Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct IUPAC name of (C_2H_5)_4C is : |
|
Answer» TETRAETHYL methane |
|
| 2. |
The correct IUPAC name of AlCl_3(EtOH)_4 is: |
|
Answer» ALUMINIUM(II)chloride-4-ethanoI |
|
| 3. |
The correct IUPAC name of acetonitrile is : |
|
Answer» ETHANENITRILE |
|
| 4. |
The correct IUPAC name of |
|
Answer» Tri `-mu-` CARBONYL bis (tricarbonyl) iron (0) |
|
| 5. |
The correct IUPAC name for the compound (CH_(3))_(3)COH is : |
|
Answer» Trimethylmethan-1-ol |
|
| 6. |
The correct IUPAC name for CH_(3)-CH_(2)-O-overset(CH_(3))overset(|)(CH)-CH_(2)-CH_(2)-CH_(2)Cl is: |
|
Answer» 2-ethoxy-5-chloropentane |
|
| 7. |
The correct IUPAC name for H_(2)C=CH- C-=CH is: |
|
Answer» but-3-en-1-yne |
|
| 8. |
The correct IUPAC name for CH_(2)=CHCH_(2)NHCH_(3) is : |
|
Answer» Allylmethylamine |
|
| 9. |
The correct IUPAC name for CH_2=CHCH_2NHCH_3 is |
|
Answer» Allylmethylamine |
|
| 10. |
The correct IUPAC name for CH_2= CHCH_2 NHCH_3is |
|
Answer» Allylmethylamine |
|
| 11. |
The correct information about the given reaction is : |
|
Answer» The compound (I) MAY show racemisation due to ENOLISATION |
|
| 12. |
The correct increasing order of trans-effect of the following species is - |
|
Answer» `CN^(-) gt BR^(-) gt C_(6)H_(5)^(-) NH_(3)` |
|
| 13. |
The correct increasing order of the acid strength of acids, butyric acid (I), 2-chlorobutyric acid (II), 3-chlorobutyric acid (III) and 2,2-dichlorobutyric acid (IV) is : |
|
Answer» `IltIIltIIIltIV ` |
|
| 14. |
The correct increasing order of solubility among the compounds follows as |
|
Answer» `CaCO_3 LT NaHCO_3 lt KHCO_3` |
|
| 15. |
The correct increasing order of reactivity of halides for S_(N)2 reaction is |
|
Answer» `CH_(3)CH_(2)X LT (CH_(3))_(2)CHX lt CH_(2)=CHCH_(2)X lt PhCH_(2)X` |
|
| 16. |
The correct Increasing order of Reactivity for following molecule towards E.S.R. |
|
Answer» `3gt4gt2gt1` |
|
| 17. |
The CORRECT increasing order of power of precipitation among the following is __________. |
|
Answer» `Cl^(-) LT PO_(4)^(3-) lt SO_(4)^(2-)` |
|
| 18. |
The correct increasing order of dipole moments of the following is |
|
Answer» `H_(2)O LT H_(2)S lt H_(2)SE lt H_(2)TE` |
|
| 19. |
The CORRECT increasing order of dipole moments of the following is _________. |
|
Answer» `H_(2)O LT H_(2)S lt H_(2)Se lt H_(2)TE` |
|
| 20. |
The correct increasing order of basicity of C_(6)H_(5)CH_(2)NH_(2)(A),(B) and is |
|
Answer» A ltBlt C |
|
| 21. |
The correct increasing order of basic strength for the following compounds is |
