Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Properties of elements X: (i) Its amide is used as a reducing agent (ii) Its amalgam is used as reducing agent Properties of element Y: (i) It is an octaatomic molecule (ii) It is diamagnetic in ground state but paramagnetic in vapour phase Properties of element Z: (i) Its diatomic molecule is paramagnetic (ii) Its allotrope is an absorber of UV radiation (iii) IT supports combustion Compound of X and Z + Compound of Y and Z rarr product(s). Oxidation number of Y in the final compound may be: |
| Answer» Answer :A | |
| 2. |
Properties like variety of oxidation states, formation of coloured ions and complex compound formation are due to |
|
Answer» PARTLY FILLED s-orbitals. |
|
| 3. |
Properties common to the elements manganese, iron, cobalt, nickel nad copper include the ready formation by them all of (I) coloured ions in aqueous solution (II)oxides of nitrogen are formed on reaction with concentrated HNO_(3) (III) chlorides of formula MCl_(2) and MCl_(3) |
|
Answer» I, II, III |
|
| 4. |
Properties of elements are periodic function of number of present in the nucleus |
|
Answer» Protons |
|
| 5. |
Properties associated with teflon are |
|
Answer» It is chemically merit. |
|
| 6. |
Propene when heated with chlorine at about 500^@ C forms |
|
Answer» `CH_2Cl.CH=CH_2` This REACTION is called allylic HALOGENATION reaction because halogenation OCCURS at the allylic POSITION of an alkene |
|
| 7. |
Propene,CH_3CH=CH_2 can be converted into 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion: |
|
Answer» `H_2O` |
|
| 8. |
Write the relavant chemical equations : Propene to propyne |
| Answer» Solution :`CH_(3)-CH=CH_(2) underset(C Cl_(4))overset(Br_(2))rarr CH_(3)-overset(overset(BR)(|))(CH)-overset(overset(Br)(|))(CH_(2)) underset(KOH)overset("Alc")rarr CH_(3)-C -= CH` | |
| 9. |
Write the relavant chemical equations : Propene to nitropropane |
| Answer» Solution :`CH_(3)-CH= CH_(2) UNDERSET("peroxide")overset(HBr)rarr CH_(3)-CH_(2)-CH_(2)-BR overset(AgNO_(2))rarr CH_(3)-CH_(2)-CH_(2)-NO_(2)` | |
| 10. |
Propene to 1-bromopropane |
|
Answer» Solution :PROPENE to 1-bromopropane : `underset("Propene")(CH_(3)-CH)=CH_(2)+HBroverset("PEROXIDE")rarrunderset("1-Bromopropane")(CH_(3)-CH_(2))-CH_(2)-Br` |
