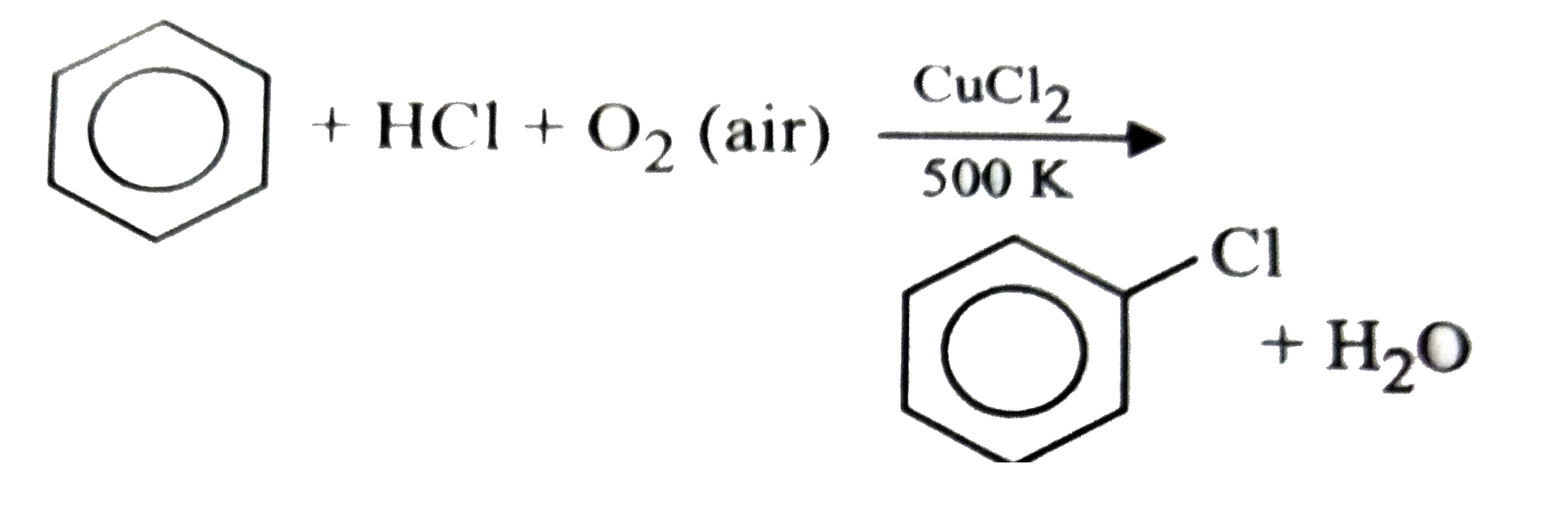Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On cracking petrol we get |
|
Answer» `CH_4` |
|
| 2. |
On cooling hot zinc oxide, the yellow colour become white. Why ? |
|
Answer» Solution :The composition of `Zn^(2+) and O^(2-)` ions in STOICHIOMETRIC `ZnO` is `n : n`. On heating ZnO, oxygen is lost reversibly. A non - stoichiometric COMPOUND with excess `Zn^(2+)`ions is formed possessing a composition of `Zn^(2+) and O^(2-)` ions `n:n-x`. Due to excess METAL in the interstities, it is YELLOW. On cooling the non-stoichiometric zinc OXIDE, it gains oxygen. A stoichiometric zinc oxide is formed and hence it is white. `Zn^(2+) + (1)/(2) O_(2) 2e^(-) to ZnO` |
|
| 3. |
On conversion into the Grignard reagent followed by treatment with absolute ethanol, how many isomeric alkyl chlorides would yield 2-methylbutane : |
|
Answer» 2 |
|
| 4. |
On complete oxidation , 1 mole of an organic compound gave 4 moles of water. The compound should be |
|
Answer» METHANOL |
|
| 5. |
On complete hydrogenation, natural rubber produces |
|
Answer» ethylene-propylene COPOLYMER 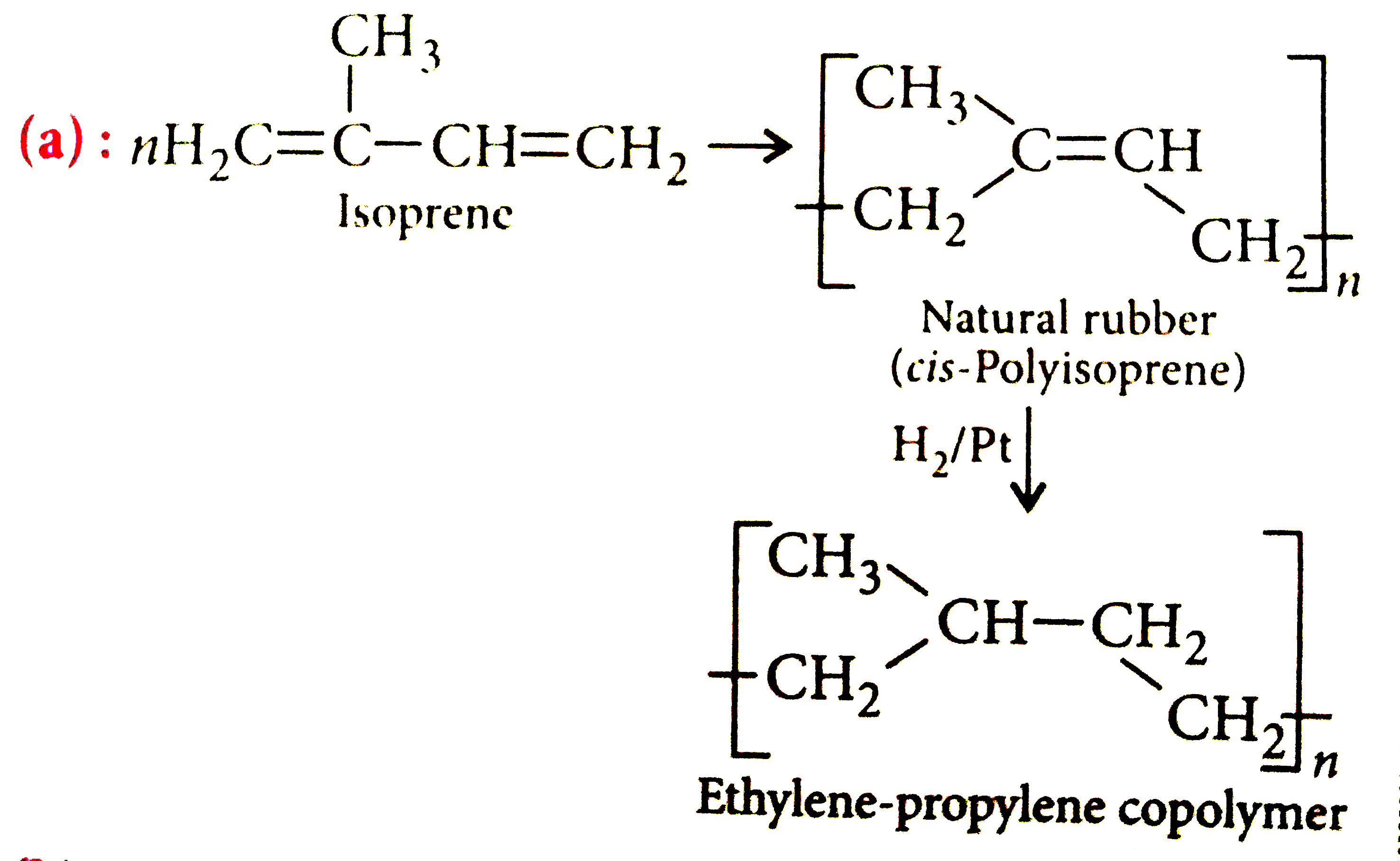
|
|
| 6. |
On complete hydrogenation , natural rubber produces. |
|
Answer» ethylene - PROPYLENE copolymer 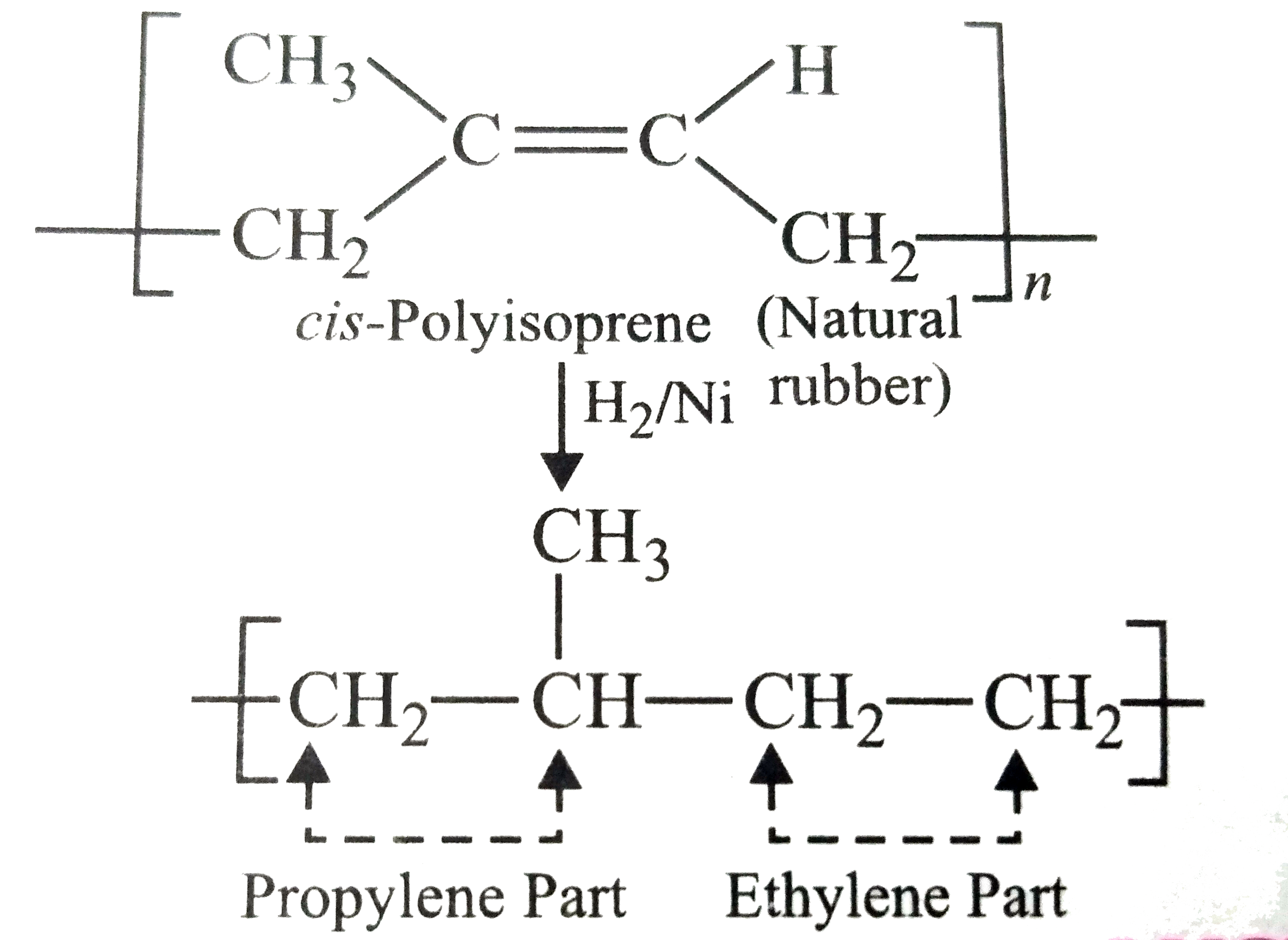
|
|
| 7. |
On commercial scale, phenol is obtained from chlorobenzene by raschig's process. Which one of the following is Raschig's process? |
|
Answer»
|
|
| 8. |
On combustion of hydrogen in hydrogen fuel cell. . . |
|
Answer» More pure water generated. Note : `H_(2)O` is FORMED by combustion of `H_(2)` with the help of `O_(2)` in fuel ELL. But the objective of this cell is to convert combusted ENERGY into electrical energy. So potential difference should be generated. So it is right. |
