Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Number of isotopes of oxygen are: |
|
Answer» One |
|
| 2. |
Number of isomers of molecular formula C_(2)H_(2)Br. Is : |
|
Answer» 1 |
|
| 3. |
Number of isomers possible for [Co(en)_(2)Cl(NO_(2))]Cl are |
| Answer» SOLUTION :Number of isomers for `[M(A-A)_(2)bc]^(npm)` of isomers POSSIBLE. | |
| 4. |
Number of isomers formed by the coordination compound with the formula MABCD is |
|
Answer» |
|
| 5. |
Number of ions which are identified by dil. HCl from the following. (i) SO_(4)^(2-) "" (ii) CO_(3)^(2-) "" (iii) SO_(3)^(2-) "" (iv) HCO_(3)^(-) (v) SO_(3)^(2-) "" (vi) NO_(3)^(-) "" (vii) CH_(3)CO O^(-) "" (viii) PO_(4)^(3-) |
|
Answer» |
|
| 6. |
Number of isomeric ethers with molecular formula C_5H_12O are |
|
Answer» 4 |
|
| 7. |
Number of intramolecular aldol condensation product is: |
|
Answer» 1 |
|
| 8. |
Number of incorrect statement are- (A) The pi bond between metal and carbonyl carbon reduces the bond order of C-O in carbon monoxide. (B) dz^(2) orbital of central metal atom/ion is used in dsp^(2) hybridisation. (C) CN^(-) is a pi_(-) acid Ligand. (D) All negative ligands are stronger than neutral ligends. |
|
Answer» |
|
| 9. |
Number of hydroxyl groups present in pyrosulphuric acid is: |
|
Answer» 3 |
|
| 10. |
number of hyperconjugable hydrogen atoms present in major product. |
Answer» 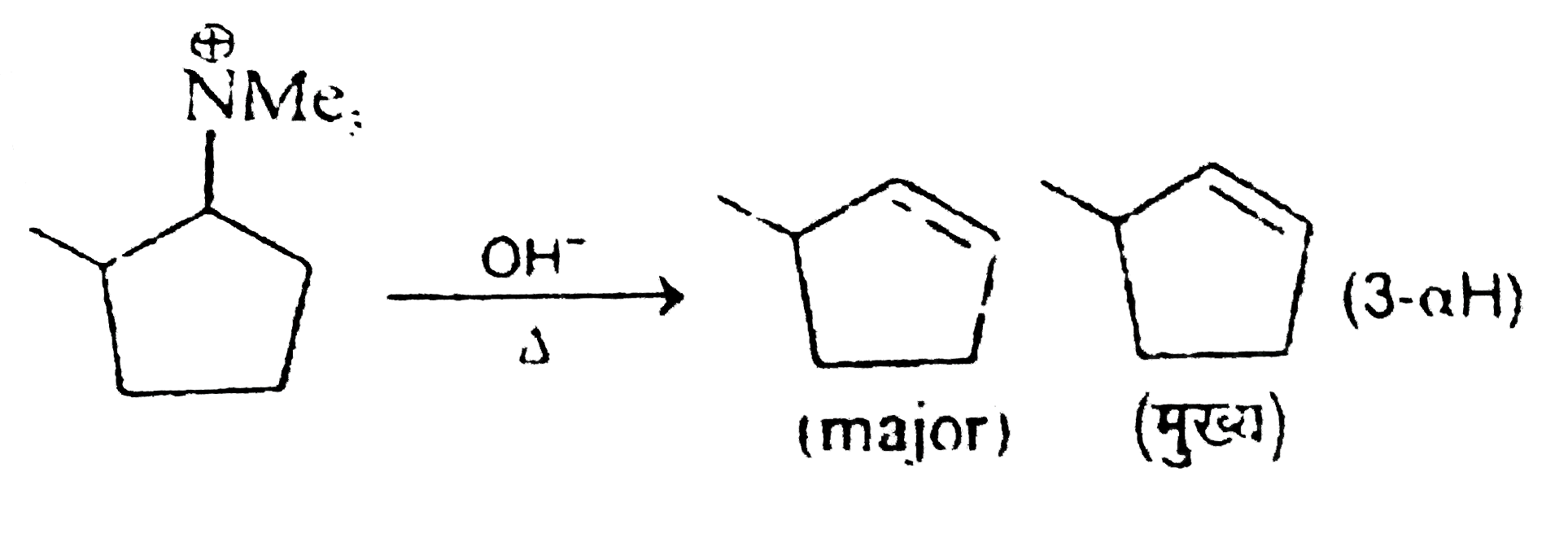
|
|
| 11. |
Number of hydrogen ions present in 10 millionth part of 1.33 cm^(3) of pure water at 25^(@)C is |
|
Answer» 6.023 million Now 1000 ml CONTAINS `10^(-7)` mole `H^(+)` 1 ml contains `10^(-7)/1000` mole `H^(+)` `1.33xx10^(-7)` ml - contains `1.33xx10^(-17)` 10 million `=10^(-7)` so, `10 "million"^(TH)` PART of `1.33 cm^(3)=1.33xx10^(-7)` ml so, no. of `H^(+)` ions `=1.33xx10^(-17)xxN_(A)`. |
|
| 12. |
Number of HlO_4molecular required to complete oxidation one mole of glucose is |
|
Answer» 4 |
|
| 13. |
Number of hydrogen ions present in 10 millionth part of 1.33 cm of pure water at 25°C is |
|
Answer» 6.023 MILLION Now, 1000 ML contains `10^(-7)` mole `H^(+)` ions 1 mL contains `10^(-7)/1000` mole `H^(+)` `1.33 xx 10^(-7)` mL contains `1.33 xx 10^(-17)` mole `H^(+)` 10 million `=10^(-7)` so, 10 million th part of 1.33 `cm^(3) = 1.33 xx 10^(-7)` mL So, no. of `H^(+)` ions `=1.33 xx 10^(-17) xx N_(A)` |
|
| 14. |
