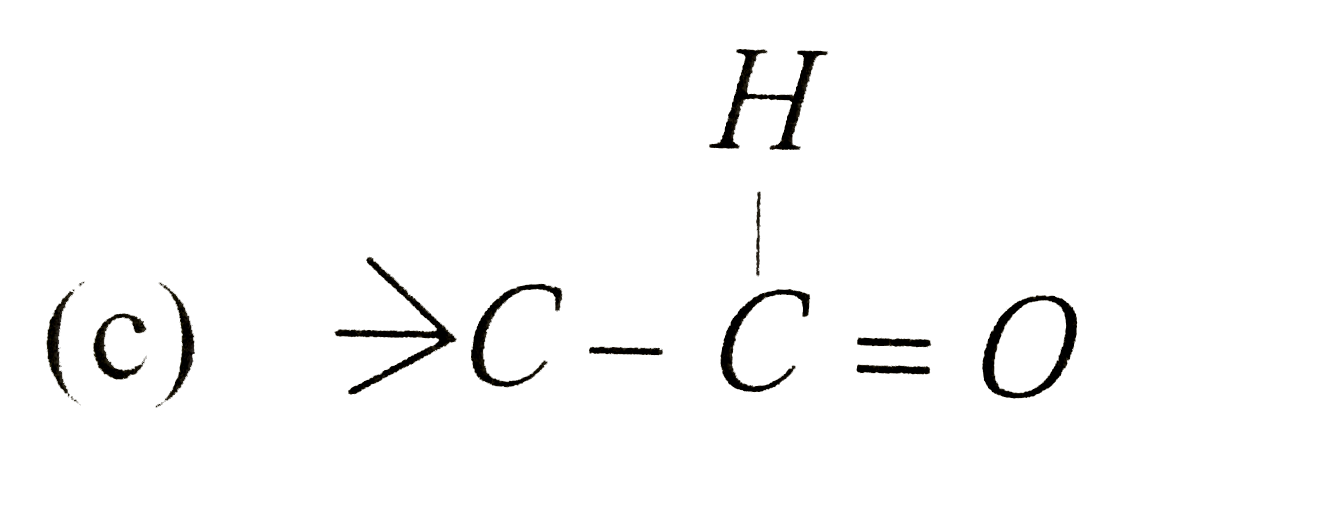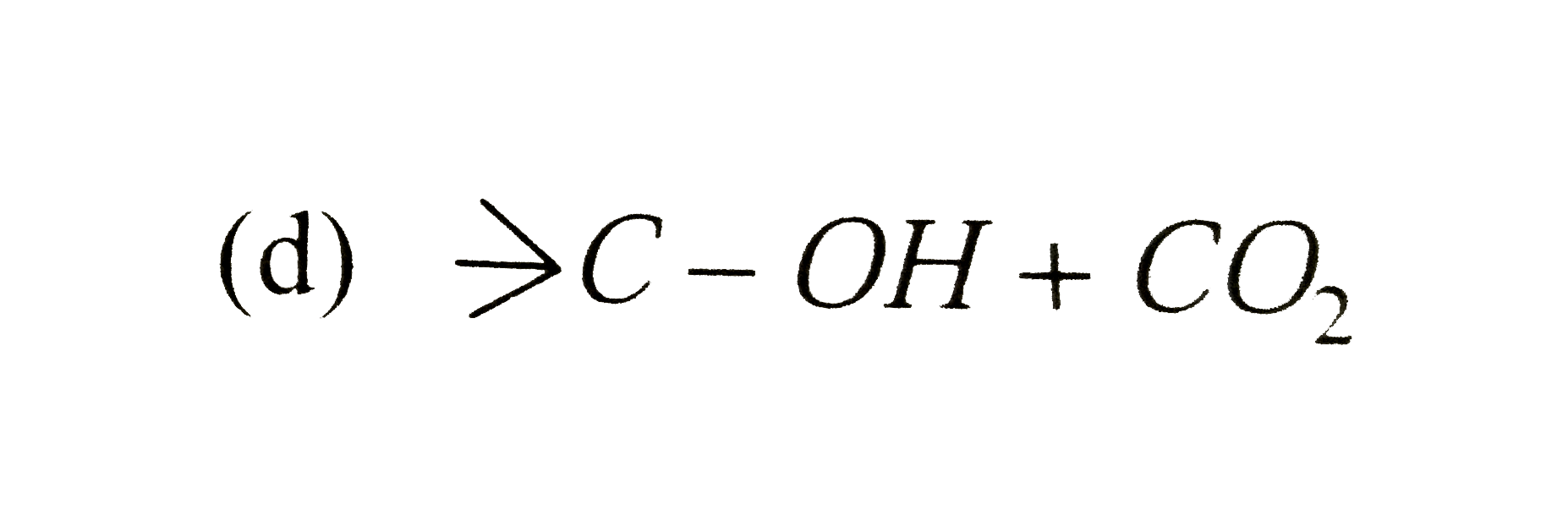Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The pair of compounds in which both the metals are in the highest possibel oxidation state is |
|
Answer» `[Fe(CN)_(6)]^(4-), [Co(CN)_(6)]^(3-)` `[Co(CN)_(6)]^(3-)` i.e. `Co^(3+)` (Highest for Co) `CrO_(2)Cl_(2)` i.e. `Cr(+6)` Highest for Cr `MnO_(4)^(-)` i.e. `Mn(+7)` Highest for Mn `TiO_(2)` i.e. `TI(+4)` Highest for Ti `MnO_(2)` i.e `Mn(+4)` `MnO_(3)` i.e. Mn(+6) |
|
| 2. |
The pair of compounds in which both the compounds give positive test with Tollen's reagent is |
|
Answer» Glucose and Sucrose |
|
| 3. |
The pair of compounds in which both the compounds give positive test Tollens reagent is : |
|
Answer» GLUCOSE and sucrose |
|
| 4. |
The pair of compounds having identical shapes for their molecules is |
|
Answer» `CH_(4), SF_(4)` `ZnCl_(2)` is also linear in shape with 2 bond pairs of electrons. |
|
| 5. |
The pair of compound which cannot exist together in solution is |
|
Answer» `NaHCO_(3) and H_(2)O` `NaHCO_(3) +NaOH to Na_(2)CO_(3) +H_(2)O` |
|
| 6. |
The pair ofcompound whichcannotexist togatherinsolution is |
|
Answer» `NaHCO_(3) and NaOH` `NaHCO_(3) + NaOH rarr Na_(2)CO_(3) +H_(2)O` |
|
| 7. |
The pair of coloured ions among the following is |
|
Answer» `Cu^(+2),Zn^(+2)` |
|
| 8. |
The pair of amphoteric oxides is….. |
|
Answer» VO, `Cr_(2)O_(3)` |
|
| 9. |
The pair in which species have the same magnetic moment is: |
|
Answer» `"["Cr(H_(2)O)^(6)"]"^(2+)[CoCl_(4)]^(2-)` No. of UNPAIRED electrons in : `Cr^(2+),[Ar]3d^(4)`( four) `Co^(2+),[Ar]3d^(7)` (three) `Fe^(2+),[Ar]3d^(6)` (four) `Mn^(2+),[Ar]3d^(5)` (five) Both `Cr^(2+)` and `Fe^(2+)` have four electrons. |
|
| 10. |
The pair in which phosphorus atoms have a formal oxidation state of +3 is- |
|
Answer» Pyrophosphorus and pyrophosphoric acids |
|
| 11. |
The pair in which phosphorous atoms have a formal oxidation state of +3 is : |
|
Answer» Pyrophosphorous and pyrophosphoric acids. Orthophosphoric acid `to H_(3)PO_(4)` OS value= +5 Pyrophosphorous acid `to H_(4)P_(2)O_(5)` OS value = +3 Pyrophosphoric acid `to H_(4)P_(2)O_(7)` OS value = + 5 Hypophosphoric acid `to H_(4)P_(2)O_(6)`, OS value = +4 |
|
| 12. |
The pair in which both the species have the same hybridization is |
|
Answer» `[NI(NH_(3))_(6)]^(2+)` and `[MN(CN)_(6)]^(4-)`  In both COMPLEX, Ni and Co have same hybridization. |