|
Answer» IgtIIgtIII Basic nature : 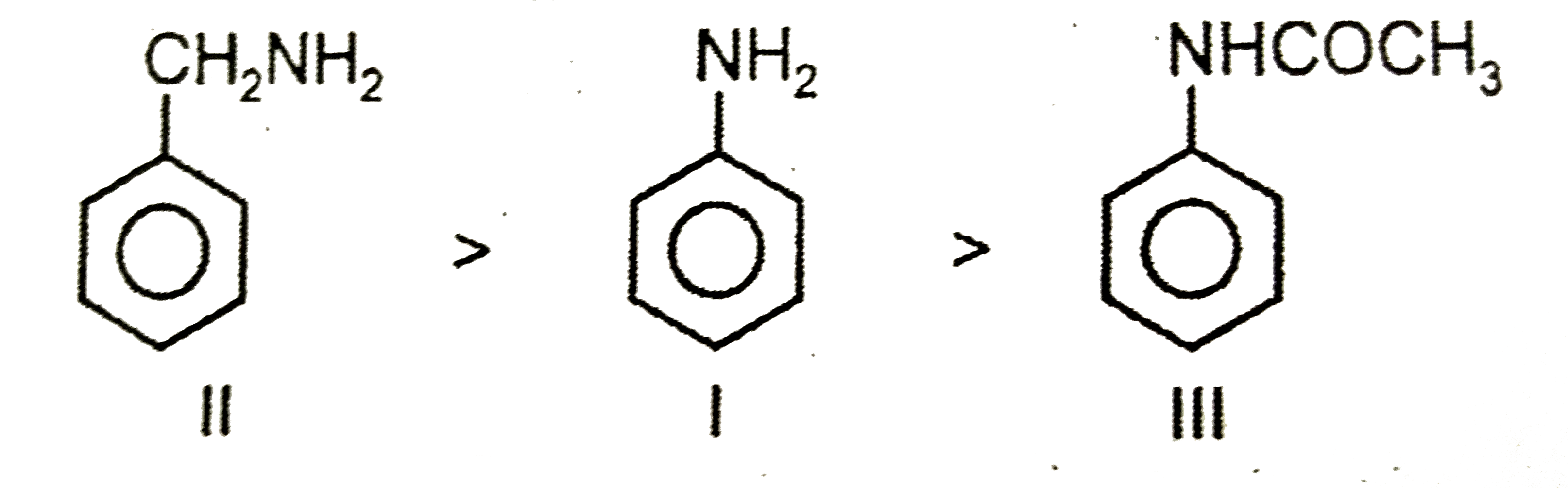
|
|
| 22. |
The correct increasing order of basic strength of following amines, C_(2)H_(5)NH_(2), NH_(3),(C_(2)H_(5))_(2)NH,C_(6)H_(5)NH_(2) in gaseous state is : |
|
Answer» `(C_(2)H_(5))_(2)NHLT C_(2)H_(5)NH_(2)LT NH_(3)lt C_(6)H_(5)NH_(2)` |
|
| 23. |
The correct increasing order of basic strength for the following compound is |
|
Answer» II LT IIIlt I |
|
| 24. |
The correct increasing order acid strength of carboxylic acid is ........... |
|
Answer» `F - CH_2 - COOH GT I - CH_2 - COOH gt Cl - CH_2 - COOH gt Br - CH_2 -COOH` |
|
| 25. |
The correct increasing order of acidic strength is |
|
Answer» `CH_3 OH gt H_2O gt (CH_3)_2 CHOH` `R-O^(+) H_2 overset(Cl^-)to RCl + H_2O ` |
|
| 26. |
The correct hybridization of transitio metal ion/atom is …... |
|
Answer» `K_4[NI(CN)_4]rarrsp^(3)` |
|
| 27. |
The correct graph showing variation of potential energy with internuclear distance when two hydrogen atoms approach each other is |
| Answer» | |
| 28. |
The correct geometry of hybridization for XeF_(4) are |
|
Answer» Octahedral, `sp^(3)d` |
|
| 29. |
The correct geometry and hybridisation for XeF_4are |
|
Answer» OCTAHEDRAL, `sp^3d^2` |
|
| 30. |
The correct formula of Zeise’s salt is: |
|
Answer» `[PtCI_3C_2H_6]^(-)K^+` |
|
| 31. |
The correct formula of salt formed by the neutralization of hypophosphorus acid with NaOH is |
|
Answer» `Na_(3)PO_(2)` `H_(3)PO_(2) + NaOH rarr NaH_(2)PO_(2) + H_(2)O` |
|
| 32. |
The correct formula of salt formed by the neutralization hypophosphorus acid with NaOH is |
|
Answer» `Na_(3)PO_(2)` |
|
| 34. |
The correct formula for diamine silver (I) chloride is : |
|
Answer» `[AG(NH_3)]CI` |
|
| 35. |
The correct formula for tetraamineaquachloridocobalt (III) chloride is : |
|
Answer» `[Co(NH_(3))_(4)(H_(2)O)Cl]Cl_(2)` |
|
| 36. |
The correct features of the enzymes are |
|
Answer» they are mainly GLOBULAR proteins |
|
| 37. |
The correct expression in SI system relating the equivalent conductance (wedge_(aq)) , specific conductance (K) and equivalent concentration (C) is (where C is the number of gram equivalents in one litre of the solution). |
|
Answer» `wedge_(aq)=K/C` |
|
| 38. |
The correct expression the rate of reaction of elementary reaction, A+B to C is |
|
Answer» `(d[C])/(dt)= k [A]` |
|
| 39. |
The correct expression the rate of reaction of elementary reaction ,A+B---->C is: |
|
Answer» `d([C])/dt=K[A]` |
|
| 40. |