|
| 11. |
Write the relavant chemical equations : Propene to 1-propanol |
| Answer» Solution :`CH_(3)-CH=CH_(2) underset(2.H_(2)O_(2)//OH^(-))overset(1.BH_(3))RARR CH_(3)-CH_(2)-CH_(2)-OH` | |
| 12. |
Propene to 2-bromopropane |
|
Answer» SOLUTION :PROPENE to 2-bromopropane : `UNDERSET("Propene")(CH_(3))-CH=CH_(2)+HBroverset("dark")rarrunderset("2-Bromopropane")(CH_(3)-overset("Br ")overset("|")(CH)-CH_(3))` |
|
| 13. |
Propene on treatment with HI gives |
|
Answer» Only `CH_(3)CH_(2)CH_(2)I` |
|
| 14. |
Propene reacts with HBr in the presence of a peroxide to form : |
|
Answer» n-propyl bromide |
|
| 15. |
Propene reacts with Cl_2 at 400-600^@Cto give: |
|
Answer» 1,2 - dichloropropane |
|
| 16. |
Propene is treated with bromine in the presence of brine (aqueous NaC).Choose the correct statement regarding the above reaction. |
|
Answer» TOTAL TWO products are formed.<BR>Total THREE products are formed. `C-overset(Cl)overset(|)underset(H)underset(|)C-C-Br,C-overset(H)overset(|)underset(OH)underset(|)C-C-Br,C-overset(OH)overset(|)underset(H)underset(|)C-C-Br,` |
|
| 17. |
Propene on hydroboraction-oxidation produces |
|
Answer» `CH_(3)CH_(2)CH_(2)OH` |
|
| 19. |
Propene is not the product obtained by dehydration of : |
|
Answer» 2-Propanol |
|
| 20. |
Propene is the product obtained by dehydration of |
|
Answer» 2-propanol |
|
| 21. |
Propene is more reactive than ethene towards HBr because : |
|
Answer» propene can more readily undergo a FREE RADICAL chain reaction |
|
| 22. |
Propene, CH_(3) - CH = CH_(2) can be converted to 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion |
|
Answer» Alkaline `KMnO_(4)` `3CH_(3)CH=CH_(2)+(1)/(2)B_(2)H_(6)underset("ETHER")overset("dry")to (CH_(3)CH_(2)CH_(3))B` `(CH_(3)CH_(2)CH_(3))B overset(H_(2)O_(2))to 3CH_(3)CH_(2)CH_(2)-OH` |
|
| 23. |
Propene, CH_(3)-CH = CH_(2), can be converted to 1-propanol by oxidation. Which set of reagents among the following is ideal to effect the conversion? |
|
Answer» ALKALINE `KMnO_4` |
|
| 24. |
Propene can be converted into 2-propanol by hydration. Which of the following reagents is ideal to affect the conversion ? |
|
Answer» Alkaline `KMnO_4` |
|
| 25. |