|
| 9. |
On combustion of 0.2475 g of an organic compound 0.4950 g of CO_(2) and 0.2025 g of H_(2)O were obtained. Find out the percentage of hydrogen. |
|
Answer» |
|
| 10. |
On combustion, carbon forms two oxides CO and CO_(2), Heat of formation of CO_(2) gas is 94.3 kcal and that of CO is 26.0 kcal. Heat of combustion of carbon is |
|
Answer» 26.0 kcal This is ALSO heat of FORMATION of `CO_(2)` `C+(1)/(2)O_(2)rarrCO, DeltaH=-26.0`. |
|
| 11. |
On combustion carbon forms two oxides CO and CO_(2). Heat of formation of CO_(2) is -393.5kJ and that of CO is -110.5kJ. Heat of combustion of CO is |
|
Answer» `-393.5kJ` (ii) `C + O_(2)(g) rarr CO_(2), Delta H = - 393.5 kJ` SUBTRACT EQ. (i) from eq. (ii), `CO + (1)/(2)O_(2) rarr CO_(2), Delta H = - 283.0 kJ` |
|
| 12. |
On charging lead storage cell. . . |
|
Answer» Pb of electrode utilize. `PbSO_(4(S))+2H_(2)O_((l))underset("Charging")overset("DC voltage") toPbO_(2(S))+SO_(4(aq))^(2-)+4H_((aq))^(+)+2e^(-)` Where, (A), (B) and (C) is INDICATION of discharging reaction of storage cell. |
|
| 13. |
On bromination, propionic acid yields two isomeric 2-bromopropionic acids. This pair is an important example of |
|
Answer» CHAIN isomers |
|
| 14. |
On catalytic hydorgenation how many isomeric alkene will give 2-Methyl butane. |
|
Answer» |
|
| 15. |
On calculating the strength of current in amperes if a charge of 840 C (coulomb) passes through an electrolyte in 7minutes, it will be |
|
Answer» 1 |
|
| 16. |
On bombardin g._(8)O^(16) with deutrons, the nuclei of the product formed will be |
|
Answer» `._(9)F^(18)` |
|
| 17. |
On boiling with concentrated hydrobromic acid, phenyl ethyl ether will yield |
