Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Propyne reacts with HOCl, which of the following cannot be formed |
|
Answer» `CH_(3)CH_(2)CHCl_(2)` 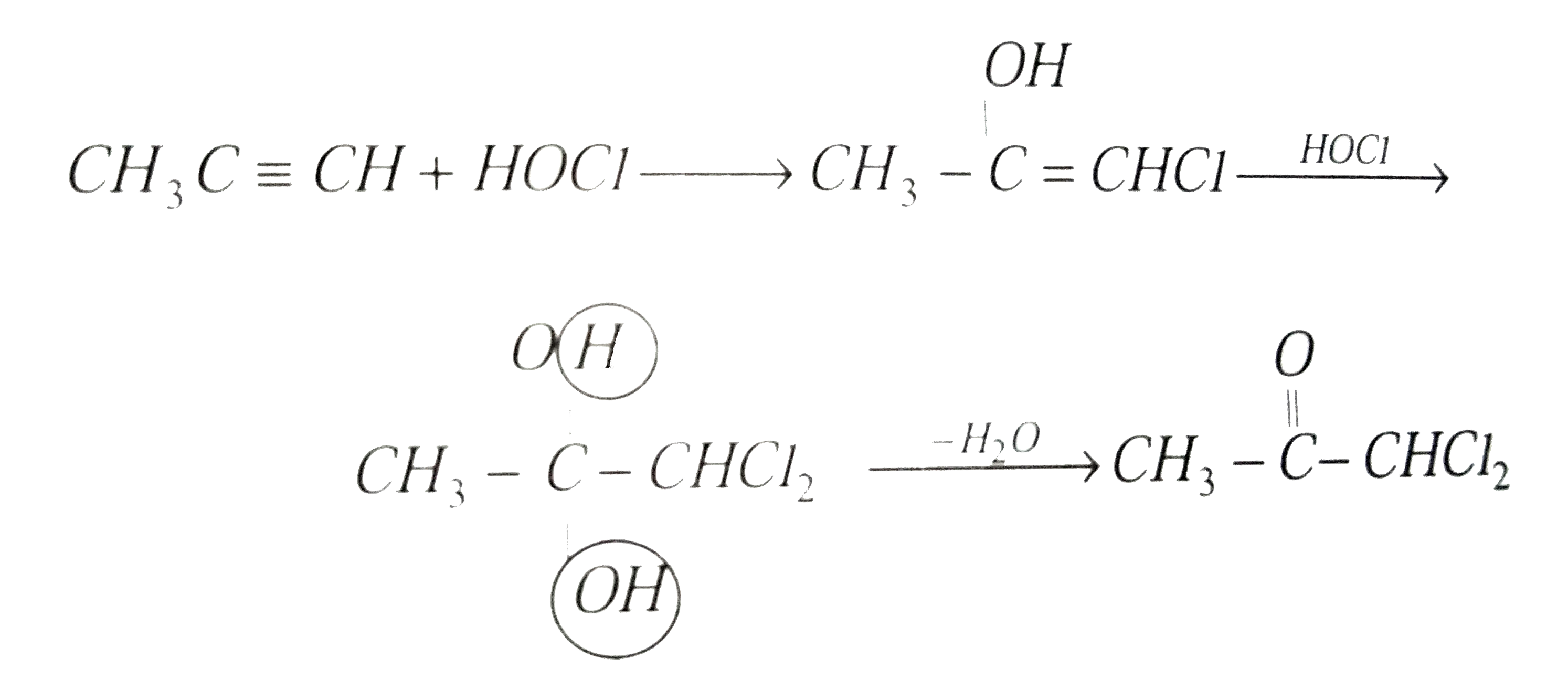 So, (a), (c) and (d) are correct ANSWERS while (b) is incorrect. |
|
| 2. |
Propyne on polymerisation gives |
|
Answer» MESITYLENE  Mesitylene or 1,3,5-trimethyl benzene |
|
| 3. |
Propyne to acetone |
|
Answer» SOLUTION :Propyne to acetone : `UNDERSET("Propyne")(CH_(3)C=CH)+H_(2)Ounderset(1%HgSO_(4))OVERSET(40%H_(2)SO_(4))rarrunderset("alkenol")underset(OH)underset(|)(CH_(3)-C=CH_(2))overset("tautomerisation")rarrunderset("acetone")underset(O)underset("||")(CH_(3)-C-CH_(3))` |
|
| 4. |
Propyne on oxidation with SeO_2 gives: |
|
Answer» CHOCHO |
|
| 5. |
Propyne is formed by the reaction of |
|
Answer» `CH_3 Br` with sodium acetylide |
|
| 6. |
Propyne and propene can be distinguished by? |
|
Answer» conc. `H_(2)SO_(4)` |
|
| 7. |
Propyne and propene can be distinguished by : |
|
Answer» Conc. `H_(2)SO_(4)` |
|
| 8. |
Propylene is passed through conc. H_2SO_4 and the product thus obtained on hydrolysis with boiling water gives |
|
Answer» 1-propanol |
|
| 9. |
Propyl propionate is prepared from which of the following |
|
Answer» `CH_(3)CH_(2)CH_(2)OH,CH_(3)CH_(2)COOH` |
|
| 10. |
Propyl iodide and isopropyl iodide are : |
|
Answer» FUNCTIONAL isomers |
|
| 11. |
Propyl benzene reacts with bromine in the presence of light and heat to give : |
|
Answer»
|
|
| 12. |
Propose the mechanism of the reaction taking place when: (a). (-)-2-Bromooctane reacts with sodium hydroxide to form (+)-Octane-2-ol. (b). 2-Bromopentane is heated with KOH (alc). To form alkene. |
Answer» Solution :(a). In this reaction, there is an inversion of configuration and `OH^(-)` ion (nucleophile) attacks from a side opposite to the side where Br atom is PRESENT. This means that the reaction FOLLOWS `S_(N^(1))` MECHANISM.  (B). In this reaction bimolecular elimination (`E_(2)`-elimination) takes place.  . .
|
|
| 13. |
Propose mechanism of the reaction taking place when (a) (-) - 2- Bromooctane reacts with sodium hydroxide to form (+) -octane -2-ol. (b) 2-Bromopentane is heated with (alc.) KOH to form alkenes. |
Answer» SOLUTION :
|
|
| 14. |
Proportions because |
|
Answer» of its acidic nature |
|
| 15. |
Propiophenone is reduced by NaBH_(4) gives |
|
Answer» 2-phenyl propan-2-ol |
|
| 16. |
Propionic acid with Br_(2)//P yields a dibromo product. Its structure would be |
|
Answer» `CHBr_(2)-CH_(2)COOH` `CH_(3)-overset(alpha)(C)H_(2)-COOH underset(-HBr)overset(Br_(2)//P)CH_(3)-CHBr-COOH underset(-HBr)overset(Br_(2)//P)toCH_(3)-CBr_(2)-COOH` |
|
| 17. |
Propionic acid with Br_(2)//P yield a dibromo product . Its structure will be |
|
Answer» `H-UNDERSET(BR)underset(|)overset(Br)overset(|)(C)-CH_(2)COOH` |
|
| 18. |
Propionic acid with Br,/P yields a dibromo product. Its structure would be |
|
Answer» `H - underset(Br)underset(|)OVERSET(Br)overset(|)C - CH_2COOH` |
|
| 19. |
Propionic acid is obtained from dry ice and what? |
| Answer» Answer :B | |
| 20. |
Propionic acid when treated with Br_(2) in the presence of P in sunlight gives : |
|
Answer» `CH_(2)BrCH_(2)COOH` |
|
| 21. |
Propionic acid on bromination in thepresence of red phosphorus gives 2- bromo propionic acids it shows |
|
Answer» OPTICAL isomers It SHOWS optical isomerism. |
|
| 22. |
Propionic acid is subjected to reduction with hydroiodic acid in the presence of a little P, the product formed is |
|
Answer» Ethane |
|
| 23. |
Propionic acid is obtainedby acidhydrolysis of |
|
Answer» `CH_(3) - CH_(2)-CH_(2)-NO_(2)` |
|