|
| 13. |
The pair in which both the species are used in the preparation of antacid is |
|
Answer» `NaHCO_(3)` and `Mg(OH)_(2)` |
|
| 14. |
The pair in which both species have the same magnetic moment (spin only value) is |
|
Answer» `[CR(H_2O)_6]^(2+) , [CoCl_4]^(2-)` `[Fe(H_2O)_6]^(2+)= Fe^(2+) =3d^6` , Four unpaired electrons. Hence, both the species have same magnetic MOMENT. |
|
| 15. |
The pair in which both species have same magnetic moment (spin only value ) is : |
|
Answer» `[Cr(H_(2)O)_(6)]^(2+), [CoCl_(4)]^(2-)` |
|
| 16. |
The pair in which both species have iron is |
|
Answer» Nitrogenase, CYTOCHROMES |
|
| 17. |
The pair having the same magnetic moment is_________. [ At. No. :Cr = 24, Mn =25 , Fe= 26 , Co =27] |
|
Answer» `[Cr(H_(2)O)_(6)]^(2+) and [CoCl_(4)]^(2-)` |
|
| 18. |
The pair having the same magnetic moment is [At. No. Cr = 24, Mn = 25, Fe = 26, Co = 27] |
|
Answer» `[Cr(H_(2)O)_(6)]^(2+)`and `[CoCl_(4)]^(2-)`  Thus, `[Cr(H_(2)O)_(6)]^(2+)` and `[Fe(H_(2)O)_(6)]^(2+)` have the same number of UNPAIRED electrons and HENCE have same magnetic MOMENT. |
|
| 19. |
The pair having the same magnetic moment is [at. No.Cr = 24, Mn = 25, Fe = 26 " and " Co = 27] |
|
Answer» `[CoCl_(4)]^(2-) and [FeH_(2)O)_(6)]^(2+)` |
|
| 20. |
The pair [Co (NH_(3))_(4) CI_(2) ] Br_(2) and [Co(NH_(3))_(4)]CI_(2) will show |
|
Answer» LINKAGE isomerism. |
|
| 21. |
The pair of compounds in which both the metals are in the highest possible oxidation state is : |
|
Answer» `TiO_3 and MnO_2` |
|
| 22. |
The packing fraction of a simple cubic structure is pi//6. Prove. |
|
Answer» Solution :In a simple cube, the FACE edge length, a = 2R Number of atoms present in one unit cell = 1 Packing fraction = `("volume OCCUPIED by atoms" )/("VOLUMEOF theunitcell" ) =(4/3pir^(3))/(a^(3))=4/3(pir^(3))/(2r)^(3)=(pi)/(6)` |
|
| 23. |
The packing fraction of a simple cubic structure is (pi)/(6). Prove. |
|
Answer» Solution :In a SIMPLE CUBE, the face edge length, `a= 2r`. Number of atoms present in one unit cell `= 1` Packing Fraction `=("VOLUME occupied by atoms")/("Volume of the unit cell")=((4)/(3) pi r^(3))/( a^(3) ) = (4)/( 3) (pir^(3) )/( (2r)^(3))= (pi)/( 6) =0.524` |
|
| 25. |
The packing fraction for a body-centred cubic is |
|
Answer» 0.42 |
|
| 26. |
The packing fraction for a body centred cube |
|
Answer» 0.74 |
|
| 27. |
The packing efficiencyof the two dimensional square unit cell shown below is |