|
Answer» PHENOL and ETHYL bromide 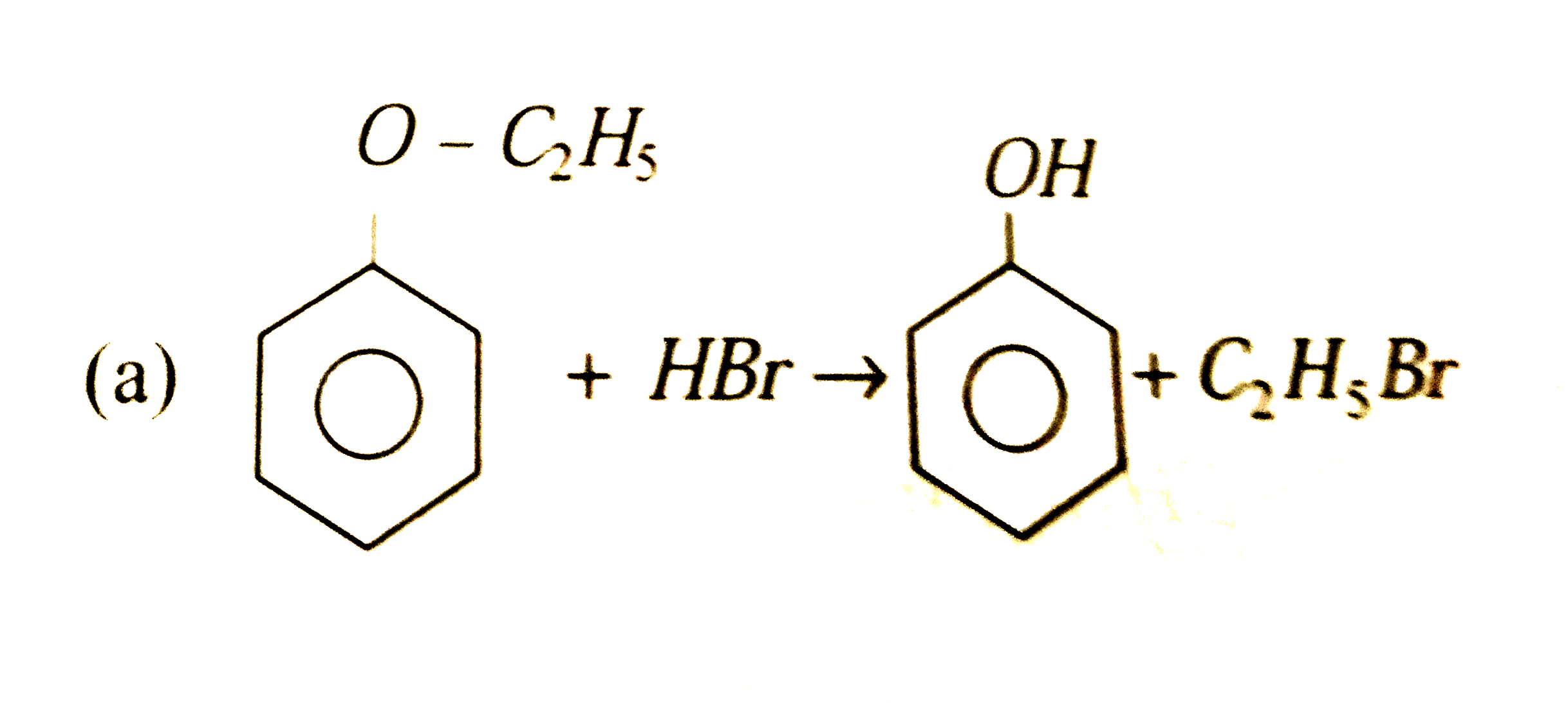
|
|
| 18. |
On boiling with concentrated HBr phenyl ethyl ether will give. |
|
Answer» Phenol and ethyl BROMIDE |
|
| 19. |
On boiling with concentrated hydrobromic acid pheyl ethyl ether yields: |
|
Answer» PHENOL and bromide |
|
| 20. |
On boiling with concentrated HBr, phenyl ether will give: |
|
Answer» Phenol and ETHYL bromide |
|
| 21. |
On boiling tin with alkali, the product is : |
|
Answer» `SnO_3^(2-)` |
|
| 22. |
On boiling the egg, what structural changes are taking place in the egg white ? |
|
Answer» The colour of the EGG changes from colourless to white. |
|
| 23. |
On boiling an aqueous solution of KClO_3 with iodine the product formed is: |
|
Answer» `KIO_3` |
|
| 24. |
On being strongly heated, a blue salt leaves a black residue. Which of the following cations can be present in the salt? |
|
Answer» `FE^(2+)` |
|
| 25. |
On being strongly heated, which of the following substances will leave a black residue |
|
Answer» `CuSO_(4), 5H_(2)O` |
|
| 26. |
On being slowly passed through water, PH_(3) forms bubbles but NH_(3) dissolves. Why is it so? |
| Answer» Solution :N-H bond is more polar than P-H bond. HENCE, `NH_(3)` forms hydrogen BONDS with `H_(2)O` MOLECLES and hence dissolves in it whereas `PH_(3)` does not dissolve and hence forms bubbles. | |
| 27. |
On being slowly passed through water, PH_3 forms bubbles but NH_3 dissolves. Why is it so ? |
| Answer» Solution :Ammonia DISSOLVES in water due to hydrogen BONDING. Phosphine being LESS soluble, escapes out in the form of BUBBLES. | |
| 28. |
On being heated with conc. HNO_(3) and ammonium molybdate solution, the salt solutions gives a yellow precipitate. The salt(s) may be |