| 24. |
Propionic acid can also be named as |
|
Answer» METHYL methanoic acid |
|
| 25. |
Propionic acid and KOH reacts to produce which one ofthe folliwng? |
|
Answer» Potassium propionate `UNDERSET("Propionic acid")(CH_(3)CH_(2)COOH)OVERSET(KOH)tounderset("Potassium propionate")(CH_(3)CH_(2)COOK+H_(2)O)` |
|
| 26. |
Propionamide when heated with amixture of bromine and caustic alkali wouldgive : |
|
Answer» PROPYLAMINE |
|
| 27. |
Propionaldoxime is reacted with trifluoroperoxy acetic acid gives |
|
Answer» propan-1-amine |
|
| 28. |
Propertyof alkaline earthmetals that increases with their atomic number is |
|
Answer» IONISATION enthalpy |
|
| 29. |
Property of the alkali metals that increases with their atomic number is : |
|
Answer» Ionic molility of their ION in water |
|
| 30. |
Property of the alkaline earth metals that increases with their atomic number is: |
|
Answer» IONISATION enthalpy |
|
| 31. |
Properties, whose values depend only on the concentration of solute particles in solution and not on the identity of the solute are called colligative properties.there may be change in number of moles of solute due to ionisation or association hence these properties are also affected.Number of moles of the product is related to degree of ionisation or association by van Hoff factor 'i' given by i=[1+(n-1)alpha] for dissociation where n is the number of products (ions or molecules ) obtained per mole of the reactant. & i=[1+(1/n-1)alpha] for association where n is number of reactant particles associated to give 1 mole product.A dilute solution contains 't' moles of solute X in 1 kg of solvent with molal elevation constant K_b.The solute dimerises in the solution according to the following equation.The degree of association is alpha. 2X hArr X_2 The equilibrium constant for the process can be expressed as |
|
Answer» `K=(K_b t/(DeltaT_b))/(1-(K_bt)/(DeltaT_f))` Here`{:(2X,hArr,X_2),(t, , 0):}` `1-talpha(talpha)/2{(DeltaT_b,=(1-alpha//2)K_bt),(alpha,=alpha=(2(K_bt-DeltaT_b))/(K_bt)):}` `implies [X_2]=talpha//2` `[X]=t-talpha` `K=(talpha//2)/((t-talpha)^2)=1/(2t^2)xxalpha/((1-alpha)^2)=1/(2t)xxalpha/((1-alpha)^2)=1/(2t)XX((2(K_bt-DeltaT_b))/(K_bt))/(1-((2(K_bt-DeltaT_b))/(K_bt))^2)` Hence , `K=(K_b(K_bt-DeltaT_b))/((2DeltaT_b-K_bt)^2)` |
|
| 32. |
Properties, whose values depend only on the concentration of solute particles in solution and not on the identity of the solute are called colligative properties.there may be change in number of moles of solute due to ionisation or association hence these properties are also affected.Number of moles of the product is related to degree of ionisation or association by van Hoff factor 'i' given by i=[1+(n-1)alpha] for dissociation where n is the number of products (ions or molecules ) obtained per mole of the reactant. & i=[1+(1/n-1)alpha] for association where n is number of reactant particles associated to give 1 mole product.A dilute solution contains 't' moles of solute X in 1 kg of solvent with molal elevation constant K_b.The solute dimerises in the solution according to the following equation.The degree of association is alpha. 2X hArr X_2 The colligaive properties observed will be |
|
Answer» `{:(DeltaP_("THEO")gt,DeltaP_("actual")),(DeltaT_(b("theo"))gt,DeltaT_(b("actual"))),(DeltaT_(f("theo"))gt,DeltaT_(f("actual"))):}` |
|
