Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On passing electric current in to a solution of salt of metal, M the reaction at cathode takes place as : M^(2+) + 2e^(-) to M. The atomic weight of M is 65. What is the equivalent weifht of the metal ? |
|
Answer» 260 `therefore ` Equivalent weight `=("ATOMIC Weight")/("VALENCY")=(65)/(2)=32.5` |
|
| 2. |
On passing electric current through molten aluminium chloride, 11.2 litre of Cl_(2) is liberated at NTP at anode. The quantity of aluminium deposited at cathode is (at. Wt. of Al=27) |
|
Answer» 9g |
|
| 3. |
On passing a current through a KCl solution, 19.5g of potassium is deposited. If the same quantity of electricity is passed through a solution of aluminium chloride, the amount of aluminium deposited is |
|
Answer» 4.5 G |
|
| 4. |
On passing CrO_2 Cl_2 in water and then adding (CH_3 COO)_2Pb, the precipitate formed is |
|
Answer» `PbCrO_4` |
|
| 5. |
On passing CrO_(2)Cl_(2) through NaOH and then adding (CH_(3)COO)_(2)Pb, the ppt. formed is |
|
Answer» `PbCl_(2)` |
|
| 6. |
On passing CrO_(2)Cl_(2) in water & then adding (CH_(3)COO)_(2) Pb, the ppt formed is |
|
Answer» `PbCrO_(4)` |
|
| 7. |
On passing C ampere of current for time t sec through 1 litre of 2(M) CuSO_(4) solution (atomic weight of Cu=63.5), the amount of m of Cu (in g) deposited on cathode will be:- |
|
Answer» `m=(Ct)/((63.5xx96500))` `mpropCt` or `m=ZCt` where C=current, t=time `Z=("Equivalent weight of metal")/(96500)` EQ. WT of `Cu=(63.5)/(2)""(becauseCu^(2+)toCu)` `Z=(63.5)/(2xx96500)""thereforem=(63.5xxCxxt)/(2xx96500)=(31.75xxCxxt)/(96500)` |
|
| 8. |
On passing certain charge through molten alminium chloride produced 18 gm of Al at cathode . The volume of chloride produced at N.T.P. at anode is |
|
Answer» `11.2 dm^(3)` `THEREFORE 1,93,000C=2xx35.5=71` G of `Cl = 22.4 dm^3` |
|
| 9. |
On passing an electric current, the quantity of a metal deposited on cathode depends upon |
|
Answer» shape of cathode |
|
| 10. |
On passing a current of 1.0 ampere for 16 min and 5 seconds through 1 L solution of CuCl_(2), all copper of the solution was deposited at cathode. The molarity of CuCl_2 solution was |
|