|
Answer» `Na_(2)HPO_(4)` |
|
| 29. |
On being heated with alcoholic KOH, neopentyl bromide gives mainly : |
|
Answer» but-2-ene |
|
| 30. |
On being heated in oxygen, 3.120 g of a metal M converts to 4.560 g of oxide (atomic weight of M= 52.0). Mark the correct statement(s). |
|
Answer» Equivalent wt. of metal M= 17.33 `=4,560 = 3,120 = 1,440` No. of equivalents = `(1,440)/8 = 0.18` Equivalent weight of metal `=3.12/1.44 xx 8 = 17.33` Valency `=52/17.33 =3` |
|
| 31. |
On applying pressure to the equilibrium, ice hArr water which phenomenon will happen: |
|
Answer» More ICE will be formed |
|
| 32. |
On analysis, a substance was found to have the following percentage composition: K=31.84, Cl=28.98 and O=39.18 Calculate its molecular formula if its molecular mass is 122.5. |
| Answer» SOLUTION :`KClO_(3)` | |
| 33. |
On analysis of an impure sample of sodium chloride. The percentage of chlorine was found to be 45.5 What is the percentage of pure sodium chloride in the given sample? |
|
Answer» SOLUTION :The MOLECULAR mass of pure sodium chloride (NACL) =At. Mass of Na+ At. Mass of chlorine. =(23+35.5)=58.5 % of chlorine in pure NaCl `=(35.5)/(58.5)xx100=60.6` Thus, % of PURITY of NaCl in the sample `=(45.5)/(60.6)xx100=75`. |
|
| 34. |
On analysis, a sample of uranium was found to contain 0.277g of ""_(82)Pb^(20) and 1.667g of ""_(92)U^(238). The half-life period of ""_(92)U^(238) is 4.51xx10^(9) years. If all the lead were assumed to have come from decay of ""_(92)U^(238), what is the age of the earth? |
|
Answer» |
|
| 35. |
On analysing an impure sample of sodium chloride, the percentage of chlorine was found to be 45.5. What is the percentage of pure sodium chloride in the sample? |