| 33. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to homegenous solution. These are called colligative properties. Applications of colligative properties are very useful in day to day life. One of its exaples is the use of ethylene glycol and water mixtue as anti-freezing liquid in the radiators of automobiles. A solution M is prepared by mixing ehanol and water. The mole fraction of ethanol in the mixture is 0.9 Given. K_(f)"of water"=1.86 "K kg mol"^(-1) K_(f)"of ethanol" = 2.0 "K kg mol"^(-1) K_(b)"for water "=0.52 "K kg mol"^(-1) K_(b)" of ethanol"=1.2 "K kg mol"^(-1) T_(f)("water")=273 K T_(f)("ethanol")=155.7 K T_(b)^(@) ("water")=373 K T_(b)^(@)("ethanol")=351.5 K P^(@)("water")32.8 mm Hg. P^(@)("ethanol")=40 mmHg M("ethanol")==46 "g mol"^(-1) M("water")=18"g mol"^(-1) In answering the following quesion, consider the solutions to be ideal dilute solutions ans solute to be non-voltile and non-dissoviative. (7) water is added to the solution M such that the mole fraction so wter in solution becomes 0.9. The boiling point of solution is : |
|
Answer» `380.4 K` `"Molality of solution (m)"=("No. of MOLES ethanol")/("Mass of water in kg")` `n_("ethanol")=0.1 `, `W_("water")=((0.9mol)xx(18" g mol"^(-1)))/1000=0.0162 kg.` `m=((0.1mol))/((0.0162 kg))=6.173 "mol kg"^(-1)` =6.173m `DeltaT_(b)=K_(b)xxm=(0.52"k kg mol"^(-1))xx(6.173" mol kg"^(-1))=3.2 K` b.p. of solution = 373+3.2= 376.2 K |
|
| 34. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to homegenous solution. These are called colligative properties. Applications of colligative properties are very useful in day to day life. One of its exaples is the use of ethylene glycol and water mixtue as anti-freezing liquid in the radiators of automobiles. A solution M is prepared by mixing ehanol and water. The mole fraction of ethanol in the mixture is 0.9 Given. K_(f)"of water"=1.86 "K kg mol"^(-1) K_(f)"of ethanol" = 2.0 "K kg mol"^(-1) K_(b)"for water "=0.52 "K kg mol"^(-1) K_(b)" of ethanol"=1.2 "K kg mol"^(-1) T_(f)("water")=273 K T_(f)("ethanol")=155.7 K T_(b)^(@) ("water")=373 K T_(b)^(@)("ethanol")=351.5 K P^(@)("water")32.8 mm Hg. P^(@)("ethanol")=40 mmHg M("ethanol")==46 "g mol"^(-1) M("water")=18"g mol"^(-1) In answering the following quesion, consider the solutions to be ideal dilute solutions ans solute to be non-voltile and non-dissoviative. (5) The freezing point of solution M is: |
|
Answer» `268.7 K` `=("No. of MOLES of WATER")/("Mass of ethanol in kg")` `n_("ethanol")=0.1 mol` `W_("ethnol")=((0.9"mol"))/((0.0414 kg))=2.415 " mol kg"^(-1)` =2.415 m. `DeltaT_(f)=K_(f)xxm=(2.0" K kg mol mol"^(-1))XX(2.415 "mol kg"^(-1))=4.83 K.` f.p. of solution =155.7-4.83=150.87 K. |
|
| 35. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to homegenous solution. These are called colligative properties. Applications of colligative properties are very useful in day to day life. One of its exaples is the use of ethylene glycol and water mixtue as anti-freezing liquid in the radiators of automobiles. A solution M is prepared by mixing ehanol and water. The mole fraction of ethanol in the mixture is 0.9 Given. K_(f)"of water"=1.86 "K kg mol"^(-1) K_(f)"of ethanol" = 2.0 "K kg mol"^(-1) K_(b)"for water "=0.52 "K kg mol"^(-1) K_(b)" of ethanol"=1.2 "K kg mol"^(-1) T_(f)("water")=273 K T_(f)("ethanol")=155.7 K T_(b)^(@) ("water")=373 K T_(b)^(@)("ethanol")=351.5 K P^(@)("water")32.8 mm Hg. P^(@)("ethanol")=40 mmHg M("ethanol")==46 "g mol"^(-1) M("water")=18"g mol"^(-1) In answering the following quesion, consider the solutions to be ideal dilute solutions ans solute to be non-voltile and non-dissoviative. (6) The vapour pressure of solution M is : |
|
Answer» <P>`39.3 MM Hg` =(40 mm Hg)xx(0.9)=36 mm Hg |
|
| 36. |
