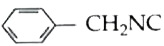Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On mixing a certain alkane with chlorine and irradiating it with ultraviolet light, it forms only one monochloroalkaane. This alkane could be |
|
Answer» pentane `CH_(3)-underset(CH_(3))underset(|)OVERSET(CH_(3))overset(|)C-CH_(3)` |
|
| 2. |
On mixing 10 mL of carbon tetrachloride with 10 mL of benzene the total volume of the solution is: |
|
Answer» `gt20 ML` |
|
| 3. |
On mixing 1 mole of C_(6)H_(6)(P^(@)=42mm) and 2 mole of C_(7)H_(8)(P^(@)=36mm) one can conclude |
|
Answer» total VAPOUR PRESSURE of mixture =38mm |
|
| 4. |
On mixing 10 mL of acetone with 40 ml of chlorofrom, the total volume of the solution is: |
| Answer» Answer :A | |
| 5. |
On mixing 1 mole of C_(6)H_(6) (P^(0) = 42 mm) and 2 mole of C_(7)H_(8) (P^(0) = 36 mm), one can conclude |
|
Answer» total VAPOUR pressure of MIXTURE = 38 mm |
|
| 6. |
On mixing 1 mole of C_(6)H_(6) (P^(@) = 42 mm) and 2 mole of C_(7)H^(6) (P^(@) = 36 mm). One can conclude |
|
Answer» total vapour pressure of MIXTURE = 38 mm |
|
| 7. |
On litre of 0.15 M Na_(2)SO_(3) aqueons solution is mixed with 500 mL of 0.2 M K_(2)Cr_(2)O_(7) aqueous solution in acid medium. What is the number of moles of K_(2) Cr_(2)O_(7) remaining in the solution after the reaction ? |
|
Answer» 0.1 `M=(M_(1)V_(1)+M_(2) V_(2))/(V_(3))` `M=(0.15xx1+0.2xx0.5)/(1 cdot 5)` M=0.15 mol/L Remaining mole of `K_(2) Cr_(2)O_(7) =0.2-0.15` `=0.05` mol/L |
|
| 8. |
On long standing bleaching powder undergoes auto oxidation and converts into |
|
Answer» CALCIUM CHLORIDE `+` Calcium perchlorate |
|
| 9. |
On long exposure to air ethanol becomes sour in taste, because of - |
|
Answer» Bacteria |
|
| 10. |
On Kumbh Mela, Solar Eclipses, festivals and other religious/social events, fairs are held on the banks of holy ponds/rivers for some stipulated days. Lacs of people participate in these fairs to pay homage to their deities. For the convenience of the pilgrims, make-shift, toilets are made in the 'Fair Area' by the district administration. in and around these toilets, a white powder is often, sprinkled. Read the above passage and answer the following questions: |
|
Answer» SOLUTION :(i). The common NAME of the COMPOUND contained in the white powder is p,p'-dichlorodiphenyltrichloroethane abbreviiated as DDT. its CHEMICAL name is 2,2-bis (4-chlorophenyl)-1,1,1-trichloroethane. it is prepared by the action of conc. `H_(2)SO_(4)` on a mixture of chlorobenzene and chloral. (ii) it is used to kill mosquitoes, LICE and other insects which spread diseases. |
|
| 11. |
On increasing the temperature , the rate of the reaction increases because of |
|
Answer» Decrease in the NUMBER of collisions |
|
| 12. |
On increasing the temperature ,the rate of the reaction increases because of ….. |
|
Answer» DECREASE in the NUMBER of collisions |
|
| 13. |
On increasing the temperature, the rate of reaction increases because |
|
Answer» the activation ENERGY of the REACTION increases |
|
| 14. |
On increasing the temperature, the pKw |
|
Answer» INCREASE |
|
| 15. |
On increasing the temperature by 10 K in the case of slow reactions: |
|
Answer» No. of COLLISIONS get DOUBLED |
|
| 16. |
On increasing temperature , surface tension of water |
|
Answer» Increases |
|
| 17. |
On increasing temperature, activation energy for a reaction decrease. Why? |
| Answer» SOLUTION :ACTIVATION ENERGY and temperature are inversely proportional to each other. | |
| 18. |
On increasing temperature, the value of Henry's constant ……… |
|
Answer» decreases |
|
| 19. |
On increasing temparature of the reacting system by 10 degrees, the rate of reaction almost doubles. The most appropriate reason for this is : |
|
Answer» COLLISION frequencyincreases |
|
| 20. |
On increasing pressure, ice melts at a tempreatue lower than its usual melting point because |
|
Answer» the BONDS break, under PRESSURE |
|
| 21. |
On increasin g the tempearture by 10K in the case of slow reactions |
|
Answer» number of collisions get doubled |
|
| 22. |
On igniting Fe_(2)O_(3) at 1400^(@)C, the product obtained is |
|
Answer» `Fe_(2)O_(3)` melts |
|
| 23. |
On hydrolysis with dil. H_2SO_4, starch and cellulose give 'X' and 'Y'. Then 'X' and 'Y' are a pair of |
|
Answer» ENANTIOMERS |
|
| 24. |
On hydrolysis , which produces only glucose |
|
Answer» GALACTOSE |
|
| 25. |
On hydrolysis of starch, we finally get |
|
Answer» GLUCOSE |
|
| 26. |
