Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Number of dative bonds around Mg^(2+) ion in chlorophyll is |
|
Answer» 2 |
|
| 2. |
Number of crystal systems having only 2 types of bravais lattices = x, number of crystal system having at least two interfacial angles equal = y and number of crystal systems having all the three edge lengths equal = z. Then find the value of x xx y xx z. |
|
Answer» |
|
| 3. |
Number of dative bonds around Ag^+ ion in [Ag(NH_3)_2]^+ |
|
Answer» 2 |
|
| 4. |
Number of Cr-O sigma bonds in dichromate ion Cr_(2)O_(7)^(2-) is |
|
Answer» 6 |
|
| 5. |
Number of Cr-O sigma bonds in dichromate in Cr_(2)O_(7)^(2-) is |
|
Answer» |
|
| 6. |
Numbers of coulombs required to deposit 90 gm of aluminium, when the electrode fraction is,Al^3+3e^(-)toAl.....9.65times10^4,8.68times10^5,9.65times10^5,6.95. |
|
Answer» `9.65 XX 10^4` |
|
| 7. |
Number of corner/oxygen atoms shared per tetrahedron in 3-D silicate and pyroxene silicate are ........and .......respectively- |
|
Answer» 2,5,3 |
|
| 8. |
Number of complexes that are paramagnetic in nature with number of unpairedare : 1.[MnCl_4]^(2-)2.[Mn_2(CO)_10] 3.[V(CO)_6]^(-)4. 5.[Pt(NH_3)_4Cl_2]6.[Co(NH_3)_2(H_2O)_4]Cl_2 7.[Ni(CN)_4]^(2-)8.[Cu(H_2O)_4]^(2+) 9. K_3[Cr[CN)_5] |
|
Answer» |
|
| 9. |
Number of CO groups that can be attached to chromium is |
|
Answer» |
|
| 10. |
Number of following compounds in which central atom has "+6" oxidation state XeF_2, XeF_6, XeO_3, XeO_4, XeF_4,XeOF_4, XeO_2F_2 |
Answer» 
|
|
| 11. |
Number of chlorine atoms present gammaxine |
Answer» 
|
|
| 14. |
Number of chlorides satisfying secondary valency in CoCl_3 . 4NH_3 |
|
Answer» 1 |
|
| 15. |
Number of chiral isomers are: |
Answer» 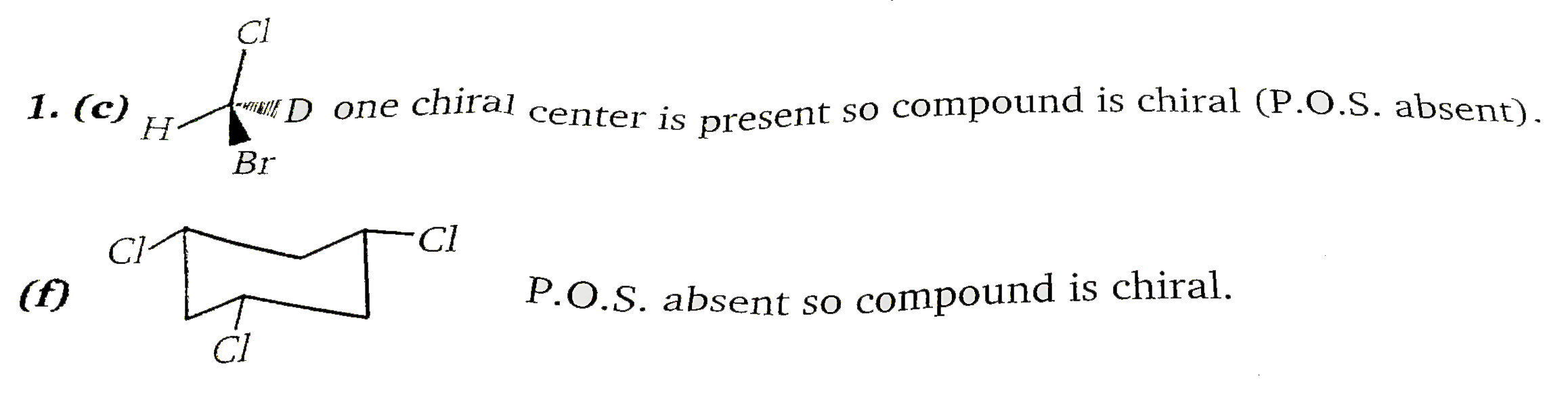 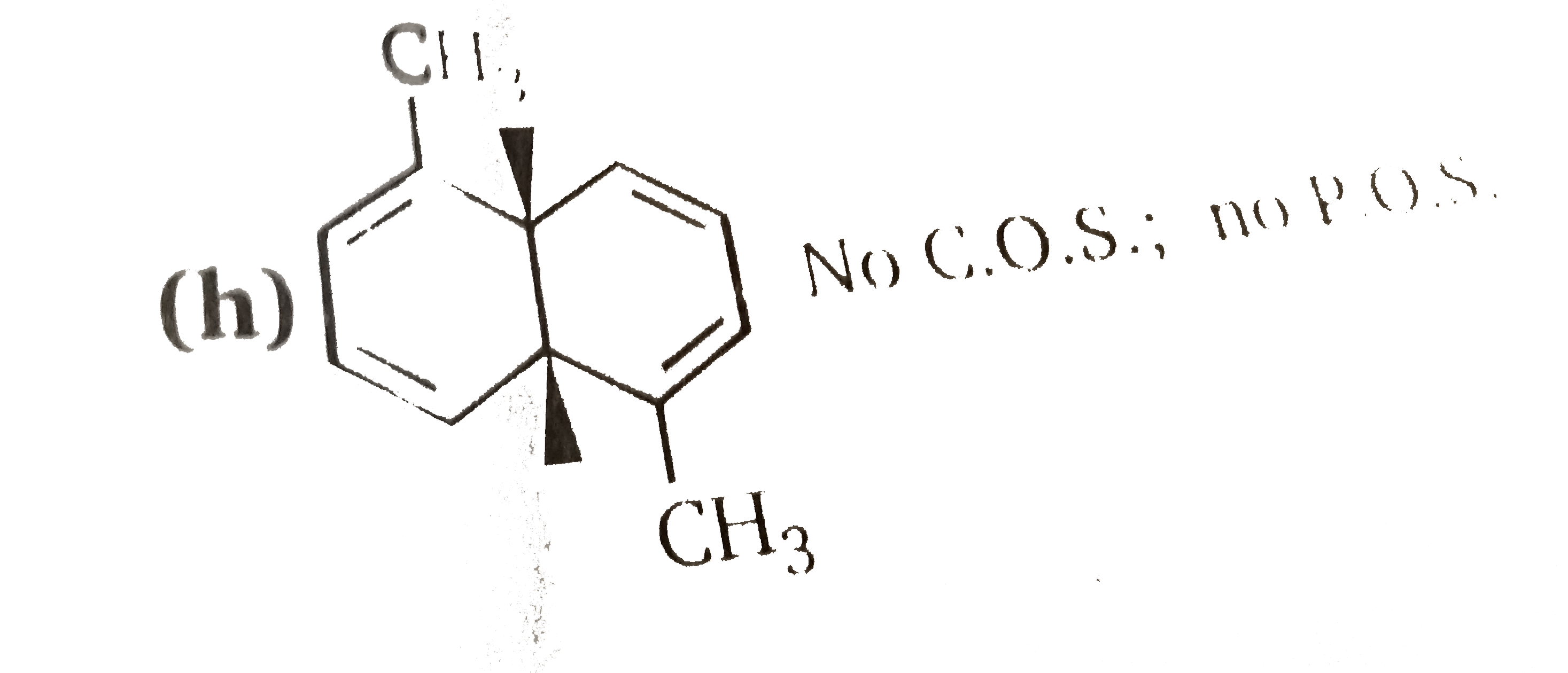
|
|
| 16. |
Number of chiral carbon atoms in the compound W, X, Y and Z respectively would be : |
|
Answer» |
|
| 17. |
Number of carbonyl compounds which form stable geminal diol in below is x then value of x/3 is |
Answer» SOLUTION :1.33 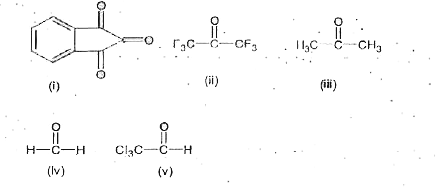
|
|
| 18. |