Answer» 0.1M `2 xx 96500 C -= 1 mol Cu=1 mol CuCl_(2)` `Q =1 xx t =1 xx (60 xx 16 +5) = 965 C` ` therefore965 ` C will deposit `-= (965)/(2 xx 96500)=(1)/(2000)` mole `CuCl_(2)` `therefore` MOLARITY of `CuCl_(2)` solution `= (1)/(200)` mol `L^(-1)` = 0.005 M |
|
| 11. |
On passing a current of 1.0 ampere for 16 min and 5 sec through one litre solution of CuCl_(2), al copper of the solution was deposited at the cathode. Calculate the strength of CuCl_(2)solution (Molar mass of Cu=63.5, faraday constant=96500 C mol^(-1)) |
|
Answer» SOLUTION :`Q=Ixxt=1xx965=965C` `Cu^(2+)+2E^(-)toCu` `therefore965C` decompose `CuCl_(2)=(1)/(2xx96500)xx965=(1)/(200)`MOLE=0.005 mole This was PRESENT in 1 L of `CuCl_(2)` solution, Hence, strength=0.005M. |
|
| 12. |
On passing a current of 1.0 ampere for 16 min and 5 sec through one litre solution of CuCl_(2) , all copper of the solution of CuCl_(2) solution was (Molar massof Cu = 63.5 , Faraday constant = 96500 C mol^(-1)). |
|
Answer» 0.07 M `Cu^(2+) + 2e^(-) to Cu`, `2 xx 96500` C decompose `CuCl_(2) = 1 ` mole `THEREFORE` 965 C decompose `CuCl_(2) = (1 xx 965)/(2 xx 96500) =(1)/(200)` mole This was PRESENT in one litre of `CuCl_(2)` solution . HENCE , strength = 0.005 M. |
|
| 13. |
On passing96,500 coulombsof electricitywhichof the followingmetalslatsolutionswillliberateone gramatomof metal ? |
| Answer» Solution :K is monovalent metal. | |
| 14. |
On passing 9.65 amperes of electricity for 1000 seconds, 2.0 grams of metal deposits . The molar of metal is |
|
Answer» Solution :`E=Zxx96,500` `=(W)/(It) = 96,500 ` =`(2)/(9.65 XX 1,000) xx96,500 =20` |
|
| 15. |
On passing 3 amperes of electricity for 50 minutes, 1.8 gm metal deposits. The equivalent mass of metal is |
|
Answer» 20.533 |
|
| 16. |
On passing 3 faraday of electricity through the three electrolytic cells connected in series containing Ag^+, Ca^(2+) and Al^(3+) ions respectively. The molar ratio in which the three metal ions are liberated at the electron is |
|
Answer» `6:3:2` |
|
| 17. |
On passing 3 faraday of electricity through the three electrolytic cells connected in series containing Ag^+,Ca^(2+) and AI^(3+)ions respectively. The molar ratio in which the three metal ions are liberated at the electrodes is : |
|
Answer» `1:2:3` |
|
| 18. |
On passing 3F electricity through three cells containing fused Na_2CO_3 fused Cu(NO_3)_2and fused AI(NO_3)_3, the no. of moles of the metals deposited are in the ratio |