Number of halide ions among (F^(-),Cl^(-),Br^(-),I^(-)) which change their oxidation number on heating with MnO_(2)+conc.H_(2)CO_(4) |
|
Answer» |
|
| 15. |
Number of haloform form reaction is given by following compounds are x. |
|
Answer» Solution :1.5 `H_(3)C-OVERSET(OH)overset(|)CH-CH_(3)""H_(3)C-overset(O)overset(||)C-CH_(3)""H_(3)C-overset(O)overset(||)C-Cl` `H_(3)C-overset(O)overset(||)C-CH_(2)-overset(O)overset(||)C-CH_(3)""H_(3)C-overset(O)overset(||)C-OH""H_(3)C-overset(O)overset(||)C-NH_(2)` Then VALUE of `(x)/(2)` is 1.5 |
|
| 16. |
Number of H^(+) ions present in 250 ml of lemon juice of pH = 3 is |
|
Answer» `1.506 xx 10^(22)` |
|
| 17. |
Number of H bonds present between adenine and thymine in formation of Nucleic acid |
|
Answer» |
|
| 18. |
Number of geometrical isomer(s) of square planar complex [RhCl(PPh_(3))(H_(2)O)(CO)] is : |
|
Answer» 0 |
|
| 19. |
Number of fused six membered rings in sterol ? |
|
Answer» |
|
| 20. |
number of fractions obtained after fractional distillation of product mixture. |
| Answer» SOLUTION :N//A | |
| 21. |
Number of functional groups in the given compound is |
|
Answer»
|
|
| 22. |
Number of following substituents those are deactivating but ortho and para directing. |
|
Answer» |
|
| 23. |
Number of Faraday's required to generate one gram atom Cacl_2 is |
|
Answer» 1 |
|
| 24. |
Number of Faradays required to liberate 2 g of hydrogen is __________. |
|
Answer» 2 |
|
| 25. |
Number of faradays of electricity required to liberate 12 g of hydrogen is |
|
Answer» 1 |
|
| 26. |
Number of faradays of electricity required to liberate 12 g of hydrogen is : |
|
Answer» 1 |
|
| 27. |
Number of faraday required to reduce a mole of Fe^(3+) to Fe^(2+)are: |
|
Answer» 1 |
|
| 28. |
Number of Faraday required to liberate 8g of H_2 is : |
|
Answer» 8 |
|
| 29. |
Number of Faraday needed to deposit 0.1 mole of copper from Cu(II) sulphate solution are : |
|
Answer» `0.1` |
|
| 30. |
Number of electrons transferred in each case when KMnO_(4) acts as an oxidising agent and converts into MnO_(2), Mn^(2+), Mn(OH)_(3) and MnO_(2)^(-) are respectively |
| Answer» Answer :A | |
| 31. |
Number of electrons transferred in each case when KMnO_4 acts as an oxidising agent to give MnO_2, Mn^(2+), Mn_2(O)_3 and MnO_4^(2-) respectively are : |
|
Answer» 3, 5, 4 and 1 |
|
| 32. |
