS_(2) in the presence of HgCl_(2) gives : |
|
Answer» <P>p - aminobenzene sulphoric acid |
|
| 46. |
On heating anhydrous Na_(2)CO_(3),..... is evolved |
|
Answer» `CO_(2)` |
|
| 47. |
On heating an ethanolic amine with chloroform and ethanolic KOH, the organic compound formed is |
|
Answer» an alky CYANIDE |
|
| 48. |
On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is |
|
Answer» ALKYL isocyanide |
|
| 49. |
On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is : |
|
Answer» an alkanol |
|
| 50. |
On heating ammonium dichromate, the gas evolved is : |
| Answer» Answer :D | |