|
Answer» `1:2:3` |
|
| 19. |
On passing 1.5 F charge, the number of moles of aluminium deposited at cathode are [Molar mass of Al 27 gram mol^(-1)] |
|
Answer» 1 |
|
| 20. |
On passing 1 Faraday charge through the electrolytic cells containing Ag^(+),Ni^(2+),Cr^(3+) ions solution, the mass of deposited Ag is (At mass = 108 u ), Ni (At mass = 59u) and Cr (At mass = 52U): |
|
Answer» `{:(,"Ag","NI","cr"),((1),108 " gm",29.5 " gm",17.3" gm"):}` |
|
| 21. |
On passing 1.5 F charge, the number of moles of aluminium deposited at cathode are- [Molar mass of Al = 27 gram "mol"^(-1)] |
|
Answer» `1.0` |
|
| 22. |
On ozonolysis, an organic compound (A) C_(6)H_(10) gives two aldehyde (B) C_(2)H_(4)O and(C ) C_(2)H_(2)O_(2). Determine the structure of A, B and C. |
|
Answer» (2) OZONOLYSIS of `(A)` suggests that it has two double bonds. `underset((A))(C_(6)H_(10))overset("Ozonolysis")rarr underset((B))(C_(2)H_(4)O)+underset((C ))(C_(2)H_(2)O_(2))` Since, `(B)` and `(C )` are aldehydes, i.e., `CH_(3)CHO` and `underset("ETHANOL(B)")(CH_(3)CHO)` and `underset("Ethane-1,2-dial (C )")(CHO.CHO)` `:. (A)` is" " `underset("Hexa-2,4-diene")(CH_(3)CH=CH-CH=CHCH_(3))` |
|
| 23. |
On ozonolysis one mole of a hydrocarbon yields 2 mole of formaldehyde. The hydrocarbon is: |
|
Answer» But-2-ene |
|
| 24. |
On ozonolysis a hydrocarbon of molecular formula C_(4)H_(6) gives succinin acid only. What is the structure of hydrocarbon ? |
Answer»  `overset(OZONOLYSIS)rarrunderset("Succinic ACID")UNDERSET(CH_(2)-COOH)overset(CH_(2)-COOH)|` `overset(OZONOLYSIS)rarrunderset("Succinic ACID")UNDERSET(CH_(2)-COOH)overset(CH_(2)-COOH)|`
|
|
| 25. |
On oxidative ozonolysis of 3-Methylhex-3-ene, two products A & B are formed. A gives CO_(2) gas with sodium bicarbonate, but B can not. The structures of A & B rae respectively : |
|
Answer» `CH_(3)-CH_(2)-underset(O)underset(||)(C )-CH_(3)- & CH_(3)-CH_(2)-COOH` 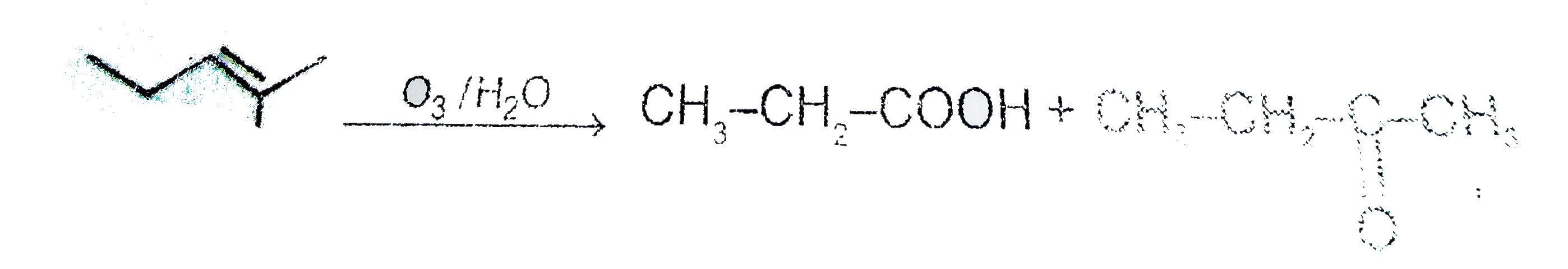
|
|
| 26. |
On oxidationwith KMnO_(4)in acidicmediumSO_(2) isoxidised to______ |
|
Answer» `SO_(2)` |
|
| 27. |
On oxidation,an alcohol gives an aldehyde having the same number of carbon atoms as that of alcohol. |
| Answer» Answer :A | |
| 28. |
On oxidation with iodine, sulphite ion is transformed to |
|