Propanone reacts with iodine in acid solution as shown in this equation. CH_(3)C(O)CH_(3)+I_(2)overset(H^(+))rarrCH_(3)C(O)CH_(2)I+HI These data were obtained when the reaction was studied. |{:([CH_(3)C(O)CH_(3)]","M,[I_(2)]","M,[H^(+)]","M,"Relative Rate"),(0.010,0.010,0.010,1),(0.020,0.010,0.010,2),(0.020,0.020,0.010,2),(0.020,0.010,0.020,4):}| What is the rate equation for the reaction? |
|
Answer» RATE =`k[CH_(3)C(O)CH_(3)][I_(2)]` |
|
| 26. |
Propanone to propene |
Answer» SOLUTION :
|
|
| 27. |
Propanone on reaction with alkyl magnesium bromide followed by hydrolysis will not produce. |
|
Answer» PRIMARY alcohol |
|
| 28. |
Propanone is usually characterised by ________ |
|
Answer» FEHLING's solution |
|
| 29. |
Propanone is the product obtained by dehydrogenation of …………………. . |
|
Answer» Propan-2-ol |
|
| 30. |
Propanone is the product obtained by dehydrogenation of |
|
Answer» propanal `2^(@)` alcohols on dehydrogenation i.e., removal of `H_(2)` gives OXIDATION product-ketones. |
|
| 31. |
Propanone does not undergo: |
|
Answer» OXIME FORMATION |
|
| 32. |
Propanone can be prepared from ethyne by |
|
Answer» passing a mixture of ethyne and steam over a catalyst, magnesium at `420^(@)C` |
|
| 33. |
Propanone can be obtained from oxidation of what ? |
|
Answer» propan -2-ol |
|
| 34. |
Propanol is heated with conc. H_(2)SO_(4) at 170^(@)C and the gas produced is reacted with HBr. What is the formula of the final product ? |
|
Answer» `CH_(3) - CH_(2) CH_(2)Br` `CH_(3) underset(Br)underset(|)(CH) - CH_(3)` (Markovnikov.s rule). |
|
| 35. |
Propanoic acid to acetic acid |
| Answer» Solution :`CH_(3) COOH overset(NAOH)(to) CH_(3) CH_(2) COONa overset(NaOH)underset(CaO)(to) C_(2) H_(6) overset(Cl_(2)//h epsilon) (to) C_(2) H_(5) CL overset(AGOH) (to) C_(2) H_(5) OH overset((O))underset(KMnO_(4)//H^(+))(to) CH_(3) COOH` | |
| 36. |
Propanoic acid is the oxidative product of , |
| Answer» Answer :B | |
| 37. |
Propanoic acid on warming withCl_2 in presence of red P gives : |
|
Answer» `CH_3CH_2Cl` |
|
| 38. |
Propanoic acid is slightly weaker than acetic acid because |
|
Answer» methyl GROUP is ELECTRON with drawing |
|
| 39. |
Propanenitrile is obtained by the action of alcholic KCN and what ? |
|
Answer» `CH_(3)CL` |
|
| 40. |
Propanoic acd undergoes HVZ reaction to give chloropopanoic acid.The product obtained is |