A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9 Given: Freezing point depression constant of water (K_(f)^(water)=1.86 K kg mol^(-1)) Freezing point depression constant to ethanol (K_(f)^(ethanol))=2.0 K kg mol^(-1)) Boiling point elevation constant of water (K_(b)^(water))=0.52 K kg mol^(-1)) Boiling point elevation constant of ethanol (K_(b)^(ethanol))=1.2 K kg mol^(-1)) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boilingpoint of water = 373 K Standard boilingpoint of ethanol = 351.5 K Vapour pressure of pure water =32.8 mm Hg Vapour pressure of pure ethanol =40 mm Hg Molecular weight of water =18 g mol^(-1) Molecular weight of ethanol =46 g mol^(-1) In anwering the following questions consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. Water is added to the solution M such that the mole fraction of water in the solution becomes 0.9. The boiling point of this solution is |
|
Answer» `380.4 K` `DeltaT_(B)=K_(b)xxm` `=0.52xx(0.1xx1000)/(0.9xx18)=3.2K` `:.`Boiling point, `T_(b)=(373+3.2)K` `=376.2K` |
|
| 37. |
A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9 Given: Freezing point depression constant of water (K_(f)^(water)=1.86 K kg mol^(-1)) Freezing point depression constant to ethanol (K_(f)^(ethanol))=2.0 K kg mol^(-1)) Boiling point elevation constant of water (K_(b)^(water))=0.52 K kg mol^(-1)) Boiling point elevation constant of ethanol (K_(b)^(ethanol))=1.2 K kg mol^(-1)) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boilingpoint of water = 373 K Standard boilingpoint of ethanol = 351.5 K Vapour pressure of pure water =32.8 mm Hg Vapour pressure of pure ethanol =40 mm Hg Molecular weight of water =18 g mol^(-1) Molecular weight of ethanol =46 g mol^(-1) In anwering the following questions consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The vapour pressure of the solution M is |
|
Answer» `39.3 MM Hg` `p= 40 xx 0.9= 36` mm Hg Since it is GIVEN that SOLUTE is taken as NON volatile, WATER does not contribute to the total vapour pressure. |
|
| 38. |
A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9 Given: Freezing point depression constant of water (K_(f)^(water)=1.86 K kg mol^(-1)) Freezing point depression constant to ethanol (K_(f)^(ethanol))=2.0 K kg mol^(-1)) Boiling point elevation constant of water (K_(b)^(water))=0.52 K kg mol^(-1)) Boiling point elevation constant of ethanol (K_(b)^(ethanol))=1.2 K kg mol^(-1)) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boilingpoint of water = 373 K Standard boilingpoint of ethanol = 351.5 K Vapour pressure of pure water =32.8 mm Hg Vapour pressure of pure ethanol =40 mm Hg Molecular weight of water =18 g mol^(-1) Molecular weight of ethanol =46 g mol^(-1) In anwering the following questions consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The freezing point of the solution M is |
|
Answer» `268.7 K` Molality of `H_(2)O=(n_(2)xx1000)/(n_(1)M_(1))` `=(0.1xx1000)/(0.9xx46)=2.415` m `DeltaT_(f)=K_(f)m=2xx2.2415=4.83` FREEZING POINT of solution `= 155.7 - 4.83 = 150.87 K` Total vapour pressure, `p=p_(A)^(@)xx A`. |
|
| 39. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solution molecules ae added to get homogeneous solution. These are called colligative properties. Application colligative properties are very useful in day-to-day life. One of its example is the use of ethylene glycol and water mixture as anti-freezing liquid in the radiator of automobiles A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9 Given: Freezing point depression of water (K_(f)^("water"))=1.86K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol")=2.0K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water")=0.52K kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol")=1.2K kg "mol"^(-1) Standard