Number of electrons liberated on cathode by passing 1 ampere current for 60 second during electrolysis is . . . .(Charge on electrone =1.60xx10^(-19)C) |
|
Answer» `3.75xx10^(20)` `Ixxt=n*e^(-)` `n=(1xx60)/(1.6xx10^(-19))=3.75xx10^(20)e^(-)` |
|
| 33. |
number of electrons present in M shell of an element with atomic number 26 in its M^(3+) state will be |
|
Answer» zero |
|
| 34. |
Number of electrons lost during oxidation of 0.355 g of Cl^(-) are : |
|
Answer» `0.01` |
|
| 35. |
Number of electrons present in the outermost orbit of Fe atom is: |
|
Answer» 3 |
|
| 36. |
Number of electronsinvolved in the electrodepositedof 63.5 g of Cu from a solution of CuSO_(4) is |
|
Answer» `6.022 XX 10^(23)` |
|
| 37. |
Number of electrons in the nucleus of an element of atomic number 14 is |
|
Answer» 14 |
|
| 38. |
Number of electrons in outer most shell of Ce (Z = 58) |
|
Answer» |
|
| 39. |
Number of electrons in 3.6mg of NH_(4)^(+) are: |
|
Answer» `1.2XX10^(21)` `=(3.6xx10^(-3))/(18)xx6.023xx10^(23)=1.2xx10^(20)` `therefore` Number of electrons in 3.6mg `NH_(4)^(+)=1.2xx10^(20)xx10` `1.2xx10^(21)` |
|
| 40. |
Number of electron in an atom having n=4 , m= 1 and m_(s)=-1//2 are |
|
Answer» 16 HALF of these i.e 16 have `m_(s)=-1//2` |
|
| 41. |
Number of electrons gained by Pd in [PdCl_4]^(-2): |
|
Answer» 4 |
|
| 42. |
Number of electrons involved in the reduction of Cr_2O_7^(2-) ion in acidic solution to Cr^(3+) is: |
|
Answer» 3 |
|
| 43. |
Number of dipeptide which can be formed by : Glycine, Alanine, Leucine, Phenylalanine. |
|
Answer» |
|
| 44. |
Number of dichloro derivatives of tetramethylbutane is |
Answer» 
|
|
| 45. |
Number of diastereomers possible for |
|
Answer» 1 |
|
| 46. |
Numberofdi - substitiutedisomers of theproduct of thereaction ,CH_(3) CH_(2) CH_(3) +Br_(2)wouldbe : |
|
Answer» 2 |
|
| 47. |
Number of diasterwomer of given compound : |
|
Answer» 2 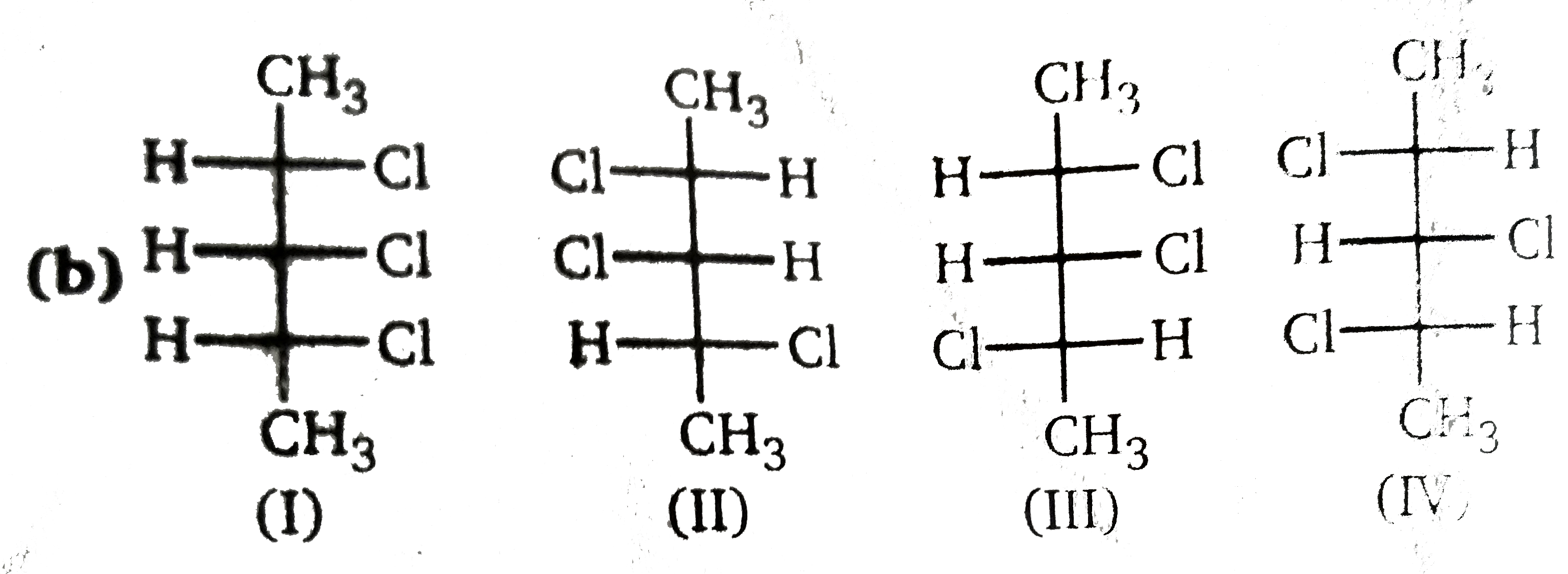 3 DIASTEREOMERS. 3 DIASTEREOMERS.
|
|
| 48. |
Number of d'electrons present in M shell of Ag+ ion? |
|
Answer» 10 |
|
| 49. |
Number of dative bonds (including sigma & pi) in P_4O_10 molecule is |
|
Answer» <P>  each P = O consists of twodative BONDS `4 - sigma ` DATIVE bonds and `4-pi` dative bonds so total 8 - dative bonds |
|
| 50. |
Number of dative bonds in the complex CoCl_(3).5NH_(3) is |
|
Answer» 5 |
|