|
Answer» 0.5025 Area of square unit cell `=(2sqrt(2)r)^(2)=8r^(2)` Area of atoms present in one unit cell 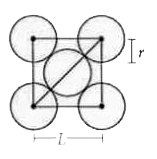 `=PIR^(2)+r((pir^(2))/4)=2pir^(2)` so packing EFFICIENCY `=(2pir^(2))/(8r^(2))XX100` `=(pi)/4xx100=78.5%` |
|
| 29. |
The packing efficiency in a simple cubic cell system of crystals is |
| Answer» ANSWER :C | |
| 30. |
Calculate the efficiency of the packaing in case of face - centered cubic crystal . |
|
Answer» 0.52 |
|
| 31. |
The packaging fration for a body centred cubic structure is |
|
Answer» 0.42 |
|
| 33. |
The P-P-P angle in P_(4) molecule is .......... Degree while S-S-S angle in S_(8) is ........... Degree. |
| Answer» SOLUTION :`60^(@), 107^(@)` | |
| 34. |
The p-p bond energy is 'x' KJ/mole. Then the energy needed for the dissociation of 124 g of white phosphorous is |
|
Answer» X KJ |
|
| 36. |
The ozonolysis of gt C= C ltproduces |
|
Answer» `gt C=O+O =C lt` |
|
| 37. |
The ozonolysis of (CH_(3))_(2) C = C (CH_(3))_(2) followed by treatment with zinc and water will give : |
|
Answer» ACETONE 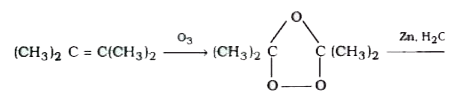
|
|
| 38. |
The ozonolysis of an olefin gives only propanone. The olefin is |
|
Answer» but-1-ene 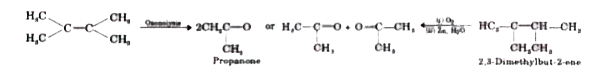
|
|
| 39. |
The ozonolysis of an alkene X followed by hydrolysis gives ethanal and propanone . X is |
|
Answer» 2 - BUTENE |
|
| 40. |
The ozonlysis of isobutene gives: |
|
Answer» `CH_3CHO` |
|
| 41. |
The ozone layer forms naturally by |
|
Answer» the interaction of CFCs with OXYGEN `O_(2(g)) + hv OVERSET(UV)to O_((g)) + O_((g))` ` O_((g)) + O_(2(g))overset(UV) to O_(3(g))` |
|
| 42. |
The oxygen of H_2O_2 used for oxidation is bound by : |
|
Answer» ELECTROVALENT bond |
|
| 43. |
The ozonelayerformsnaturallyby |
|
Answer» theinteraction of CFCwith OXYGEN |
|
| 44. |
The oxygen present today in atmosphere : |
|
Answer» Is a PLANT product |
|
| 45. |
The oxygen and hydrogen formed during electrolysis of water are in the weight ratio of |
|
Answer» `8:1` |
|
| 46. |
The oxygen atom in a either molecule is |
|
Answer» Very active |
|
| 47. |
The oxyacid of SO_2 is : |
|
Answer» `H_2SO_3` |
|
| 48. |
The oxyacid which acts both as oxidising and reducing agent is : |
|
Answer» `H_2SO_4` |
|
| 49. |
The oxyacid of phosphorous in which phosphorus has lowest oxidation state is. |
|
Answer» <P>Hypophosphorus ACID |
|