On hydrolysis, PCl_(5) gives ………………. . |
|
Answer» `H_(3)PO_(3)` on heating undergoes SELF - OXIDATION reduction as : |
|
| 27. |
On hydrolysis of starch by dilute acids we get finally |
|
Answer» GLUCOSE and fructose |
|
| 28. |
On hydrolysis of salts of strong acid and strong base, the solution obtained is neutral.Justify your answer with a suitable example. |
|
Answer» Solution :Let us consider the REACTION between `NaOH` and NITRIC acid to give sodium nitrate and water. `NaOH_((aq))+HNO_(3(aq)) to NaNO_(3(aq))+H_2O(I)` The salt `NaNO_3` completely dissociation in water to produce `Na^+ and NO_3^-` ions. `NaNO_(3(aq)) to NO_((aq))^(+)+NO_(3(aq))^-` Water dissociation to a small extent as `H_2O(I) hArr H_((aq))^(+)+OH_((aq))^-` Since `[H^+]=[OH^-]`,water is neutral `NO_3^-` ion is the conjugate base of the strong acid `HNO_3` and hence it has no tendency to react with `H^+` Similarity, `Na^+` is the conjugate acid of the strong base `NaOH` and it has no tendency to react with `OH^-` it means that there is no hydrolysis. In such cases `[H^+]=[OH^-] PH` is maintained and, THEREFORE, the solution is neutral . |
|
| 29. |
On Hydrolysis of phosphorus oxychloride which acid is formed ? |
|
Answer» `H_(3)PO_(3)` |
|
| 30. |
On hydrolysis of a "compound", two compounds, are obtained. One of which on treatment with sodium nitrite and hydrochloric acid gives a product which does not respond to iodoform test. The second one reduces Tollen's reagent and Fehling's solution The "Compound: is |
|
Answer» `CH_(3)CH_(2)CH_(2)NC` |
|
| 31. |
On hydrolysis, NCl_(3) gives NH_3 and HOCl while PCl_3 gives H_3PO_3 and HCl - explain. |
| Answer» Solution :The acidity of a MOLECULE DEPENDS on BOND polarity of central atom and hydrogen. GREATER the polarity, higher will be the acidity. The polarity of bond depends on the electronegativity of the central atom. Since, S is more ELECTRONEGATIVE then p, `H_2S` is more acidic than `PH_3` | |
| 32. |
On hydrolysis nitro ethane gives |
|
Answer» METHYL AMINE and HYDROXYL amine |
|
| 33. |
On hybridisation of one s and one p orbital we get |
|
Answer» THREE orbitals in plane |
|
| 34. |
On hydrolusis, all lipids yield |
|
Answer» Monocarboxylic acids |
|
| 35. |
On heating a salt with NaOH, smell of NH_3 is obtained, The salt contains: |
|
Answer» `NH_4^+` |
|
| 36. |
On heating ZnCl_2.2H_2O the compound obtained is: |
| Answer» Answer :B | |
| 37. |
On heating ZnCl_(2).2H_(2)O, the compounds obtained is |
|
Answer» `ZnCl_(2)` |
|
| 38. |
On heating with P_(2)O_(5) , acetic acid gives : |
|
Answer» ACETONE |
|
| 39. |
On heating with soda-lime, salicylic acid gives |
|
Answer» Phenol |
|
| 40. |
On heating with conc.H_2SO_4 sucrose gives: |
|
Answer» CO and `CO_2` |
|
| 41. |
On heating with concentrated NaOH solution in an inert atmosphere of CO_(2) , white phosphorus gives a gas. Which of the following statement is incorrect about the gas ? |
|
Answer» It is highly poisonous and has smell like ROTTEN fish. `P_(4)+ 3NaOH + 3H_(2)O to PH_(3) + 3NaH_(2)PO_(2)` |
|
| 42. |
On heating with concentrated NaOH solution in an inert atmosphere of CO_2, white phosphorus gives a gas. Which of the following statements is incorrect about the gas ? |
|
Answer» It is highly poisonous and has SMELL like rotten fish. |
|
| 43. |
On heatingwith concentratedNaOH solution in an inert atmosphere of CO_(2) white phorusgivesa gas . Which of the following statementsis incorrect about the gas ? |
|
Answer» It ismore basicthan `NH_(3)` `PH_(3)`is less basic than `NH_(3)` as the electron DENSITY is diffused over the LARGER phosphorus atom . |
|
| 44. |
On heating with concentrated NaOH solution in an inert atmosphere of CO_(2), white phosphorus gives a gas. Which of the following statement is incorrect about the gas ? |
|
Answer» It is highly poisonous and has smell like rotten fish |
|
| 45. |
On heating with concentration NaOH solution in an inert atmosphere of CO_2, white phosphorous gives a gas. Which of the following statement is incorrect about the gas ? |
|
Answer» It is highly poisonous and has SMELL like rotten fish. |
|
| 47. |
On heating with conc. HNO_3 proteins give yellow colour. This test is called |
|
Answer» OXIDISING test |
|
| 48. |
On heating with aqueous alkali,chloroform yields: |
|
Answer» HCHO |
|
| 49. |
On heating vapours of S_(8)(g) decomposes to S_(2)(g). Due to this, the van-der Waal's constant 'b' for the resulting gas |
|
Answer» INCREASES |
|