Number of chelate rings in [Fe(edta)]^(-)=a Numberof chelate rings in [Co (en)_2(NH_(3))Cl]^(2+)=b Number of chelate rings in [Ni(dmg)_2]=c Number of chelate rings in brown ring complex [Fe(H_2O)_5NO]SO_4=d Calculate the value of (a+b+c+d) is .... |
|
Answer» |
|
| 19. |
Number of chiral C-atom in glucose are …...... |
|
Answer» 4 |
|
| 20. |
Number of carbon atoms in parent carbon chain of iso-octane is : |
|
Answer» 6 |
|
| 21. |
Number of carbon atom present linearly in C_(3)O_(2) :- |
|
Answer» 2 [FOUR CARBON ATOMS PRESENT LINEARLY] |
|
| 22. |
Number of C = O groups in thymine and in X are equal. Now, X is |
|
Answer» CYTOSINE |
|
| 23. |
Number of bond pairs and lone pairs respectively that the central atom in xenon difluoride has |
|
Answer» 2,6 |
|
| 24. |
Number of beta particles emitted during the decay of ""_(92)^(235) U to ""_(82)^(207)Pb |
|
Answer» `92 = 82 + (2 xx 7) - b , b = 14 + 82 - 92 = 14- 10 = 4 beta` |
|
| 25. |
Number of B-O-B bond in borax |
|
Answer» `[B_(4)O_(5)(OH)_(4)]^(2-)`  Structure of `[B_(4)O_(5)(OH)_(4)]^(2-)` ion |
|
| 26. |
Number of atoms of oxygen present in 10.6 g Na_(2)CO_(3)will be |
|
Answer» `6.022 xx 10^(22)` `THEREFORE 10.6 g Na_(2)CO_(3) =0.1 mol` `=0.1 xx 6.02 xx 10^(23)` molecules `=3 xx 0.1 xx 6.02 xx 10^(23)` atoms of `O = 1.806 xx 10^(23)` |
|
| 27. |
Number of atoms of oxygen present in 10.6 g of Na_(2)CO_(3) will be |
|
Answer» `6.02xx10^(22)` `=46+12+48` `=106gmol^(-1)` `:.` 10.6 g of `Na_(2)CO_(3)-=0.1` MOL of `Na_(2)CO_(3)` `=0.3` mol of oxygen atoms `=0.3xx6.02xx10^(-23)` oxygen atom `=1.806xx10^(23)` oxygen atom |
|
| 28. |
Number of atoms in one molecule of sulphur |
|
Answer» `1.8` |
|
| 29. |
Number of atoms in 558.5 g Fe (molar mass Fe= 55.85 g mol^(-1)) |
|
Answer» twice than in 60 g carbon `=10 xx 6.023 xx 10^(23)`atoms. 60 g C = 5 moles `=5 xx 6.023 xx 10^(23)` atoms. |
|
| 30. |
Number of atoms in 588.6 g Fe (atomic mass of Fe = 55.86 g "mol"^(-1) ) is |
|
Answer» twice that in 60 g carbon ` = 10 xx 6.02 xx 10^23` No . OFATOMS in 558.6 g = twice the no. of atoms in 60 g C . |
|
| 31. |
Number of atoms in 4.25 g of NH_(3) is: |
|
Answer» `6.023xx10^(23)` |
|
| 33. |
Number of atoms 558.5g Fe (At. Wt. of Fe=55.85g "mol"^(-1)) is: |
