Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On the basic of data given below, predict which of the following gases shows least adsoprtion on a definite amount of charcoal ? {:("Gas", CO_(2), SO_(2), CH_(4), H_(2)), ("Critical temp/K", 304, 630, 190, 33):} |
|
Answer» `CO_(2)` |
|
| 2. |
On the addition of mineral acid to an aqueous solution of borax, the compound formed is : |
|
Answer» ORTHOBORIC acid |
|
| 3. |
On the addition of a solution containing CO_(4)^(2–) ions to the solution of Ba^(2+), Sr^(2+) and Ca^(2+) ions, the ppt obtained first will be of- |
|
Answer» `CaCO_(4)` |
|
| 4. |
On t reatingPCl_(5)with conc. H_(2)SO_(4), SO_(2)Cl_(2) is formed as the final product this showsthat H_(2)SO_(4) |
|
Answer» is DERIVATIVEOF `SO_(2)` |
|
| 5. |
On sulphonation of C_(6)H_(5)Cl |
|
Answer» m-Chlorobenzenesulphonic ACID is formed |
|
| 6. |
On sulphonation of C_6H_5CI |
|
Answer» m-chlorobenzenesulphonic ACID |
|
| 7. |
On sulhonation , phenol gives two products, ortho and para phenol sulphonic acids . The variation in productsdepend upon the __________. |
|
Answer» CONCENTRATION of sulphuric acid |
|
| 8. |
On strongly heating anhydrous calcium acetate gives : |
| Answer» Answer :A | |
| 9. |
On strongly heating AgNO_3 we get : |
|
Answer» `N_2O` and NO |
|
| 10. |
On strongly heatingAgNO_3 the gases evolved are: |
|
Answer» `N_2O` and NO |
|
| 11. |
On strong heating lead nitrate gives : |
|
Answer» `PBO,NO,O_2` |
|
| 12. |
On strong heating CaO and C the products formed are: |
|
Answer» CA and CO |
|
| 13. |
On strong heating 4 gm of a solid compound produced 528 ml of a diatomic gas (A_(2)) at NTP condittion and 2.68 gm of solid residue. The atomic mass of element A is - |
|
Answer» Solution :`X overset(DELTA)(rarr) A_(2) (g) + Y` `4 gm "" 2.68 gm` `A_(2) (g) = 4 - 2.68 = 1.32gm` `n_(A_(2)) (g) = (V ("gas") NTP "in litre")/(22.4) = (w ("gas") H_(2))/(M.Wt.A_(2))` `n_(A_(2)) (g) = (528)/(22400) = (1.32)/(M.Wt.A_(2))` `M.Wt.A_(2) = 56 gm = 2 xx` Atomic mass of A Atomic mass of `A = 28 gm` |
|
| 14. |
On strong heating ammonium acetate gives - |
|
Answer» Acetamide |
|
| 15. |
On solidification of molten silver gives ………….. crystals. |
|
Answer» SHOWS ccp type of arrangement |
|
| 16. |
On reduction with Na-Hg and water a carbohydrate gives a mixture of sorbitol and mannitol. The carbohydrate can be |
| Answer» Solution :The carbohydrate can be fructose | |
| 17. |
On (reductive) ozonolysis of natural rubber, 4-oxo-pentanal is obtained the monomer of natural rubber would be : |
|
Answer» `H_(2)C=UNDERSET(CH_(3))underset(|)(CH)-CH=CH_(2)` |
|
| 18. |
On reduction with LiAlH_4 a ketone yields : |
|
Answer» PRIMARY alcohol |
|
| 19. |
On reduciton with Sn+conc. Hclof C_(2)H_(5)NO_(2)yields |
|
Answer» ESTERS |
|
| 20. |
On reduction , primary amine is formed by |
|
Answer» NITROETHANE |
|
| 21. |
On reaction with water, calcium phosphide produces |
|
Answer» `Ca_(3)(PO_(4))_(2)` |
|
| 22. |