|
Answer» as stronger as PROPANOIC acid Stonger acid than propanoic acid because of the presence of electron WITHDRAWING -Cl group |
|
| 41. |
Propane on dichlorinatin gives......isomers and out of them .....are optically active. |
|
Answer» `4,1` 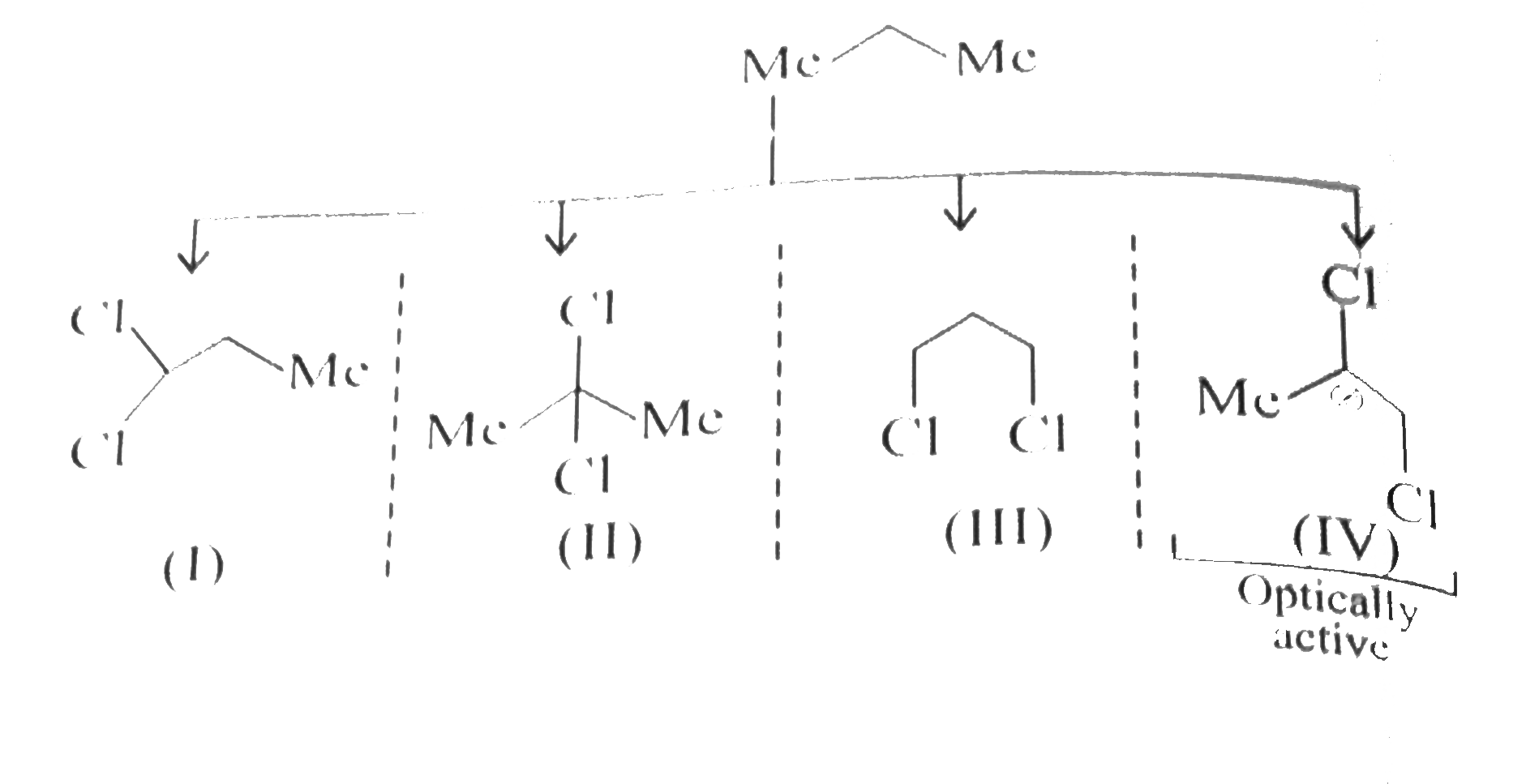
|
|
| 42. |
Propanenitrilereactswith Ato giveB . ThecompoundB onadditionof HCNgives C .ThecompoundC onreductiongive 1-Amino-2- ethyl-4-methyl -2- pentanolwhich of thefollowis A gt |
|
Answer» `(CH_(3))_(3)CCH_(2) MgX` `(A)` `CH_(3) CH_(2) C= Ooverset(HCN) (to)CH_(3) CH_(2) - overset(OH) overset(|)underset(CH_(2) CH (CH_(3))_(2))(C )- CH` `overset("reduction") (to)CH_(3) CH_(2)- overset( OH) overset(|)underset( CH_(2)) CH(CH_(3))_(2) overset(|) (C ) - CH_(2) NH_(2)` `1-` amino-2-ethyl -4-methyl-2-pentanol |
|
| 43. |
Propane is treated with methyl magnesium bromide and them subjected to hydrolysis. Write the final product. |
| Answer» SOLUTION :`UNDERSET("2-methylpropan-2-ol")((CH_3)_3-COH)` | |
| 44. |
Propane + Cl_(2)overset("UV light")toAoverset(Na//"ether")toB. The compound (B) is. |
|
Answer» n-hexane |
|
| 45. |
Propane-1-ol and Propane-2-ol are distinguish by which method ? |
|
Answer» Ozonolysis |
|
| 46. |
Propane cannot be prepared from which reaction |
|
Answer» `CH_3-CH=CH_2underset(OH^-)OVERSET(B_2H_6)to ` `R-CH=CH_2overset(B_2H_6)to (R-CH_2-CH_2)_3 -B overset(OH^-)to R-CH_2-CH_2OH` |
|
| 48. |
Propanal on treatment with dilute sodium hydroxide gives |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)CHO` |
|
| 49. |
Propanal and propanone undergoes condensation reaction in presence of dil . KOH to form, |
|
Answer» `CH_(3)CH_(2)CH(OH)CH_(2)COCH_(3)` `CH_(3)CH_(2)CHO + HCH_(2)COCH_(3) overset("dil . NaOH") tounderset(" 4- HYDROXY 2- hexanone")( CH_(3)CH_(2)CH(OH) CH_(2)COCH_(3))` |
|
| 50. |
Propan-2-ol to 2-methylpropan-2-ol. |
| Answer» Solution :`underset("Propan-2-ol")(CH_(3)-OVERSET(OH)overset(|)(C)H-CH_(3)) underset("or "C_(5)H_(5)NH^(+)CrO_(3)Cl^(-))overset(Cu//573K)to underset("Propanone")(CH_(3)-overset(O)overset(||)(C)-CH_(3)) underset((ii)" "H_(3)O^(+))overset((i)" "CH_(3)MgBr)to underset("2-Methylpropan-2-ol")(CH_(3)-underset(OH)underset(|)overset(CH_(3))overset(|)(C)-CH_(3))` | |