freezing point of water=273K Standard freezing point of ethanol=155.7K Standard boiling point of water =373K Stadndard boiling point of ethanol =351.5K Vapour pressure of pure water =32.8mm Hg Vapour pressure of pure ethanol=40mm Hg Molecular weight of water =18 g "mol"^(-1) Molecular weight of ethanol =46g "mol"^(-1) In aswering the following questions, consider the solution to be ideal dilute solutions and solutes to be non volatile and non-dissociative. the Henry's law constant for the solubility of N_(2) gas in water jat 298K is 1.0xx10^(5) atm. The mole fraction of N_(2) in air 0.8. The number of moles of N_(2) from air dissoved in 10 moles of water of 298K and 5 atm pressure is : |
|
Answer» `4xx10^(-4)` `x_(N_(2))=(1)/(10^(5)xx0.8xx5=4xx10^(-5)` PER MOLE In `10` mole SOLUBILITY is `4xx10^(-4)`. |
|
| 40. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. one of its examples is the use of ethylene glycol adn water mixtures as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given: Freezing point depression constant of water (K_(f)^(water)) = 1.86 K kg mol^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol")) = 2.0 K kg mol^(-1) Boiling point elevation constant of water (K_(b)^(water)) = 0.52 K kg mol^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol")) = 1.2 K kg mol^(-1) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressure of pure ethanol = 40 mm Hg Molecular weight of water=18 g mol^(-1) Molecular weight of ethanol = 46 g mol^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The vapour pressure of the solution M is |
|
Answer» <P>`39.3 MM HG` `= 40 XX 0.9` `= 36.0 mm Hg` |
|
| 41. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. one of its examples is the use of ethylene glycol adn water mixtures as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given: Freezing point depression constant of water (K_(f)^(water)) = 1.86 K kg mol^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol")) = 2.0 K kg mol^(-1) Boiling point elevation constant of water (K_(b)^(water)) = 0.52 K kg mol^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol")) = 1.2 K kg mol^(-1) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressure of pure ethanol = 40 mm Hg Molecular weight of water=18 g mol^(-1) Molecular weight of ethanol = 46 g mol^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The freezing point of the solution M is |
|
Answer» `268.7K` `= 115.7 - 2 xx (0.1)/(0.9 xx 46//1000)` `= 155.7 - 2 xx 2.415 = 150.9 K` |
|
| 42. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Application of colligative properties are very useful in day- to - day life. One of its examples is the use of ethylene glycol and water mixture as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given : Freezing point depression constant of water (K_(f)^("water"))="1.86 K kg mol"^(-1) Freezing point depression constant of ethanol (e_(f)^("ethanol"))="2.0 K kg mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))="0.52 K kg mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))="1.2 K kg mol"^(-1) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressusre of pure ethanol = 40 mm Hg Molecular weight of water = "18 g mol"^(-1) Molecular weight of ethanol = "46 g mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. Water is added to the solution M such that the mole fraction of water in the solution becomes 0.9. The boiling of the solution is |
|
Answer» 380.4 K Molality of ethanol in the solution `=(0.1)/(0.9xx13)xx1000=6.17` `DeltaT_(B)=K_(b) "(water)"xxm=0.52 xx6.17K=3.2K` `THEREFORE"Boiling point of solution"` `=T_(b)^(@)"(water)"+DeltaT_(b)=373+3.2K=376.2K` |