On reaction with Cl_(2), phosphorus forms two types of halides 'A' and 'B'.Halide A is yellowish-white powder but halide 'B' is colourless oily liquid. Identify A and B and write the formulas of their hydrolysis products. |
|
Answer» Solution :Since `Cl_(2)` reacts with phosphorus to form two halides (A and B), therefore, these halides must be `PCl_(5) and PCl_(3)`. Since halide 'A' is a yellowish-white solid but halide 'B' is a colourless oily liquid, therefore, due to HIGHER molecular mass and hence stronger FORCES of attraction, halide 'A' must be a solid, i.e., `PCl_(5)` and `{:(P_(4)+10Cl_(2) rarr 4 underset((A))(PCl_(5))("yellowish-white solid")),(P_(4)+6Cl_(2) rarr underset((B))(4 PCl_(3))("Colourless oily liquid")):}` halide 'B' must be a liquid, i.e., `PCl_(3)`. Their hydrolysis products are shown below : `{:(underset("Phosphorus pentachloride")(PCl_(5))+4H_(2)O rarr underset("Orthophosphoric acid")(H_(3)PO_(4))+5HCl),(underset("Phosphorus trichloride")(PCl_(3))" "+3H_(2)O rarr underset("Phosphorus acid")(H_(3)PO_(3))""+3HCl):}` |
|
| 23. |
On reaction with ketone with hydroxyl amine give ketoxime which on reduction produces |
| Answer» Answer :B | |
| 24. |
On reaction with dil. H_(2)SO_(4) which of the following salts will give out a gas that turns an acidified dichromate paper green? |
|
Answer» `Na_(2)CO_(3)` `FeS+H_(2)SO_(4) to FeSO_(4)+H_(2)S uarr` `H_(2)S` and `SO_(2)` can REDUCE dichromate PAPER to green `Cr^(3+)` |
|
| 25. |
On reaction with hot conc.H_2SO_4, which of the following compounds loses a molecule of water : |
|
Answer» `CH_3COCH_3` |
|
| 26. |
on reaction with cold conc. HBr gives : |
|
Answer»
|
|
| 27. |
On reaction with Cl_2, phosphorus forms two types of halides 'A' and 'B' Halide A is yellowish-white powder but halide 'B' is colourless oily liquid. Identify A and B and write the formulas of their hydrolysis products. |
|
Answer» Solution :`A: PCl_(5)` `B: PCl_(3)` `PCl_(3)` Hydrolyse to give `H_(3)PO_(3)` and `PCl_(5)` hydrolysis to give `H_(3)PO_(4)` `P_(4) + 10Cl_(2) to 4PCl_(5)` `PCl_(5) + 4H_(2)O to H_(3)PO_(4) + 5HCl` `P_(4) + 6Cl_(2) to 4PCl_(3)` `PCl_(3) + 3H_(2)O to H_(3)PO_(3) + 3HCL` |
|
| 28. |
On reaction with Cl_2, phosphorus forms two types of halides A and B. Halide A is yellowish white powder but halide B is colourless oily liquid. Identify A and B and write the formulas of their hydrolysis products. |
|
Answer» SOLUTION :A is `PCl_5` ( It is yellowish-white POWDER) `P_4 + 10 Cl_2 to 4PCl_5` B is `PCl_3` ( It is a colourless OILY liquid ) `P_4 + 6Cl_2 to 4PCl_3` Hydrolysis products are formed as follows: `PCl_3 + 3H_2O to H_3PO_3 + 3HCL` `PCl_5 + 4H_2O to H_3PO_4 + 5HCl` |
|
| 29. |
On reacting with aqueous bromine at room temperature phenol forms which of the following ? |
|
Answer» meta-Bromophenol |
|
| 30. |
On reacting with neutral ferric chloride, phenol gives |
| Answer» Solution :violet colour | |
| 31. |
On reacting with electropositivemetal like Na, alcohols give sodium alkoxides on liberates H_2. In this reaction alcohols behave as _________. |
|
Answer» weak ACIDS |
|
| 32. |
