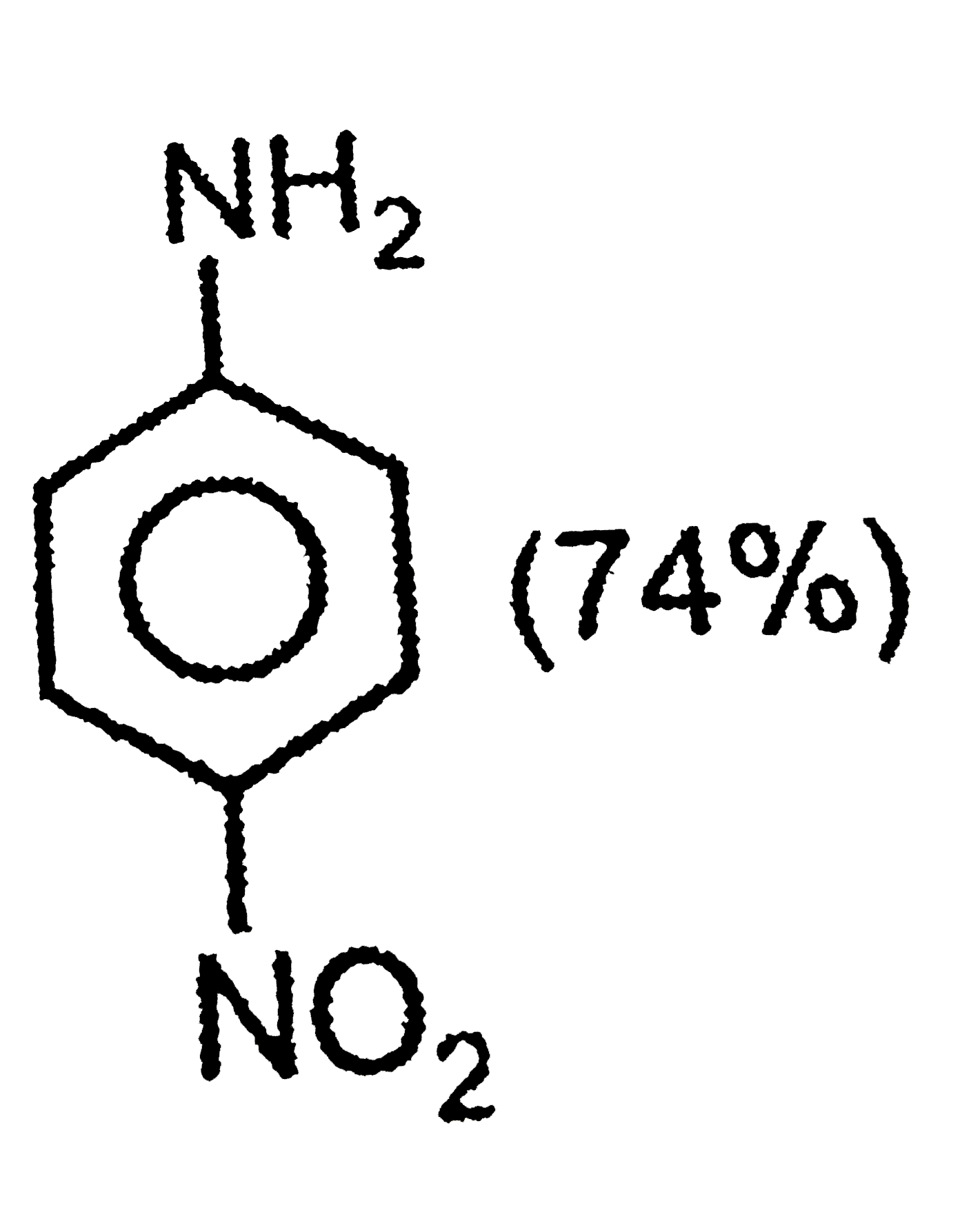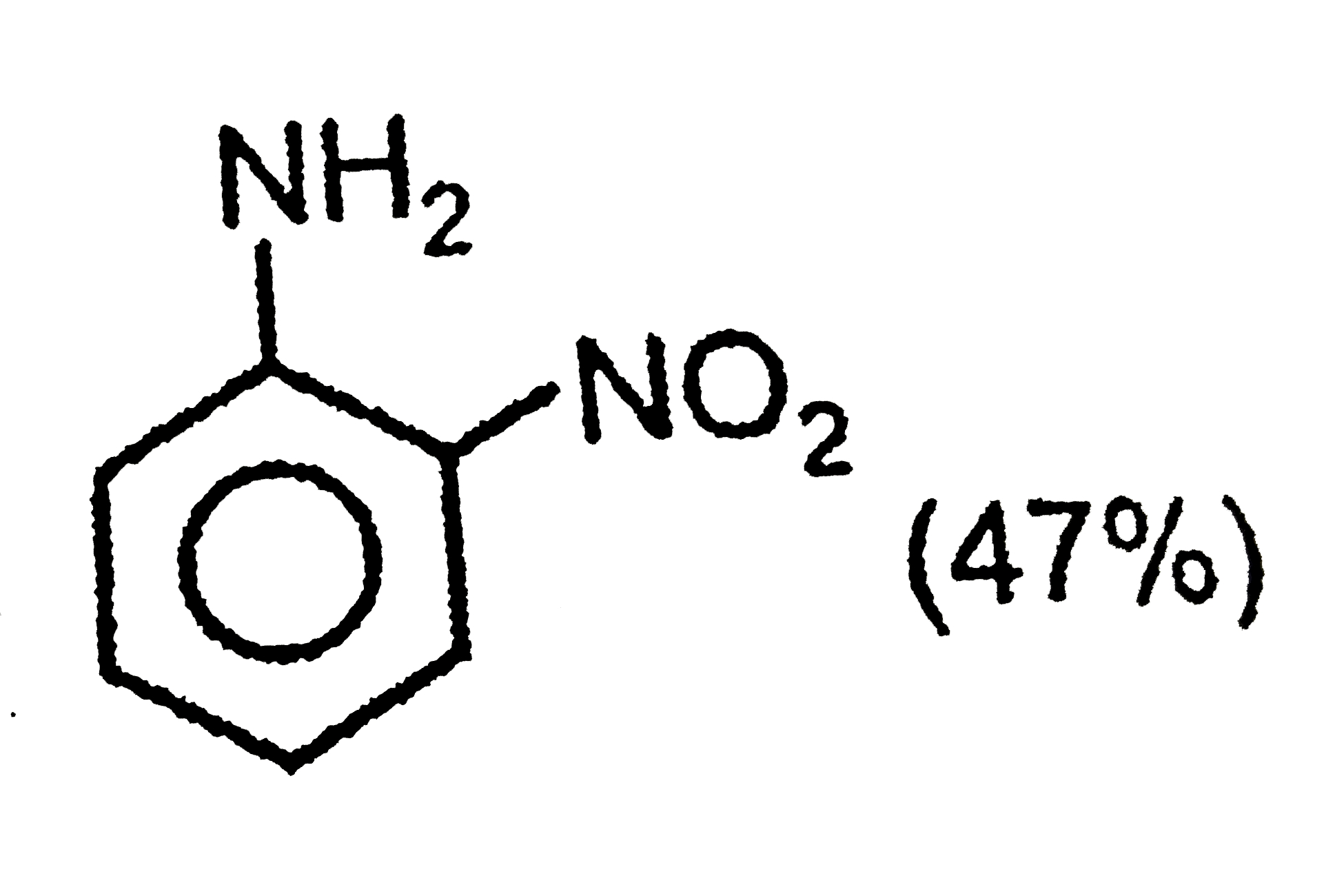Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct order of acid strengths of benzoic acid (X), peroxybenzoic acid (Y) and p-nitrobenzoic acid (Z) is |
|
Answer» `Y GT Z gt X`  Hence, acidic strength ORDER is `Z gt X gt Y`. |
|
| 2. |
The correct order of acidic nature of oxides is in the order |
|
Answer» `NO lt N_(2)O lt N_(2)O_(3) lt NO_(2) lt N_(2)O_(5)` `UNDERSET("Neutral")(ubrace(N_(2)O" "NO))" "underset("increases")underset("Acidic character")(ubrace(N_(2)O_(3),NO_(2),N_(2)O_(5)))` |
|
| 3. |
The correct order of acid strength of the following substituted phenols in water at 28^(@)Cis |
|
Answer» p-nitrophenol `LT` p-fluorophenol `lt `p-chlorophenol |
|
| 4. |
The correct order of acid strength of the followingsubstituted phenol in water at 28^(@)Cis : |
|
Answer» p-nitrophenol ` LT ` p-fluorophenol ` lt ` p-chlorophenol |
|
| 5. |
The correct order of acid strengthis |
|
Answer» `HClO_(4) LT HClO_(3) lt HClO_(2) lt HClO` |
|
| 6. |
The correct order of acid strength is |
|
Answer» `HCIO_4ltHCIO_3ltHCIO_2ltHCIO` |
|
| 7. |
The correct order of acidic strength is |