Answer» `S_(4)O_(6)^(2-)` |
|
| 29. |
On oxidation with iodine, sulphite ion is transformed to ……………… . |
|
Answer» `S_(4)O_(6)^(2-)` |
|
| 30. |
On oxidation of alcohol gives an acid having the same number of carbon atoms. The alcohol is, |
| Answer» Answer :A | |
| 31. |
On oxidation of an alcohol gives an aldehyde having the same number of carbon atoms as that of alcohol. The alcohol is |
|
Answer» `1^(@)` ALCOHOL |
|
| 32. |
On moving down the group, the reducing power of alkaline earth metals. |
|
Answer» decreases |
|
| 33. |
On moving from left to right across a period in the periodic table the size of the atom generally: |
|
Answer» decreases |
|
| 34. |
On moving down the group, stability of +1 oxidation stateof the members of boron family __________ , while that of +3 oxidation state __________ . |
| Answer» SOLUTION : INCREASES, DECREASES | |
| 35. |
On moving down the group 13, density ________ |
|
Answer» DECREASES |
|
| 36. |
On mixing two reactants, the substance that reacts completely is called ………………whereas the other iscalled…………. . |
| Answer» SOLUTION :LIMITING REAGENT, EXCESS reagent. | |
| 37. |
On monochlorination of 2-methylbutane, the total number of chiral compounds is: |
|
Answer» 2 |
|
| 38. |
On monochlorination of 2-methy butane, the total number of chiral compounds is |
|
Answer» 2 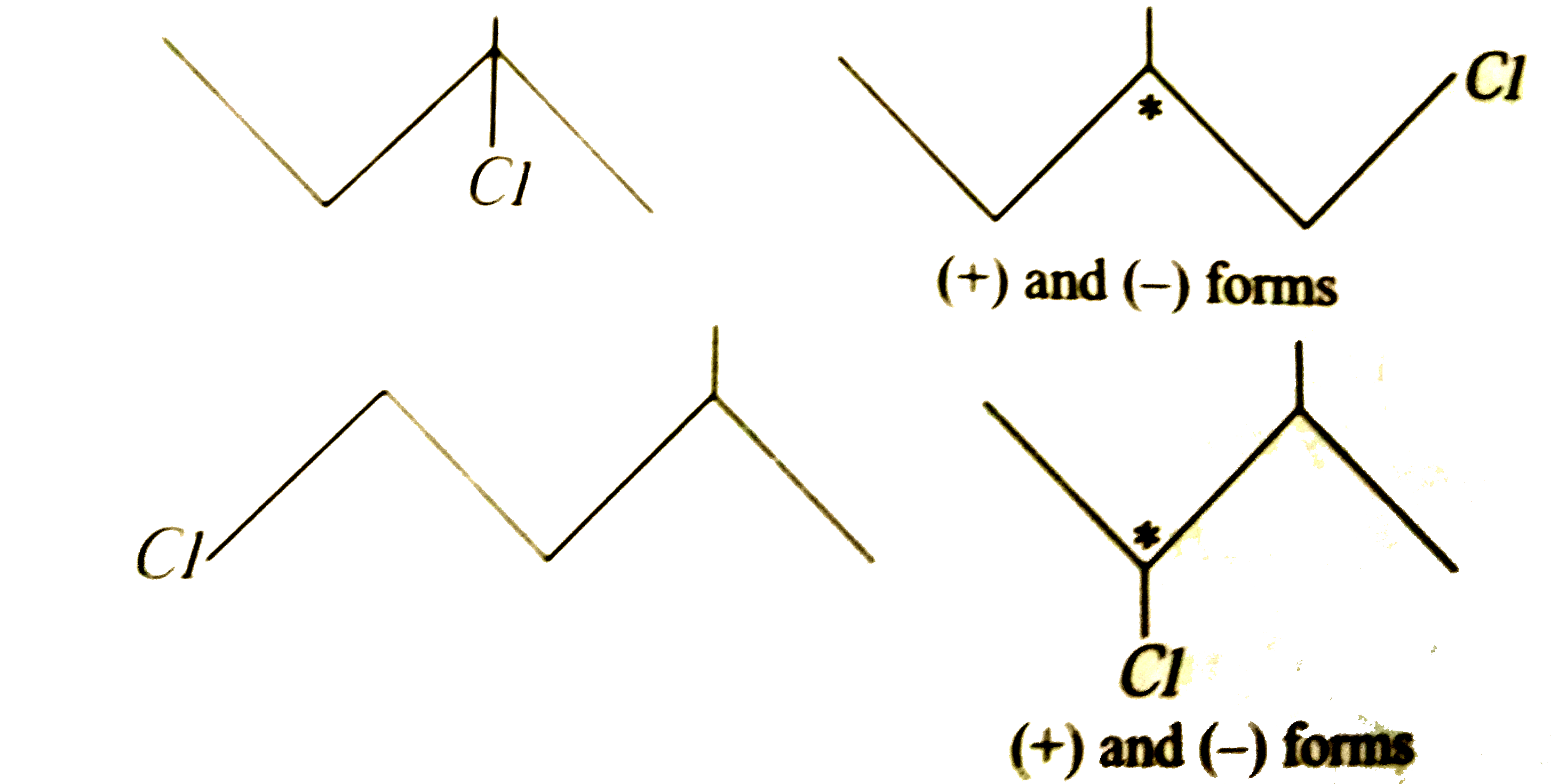 THEREFORE, a TOTAL of four CHIRAL compounds are OBTAINED. |
|
| 39. |
On mixing tetraethyl lead to gasoline available at petrol pumps |
|
Answer» CALORIFIC value of the FUEL INCREASES |
|
| 40. |
On mixing , heptate and octane form an ideal solution. At 373 K. The vapour pressure of the two liquid components (heptaane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure of the solution obtainde by mixing 25.0 g of heptane and 35 g of octane will be (molar mass of heptane=100 g mol^(-1) ) |
|
Answer» 0.0186 K Since `alpha` is eaual to 1 `"Molality (m)"=((0.01mol))/(1kg)` `DeltaT_(f)=ixxK_(f)xxm` `=3XX(1.86 " K kg"MOL^(-1)) xx (0.01 mol kg^(-1))` =0.0558 K. |
|
| 41. |
On mixing, heptane and octane form an ideal solution. At 373 K. The vapour pressure of the two luquid components (heptane and octane) are 105 kPa and 45 kParepectively. Vapour pressure of the solution obtained by mixing 25.0 g of heptane and 35 g of octane will be (molar mass of heptane= 100 g mol^(-1) and of octane= 114 g mol^(-1)) |