|
Answer» SOLUTION :Molar MASS of pure NaCl = `23+35.5=58.5"g mol"^(-1)` `% " of Cl in pure NaCl "=(35.5)/(58.5)xx100=60.6` Thus, 60.6 parts of Cl are PRESENT in 100 parts of pure NACl 45.5 parts of Cl will be present in pure NaCl `=(100)/(60.6)xx45.5="75 parts"` The remaining `25%` is impurity present in impure sample. HENCE, `%` of pure NaCl in impure sample = `75%`. |
|
| 36. |
on aldol condensation followed by heating gives |
|
Answer»

|
|
| 37. |
On addition of small amount of KMnO_(4) to concentrated H_(2)SO_(4) , a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following : |
|
Answer» `Mn_(2)O_(7)` |
|
| 38. |
On addition of small amount of KMnO_(4) toconcentrated H_(2)SO_(4), a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following |
|
Answer» `Mn_(2)O_(7)` |
|
| 39. |
On addition of small amount of KMnO_(4) to concentrated H_(2)SO_(4), a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following. |
| Answer» Answer :a | |
| 40. |
On addition of one ml solution 10% NaCl to 10 ml gold sol in the presence of 0.0250g of starch , the coagulation is just prevented starch has the gold number : |
|
Answer» `2.5` |
|
| 41. |
On addition of one mL solution of 10% NaCl to 10 mL gold sol in the presence fo 0.0250 g of starch, the coagulation is just prevented. Starch has the following gold number |
|
Answer» `0.025` |
|
| 42. |
On addition of one mL of 10% NaCl solution to 10 mL gold sol in the presence of 0.25 g of starch, the coagulation is just prevented. Starch has the gold number |
|
Answer» 0.025 |
|
| 43. |
On addition of increasing amount of AgNO_(3)(g)to 0.1 M each of NaCl and NaBr in a solution, what % of Br^(-)ion gets precipitated when Cl^(-)ion starts precipitating? Given: K_(sp)(AgCl) = 1.0 xx 10^(-10), K_(sp) (AgBr) = 1 xx 10^(-13). |