On reacting copper with dilute nitric acid gives a colourless gas (A). The gas (A) on reacting with Fe^(2+) ionin concentrated sulphuric acid and water forms a brown coloured complex. The EAN of Iron in the brown coloured complex is : [Given: Atomic No.: Fe = 26] |
|
Answer» 26 The oxidation number of Iron in `[Fe(H_(2)O)_(5)(NO)]^(2+)rArr x + (5xx0)+1=+2 rArr x = +1` EAN of Fe = Atomic no. of metal - no. of electrons lost + no. of electrons gained from ligands `= 26-1+12rArr 37` |
|
| 33. |
On reacting with halogens, H_2S is |
|
Answer» oxidized |
|
| 34. |
On Pluto, where everything is frozwn, astronauts discovered two froms of butane gauche and anti. Assuming that there are no rotations around single bonds, which statement about the two forms is correct ? |
|
Answer» They are enantiomers 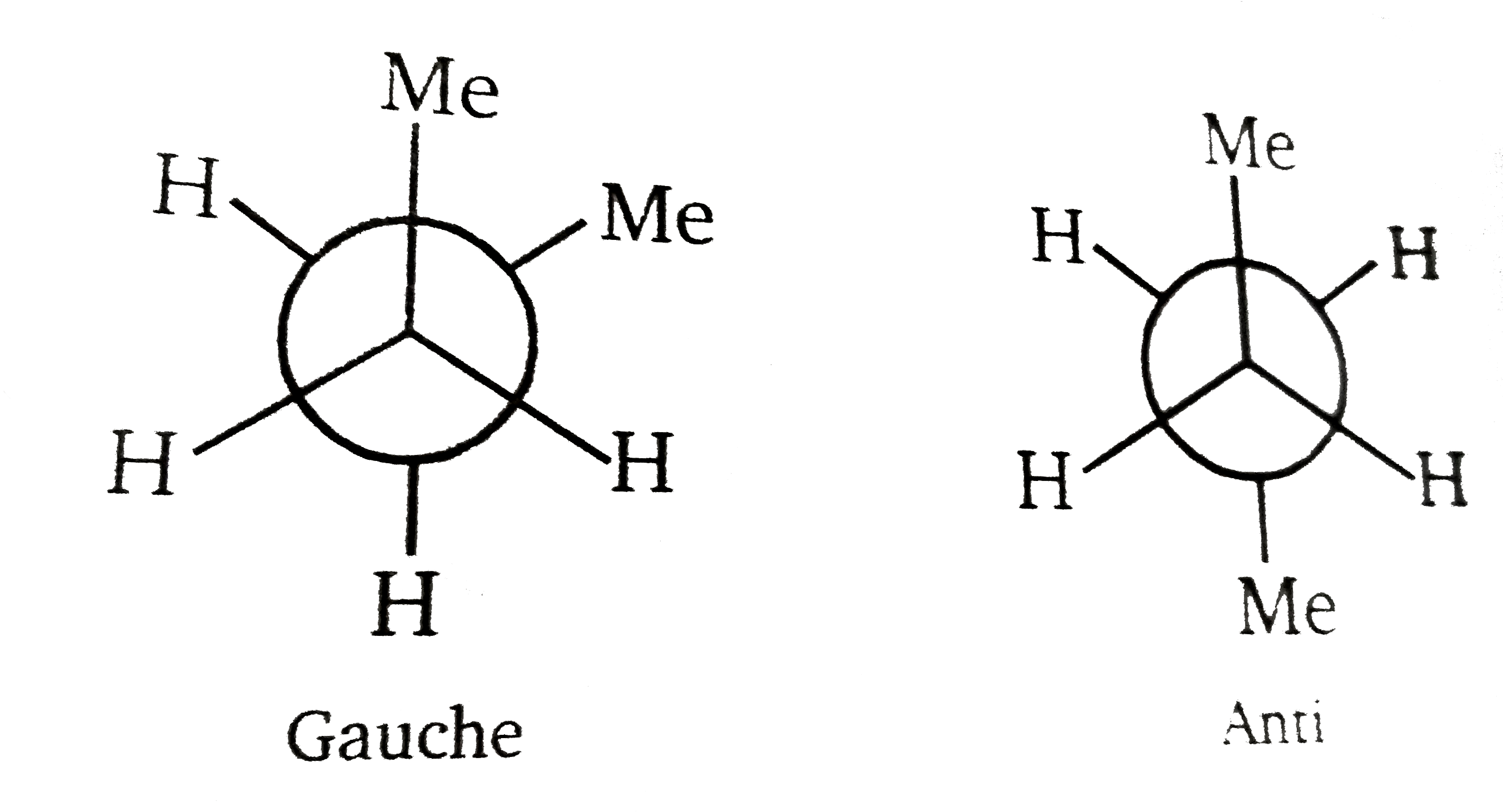 If no ROTATION then these two are not INTER convertible and are diastereoisomer. |
|
| 35. |
On prolonged exposure to air, sodium finally changes to: |
|
Answer» `Na_2CO_3` |
|
| 36. |
On Pauling scale which of the following does not have electronegativity ge3.0 |
|
Answer» Oxygen |
|
| 37. |
On performing a borax-bead test with a given inorganic mixture for qualitative analysis, the colour of the bead was found to emerald green both in oxidising and reducing flame. It indicates the possibility of the presence of |
|
Answer» `Co^(+2)` |
|
| 38. |
On passing vapours of an organic liquid over finely divided Cu at 573K the product was an alkene. This reaction is |
|
Answer» CATALYTIC OXIDATION of primary alcohol |
|
| 39. |
On passingle C ampere of electricity through a electrolyte solution for t second, m gram metal deposits on cathode. The equivalent weight E of the metal is |
|
Answer» `E=(Cxxt)/(mxx96500)` |
|
| 40. |
On passingH_2S in II gp, sometimes a white turbidity is formed. This is due to: |
| Answer» Answer :A | |
| 41. |
On passing through a magnetic field, the greatest deflection is experienced by : |
| Answer» Answer :B | |
| 42. |
On passing SO_(2) gas through an acidified solution of K_(2)Cr_(2)O_(7) |
|
Answer» The Solution GETS decolourised |
|
| 43. |
What happens when H_2S passed through FeCl_3 solution ? |
|
Answer» `FeCl_2` |
|
| 44. |
On passing H_(2)S gas through a highly acidic solution containing Cd^(2+) ions, CdS is not precipitated because |
|
Answer» Of common ION effect |
|
| 45. |
On passing H_2S through HNO_3 we get |
|
Answer» Colloidal SULPHUR |
|
| 46. |
Draw structure of N_2O_5 and also find numbers of lone pairs in N_2O_5 :- |
| Answer» | |
| 47. |
On passing H_2 S into saturated solution of BaCl_2, white ppt. obtained is of |
|
Answer» hydrogen chloride |
|
| 48. |
On passing H_(2)S black ppt. of II group is obtained. The mixture may not contain |
|
Answer» `Pb^(++)` |
|
| 49. |
On passing excess of CO_(2) in lime water, its milky appearance disappears because- |
|
Answer» Soluble `Ca(OH)_(2)` is formed |
|
| 50. |
On passing electricity through diluteH_2SO4 solution the amount of substance libeated at the cathode and anode are in the ratio: |
|
Answer» `1:8` |
|