|
Answer» twice that in 60 G CARBON |
|
| 34. |
Number of angular nodes for 4 d orbital is |
|
Answer» 4 |
|
| 35. |
Number of Amino acids present in Aspartame ? |
|
Answer» |
|
| 36. |
Number of amines among the following which give positive carbylamine test are C_(2)H_(5)NH_(2), (C_(2)H_(5))_(2)NH |
|
Answer» |
|
| 37. |
Number ofamine isomer of the molecular formula C_(4)H_(11)N is/are structural isomers |
|
Answer» `C-underset(NH_(2))overset(C)overset(|)(C)-C C-underset(C)underset(|)(C)-overset(H) overset(|)(N)-C C-C-underset(H)underset(|)(N)-C-C C-underset(C)underset(|)(N)-C-C underset(C)underset(|)(C)-C-overset(H)overset(|)(N)-C` |
|
| 38. |
Number of alpha- particles emitted per second by a radioactive element falls to 1//32 of its original value in 50 days. The half-life-period of this element is |
|
Answer» 5 days `N = N_(0) xx ((1)/(2))^(n) or (1)/(32) = 1 xx ((1)/(2))^(n)`, or `((1)/(2))^(5) = ((1)/(2))^(n) or n = 5` `T = t_(1//2) xx 2, or t_(1//2) = (50)/(5) = 10` days. |
|
| 39. |
Number of alpha - particles produced during the decay process of U - 235 to Pb - 207 |
|
Answer» |
|
| 40. |
Number of aldehydes isomers of butanone is |
Answer» 
|
|
| 41. |
Number of acylon product(s) formed during this reaction. Me-overset(O)overset(||)C-OEt+Et-overset(O)overset(||)C-OMeunderset(liq.NH_(3))overset(Na)to |
|
Answer» |
|
| 42. |
Number of acyclic structural isomers represented by molecular formula C_(4)H_(10)O is: |
|
Answer» 7 
|
|
| 45. |
Number of acidic hydrogen in 1-butyneis |
|
Answer» 1 |
|
| 47. |
Number of 2^(@)C nitrogens present in given compound are |
|
Answer» 2 |
|
| 48. |
Number average molecular mass overset-(M_n) of synthetic and weigth average mol. Mass overset-(M_w) of synthetic polymer are related as : |
|
Answer» `OVERSET-(M_n)ltoverset-(M_w)` |
|
| 49. |
Number average molecular mass, bar(Mn) and weight average molecular mass (barMw) of synthetic polymers are related as |
|
Answer» `bar(M_n) = (barM_(W))^(1//2)` For synthertic polymer, PDI > 1 `:. barM_w gt barM_n` |
|
| 50. |
Nueleicacids are bio polymers of …………. |
| Answer» SOLUTION :NUCLEOTIDES | |