|
Answer» `=1.0 xx 10^(-9)` M `[Br^(-)]` remaining at this stage % of `Br^(-)` remaining unpredicted % of `Br^(-)` precipitated =100-0.1 = 99.9 |
|
| 44. |
On addition of excess of sodium hydroxide solution to stamnous chloride solution, we obtains: |
|
Answer» `Sn(OH)_(2)` `Sn(OH)_(2)+2NaOH to Na_(2)SnO_(2) +2H_(2)O` |
|
| 45. |
On addition of cone. H_2SO_4 to a chloride salt, colourless fumes are evolved but in case of iodide salt, violet fumes come out. This is because ……... |
|
Answer» `H_2SO_4` REDUCES HI to `I_2` `2Nal + H_(2)SO_(4) to Na_(2)SO_(4) + underset(2H_(2)O + SO_(2) + I_(2))underset(darr H_(2)SO_(4))(2HI)` |
|
| 46. |
On addition of conc. H_2SO_4 to a chloride salt, colourless fumes are evolved but in case of iodide salt, violet fumes come out. This is because |
|
Answer» `H_2SO_4` REDUCES HI to `I_2` |
|
| 47. |
On addition of conc. H_(2)SO_(4) to a chloride salt, colourless fumes are evolved but in case of iodide salt, fumes come out. This is because |
|
Answer» `H_(2)SO_(4)` reduces `HI` to `I_(2)` |
|
| 48. |
On addition of conc. H_(2)SO_(4) to a chloride salt, colourless fumes are evolved but in case of iodide salt, violet fumes come out. This is because |
|
Answer» `H_(2)SO_(4)` reduces HI to `I_(2)` |
|
| 49. |
On addition of an inert gas at constant volume to the reaction N_(2)+3H_(2)hArr2NH_(3) at equlibrium |
|
Answer» The reaction remains unaffected |
|
| 50. |
On addition of aqueous NaOH to a salt solution, white gelatinous precipitate is formed, which dissolves in excess alkali. The salt solution contains |
|
Answer» CHROMIUM ions Which is soluble in excess alkali. |
|