|
Answer» <P>144.5 kPa ` X_("OCTANE")=((35g))/((114 G mol^(-1)))=0.30 "mol"` `X_("heptane")=(0.25)/(0.25+0.30)=0.45` `"Xoctane"=(0.30)/(0,25+0.30)=0.55` `P=(0.45xx105 "K pa")xx(0.55xx45"K pr")` `=47.25+24.75=72 "K pa"`. |
|
| 42. |
On mixing, heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components (heptane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure of the solution obtained by mixing 25.0 g of heptane and 35 g of octane will be (molar mass of heptane =100 and octane = 114 g mol^(-1)) |
|
Answer» 72.0 kPa |
|
| 43. |
On mixing solutions of A and B, the solution formed is warm. What type of deviation is shwon ? |
| Answer» SOLUTION :NEGATIVE DEVIATION from RAOULT's LAW. | |
| 44. |
On mixing, heptane and octane form an ideal solution. At 373 K, the vapour pressure of the two liquid components ( heptane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure of the solution obtained by mixing 25.0 g of heptane and 35 g of octane will be ( molar mass of heptane = 100 "g mol"^(-1) and of octane = 114 "g mol"^(-1) ): |
|
Answer» 96.2 KPA `x_(h)=(0.25)/(0.25 + 0.307)=0.45` `:.x_(0)=1-0.45=0.55` `p=x_(h)p_(h)^(@)+x_(o)p_(o)^(@)` `=0.45xx105+0.55xx45` `=47.25+24.75=72` kPa |
|
| 45. |
On mixing equal volumes of acetone and ethanol, what type of deviation from Raoult's law is expected? |
| Answer» SOLUTION :POSITIVE DEVIATION | |
| 46. |
On mixing equal volumes of acetone and ethanol, what type of deviation from Raoult' s low is expected ? |
| Answer» SOLUTION :POSITIVE DEVIATION. | |
| 47. |
On mixingcertainalkanewithchlorineandirradiatingitwithUltravioet light, itformsonlyonemonochloro alkane . thealkanexouldbe : |
|
Answer» PROPANE |
|
| 48. |
On mixing an alkane with chlorine and irradiating with ultra-violet light,it forms only one mono-chloroalkane. The alkane is |
|
Answer» propane `underset(" "CH_(3))underset("|")overset(" "CH_(3))overset("|")(H_(3)C-C-CH_(3))overset(Cl_(2)//UV)rarrunderset(""CH_(3))underset("|")overset(""CH_(2)Cl)overset("|")(H_(3)C-C-CH_(3))` |
|
| 49. |
On mixing certain alkane with chlorine and irradiating it with ultraviolet light ,one forms only one monochloro alkane .The alkane could be : |
|
Answer» neopentane |
|