|
| 43. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogenous solution. These are called colligative properties. Application of colligative properties are very useful in day-to-day life. One of its example is the use of ethylene glycol and water mixture as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixingethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given : Freezing point depression constant of water (K_(f)^("water")) = 1.86 K "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethonal")) = 2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water")) = 0.52 K kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethonal")) = 1.2 K kg mol^(-1) Standard freezing point of water = 273 K Standard freezing point of ethonal = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressure of pure ethonal = 40 mm Hg Molecular weight of water = 18 g "mol"^(-1) Molecular weight of ethonal = 45 g"mol"^(-1) In answering the following questions, consider the solution to be ideal ideal solutions and solutes to be non-volatile and non-dissociative. Water is added to the solution M such lthat the molecules fraction of water in t he solution becomes 0.9. The boiling point of this solution is : |
|
Answer» <P>268.7K `DeltaT_(f)=underset("ethanol")(iK_(f).m)=(2.0)(2.42)=4.84` F. P. of solution` = 155.7 – 4.8 = 150.9` |
|
| 44. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Application of colligative properties are very useful in day- to - day life. One of its examples is the use of ethylene glycol and water mixture as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given : Freezing point depression constant of water (K_(f)^("water"))="1.86 K kg mol"^(-1) Freezing point depression constant of ethanol (e_(f)^("ethanol"))="2.0 K kg mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))="0.52 K kg mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))="1.2 K kg mol"^(-1) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressusre of pure ethanol = 40 mm Hg Molecular weight of water = "18 g mol"^(-1) Molecular weight of ethanol = "46 g mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The vapour pressure of the solution M is |
|
Answer» <P>39.3 MM Hg `=36+3.28=39.28mm~=39.3mm` |
|
| 45. |
Properties such as boiling point, freezing point and vapour pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Application of colligative properties are very useful in day- to - day life. One of its examples is the use of ethylene glycol and water mixture as anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing ethanol and water. The mole fraction of ethanol in the mixture is 0.9. Given : Freezing point depression constant of water (K_(f)^("water"))="1.86 K kg mol"^(-1) Freezing point depression constant of ethanol (e_(f)^("ethanol"))="2.0 K kg mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))="0.52 K kg mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))="1.2 K kg mol"^(-1) Standard freezing point of water = 273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water = 373 K Standard boiling point of ethanol = 351.5 K Vapour pressure of pure water = 32.8 mm Hg Vapour pressusre of pure ethanol = 40 mm Hg Molecular weight of water = "18 g mol"^(-1) Molecular weight of ethanol = "46 g mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The freezing point of solution M is |
|
Answer» 268.7 K i.e., 0.9 mole of ethanol is mixed with 0.1 mole of water, i.e., water is the SOLUTE and ethanol is the solvent. `therefore"MOLALITY of water in the solution"` `=(0.1)/(0.9xx46)xx1000=2.415` `DeltaT_(F)=K_(f)"(ethanol)"xxm=2.0xx2.415=4.83K` `therefore"FREEZING point of the solution (ethanol)"` `=T_(f)^(@)"(ethanol)"-DeltaT_(f)=155.7-4.83=150.87K` = 150.9K |