The correct expression in SI system relating the equivalent conductance (lamda_(C)), specific conductance (kappa) and equivalent (C) is:- |
|
Answer» `lamda_(C)=(KAPPA)/(C)` |
|
| 41. |
The correct expression for the the rate constant for reactions of zero order is |
|
Answer» `K = [A_0]//2t` |
|
| 42. |
The correct expression for the rate constant for reactions of zero order is |
|
Answer» `K-[A_(o)]//2T` |
|
| 43. |
The correct equation for the degree of association 'alpha' of an associating solute, 'n' molecules of which undergoes association in solution, is |
|
Answer» `alpha = (n(i-1))/(1-n)` `nA HARR (A)_n` `{:("Initial mole","1 mole","0"),("Associated change",-alpha"mole",+alpha//n"mole"),("Equilibrium mole",(1-alpha)"mole",alpha//n"mole"):}` Total NUMBER of moles after association of 1 mole of A `=(1 - alpha) + (alpha //n)`mole Total no. of moles `=1 - alpha + alpha/n` van.t Hoff factor (i) `=("Number of moles after association")/("Normal number of mole taken")` `=(1-alpha + alpha/n)/1` |
|
| 44. |
The correct equation for the degree of association 'alpha' of an associating solute, 'n' molecules of which undergo association in solution is |
|
Answer» `alpha=(N(i-1))/(1-n)` `1-alpha""(alpha)/(n),"Total"=1-alpha+(alpha)/(n)` `therefore i=1-alpha+(alpha)/(n) or 1-i=alpha-(alpha)/(n)=alpha(1-(1)/(n))` `=alpha((n-1)/(n))` `or alpha=(n)/((n-1))(i-i)=(n(i-1))/((1-n))` |
|
| 45. |
The correct electronic configuration of nitrogen is : |
|
Answer» `1s^2 2s^2 2p_x^@ 2p_x^@ 2p_x^@` |
|
| 46. |
the correct electronic configuration of Cu^(2+) ion is |
|
Answer» `[Ar] (3d)^8(4S)^1` |
|
| 47. |
The correct electronic configuration of Cr is…………… |
|
Answer» `[AR] 3d^(4) 4s^(2)` |
|
| 48. |
The correct electronic configuration for Ni^(2=) (Z=28) is |
|
Answer» `[Ar]4S^(2)3d^(6)` |
|
| 49. |
The correct difference between first and second order reactions is that ........... |
|
Answer» A FIRST order reaction can be cataylsed , a SECOND order reaction cannot b cataysed . `t_(1//2)=(0.6932)/k` For a second order reaction `t_(1/2)=(2^(n-1)-1)/((n-1)k[A_0]^(n-1))` n = 2 `t_(1//2)=(2^(2-1)-1)/((2-1)k[A_0]^(2-1))` `t_(1//2)=1/(k[A_0])` |
|
| 50. |
The correct difference between first and second order reactions is that |
|
Answer» A FIRST order reaction can be catalysed, a second order reaction cannot be catalysed. `t_(1//2)=0.6932/k` For a second order reaction `t_(1//2)=(2^(n-1)-1)/((n-1)k[A_(0)]^(n-1)),n=2` `t_(1//2)=(2^(2-1)-1)/((2-1)k[A_(0)]^(2-1)) RARR t_(1//2)=1/(k[A_(0)])` |
|