|
| 46. |
Properties such as boiling point, freezing point and vapour, pressure of a pure solvent change Propeties such as boiling point, freezing point and vapour, pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. The freezing point of the solution M is |
|
Answer» `268.7 K` `2xx(0.1)/(0.9xx46)xx100=4.83K` FREEZING point of solution `M=155.7-4.83=150.87 K=150.9K` |
|
| 47. |
Properties such as boiling point, freezing point and vapour, pressure of a pure solvent change Propeties such as boiling point, freezing point and vapour, pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative.Water is added to the solution M such that the fraction of water in the solution becomes 0.9. The boiling point of this solutions is |
|
Answer» `380.4 K` `0.52xx(0.1)/(0.9xx18)xx1000=3.2 K` `T_(b)=373+3.2=376.2 K` |
|
| 48. |
Properties such as boiling point, freezing point and vapour, pressure of a pure solvent change Propeties such as boiling point, freezing point and vapour, pressure of a pure solvent change when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative. when solute molecules are added to get homogeneous solution. These are called colligative properties. Applications of colligative properties are very useful in day-to-day life. One of its examples is the use of ethylene glycol and water mixtureas anti-freezing liquid in the radiator of automobiles. A solution M is prepared by mixing athanol and water. The mole fraction of ethanol in the mixture is 0.9 GivenFreezing point depression constant of water (K_(f)^("water"))=1.86 K kg "mol"^(-1) Freezing point depression constant of ethanol (K_(f)^("ethanol"))=2.0 K kg "mol"^(-1) Boiling point elevation constant of water (K_(b)^("water"))=0.52 kg "mol"^(-1) Boiling point elevation constant of ethanol (K_(b)^("ethanol"))=1.2 kg "mol"^(-1) Standard freezing point of water =273 K Standard freezing point of ethanol = 155.7 K Standard boiling point of water =373 K tandard boiling point of ethanol =351.5 K Vapour pressure of pure water =32.8 mmHg Vapour presure of pure ethanol =40g Hg Molecular weight of water =18 g"mol"^(-1) Molecules weight of ethanol =46 g "mol"^(-1) In answering the following questions, consider the solutions to be ideal dilute solutions and solutes to be non-volatile and non-dissociative.The vapour pressure of the solution M is |
|
Answer» `39.3 MM HG` |
|
| 49. |
Properties of elements X: (i) Its amide is used as a reducing agent (ii) Its amalgam is used as reducing agent Properties of element Y: (i) It is an octaatomic molecule (ii) It is diamagnetic in ground state but paramagnetic in vapour phase Properties of element Z: (i) Its diatomic molecule is paramagnetic (ii) Its allotrope is an absorber of UV radiation (iii) IT supports combustion Which statement is not correct about these following compounds? |
|
Answer» COMPOUND of X and Z is BASIC in aq solution |
|
| 50. |
Properties of elements X: (i) Its amide is used as a reducing agent (ii) Its amalgam is used as reducing agent Properties of element Y: (i) It is an octaatomic molecule (ii) It is diamagnetic in ground state but paramagnetic in vapour phase Properties of element Z: (i) Its diatomic molecule is paramagnetic (ii) Its allotrope is an absorber of UV radiation (iii) IT supports combustion Compound of X,Y and Z on reaction with Y form a compound A. Out of the following which statement is not correct about compoundA? |
|
Answer» used in iodometry |
|