|
Answer» `CH_3COOHgtCH_2ClCOOHgtCHCl_2COOH` |
|
| 8. |
The correct order ionic radii of Y^(3) ,La^(3+), En^(3+) and Lu^(3+) is |
|
Answer» `Y^(3+) LT La^(3+) lt Eu^(3+) lt Lu^(3+)` `Y(Z=39)` is first member of 4d-series so that `Y^(3+)` is smallest in SIZE. Thus, order of ionic radii is `La^(3+) GT Eu^(3_) gt Lu^(3+) gt Y^(3+)` |
|
| 9. |
The correct order of 2^(nd) ionization potential of carbon, nitrogen , oxygen and fluorine is |
|
Answer» CgtNgtOgtF `O(8) 1s^(2)2s^(2)2p_(x)^(2) 2p_(y)^(1)2p_(z)^(1) "" F(9)1s^(2)2s^(2)2p_(x)^(2)2p_(y)^(2)2p_(z)^(1)` O has highest `2^(nd)` ionization potential i.e., ionization ENERGY because after LOSING one electron, it acquires stable half filled p-electronic configuration. F is smaller than N, therefore, it has higher second ionization potential. C has least due to BIGGER size. |
|
| 10. |
The correct order of acedic strength of the following compounds is |
|
Answer» `HClOgtHClO_(2)gtHClO_(3)gtHClO_(4)` |
|
| 11. |
The correct order of 1st ionisation enthalpy among the following elements Be, B,C,N and O is |
|
Answer» `BltBeltCltOltN` |
|
| 12. |
The correct order of the O-O bond length in O_(2), H_(2)O_(2) and O_(3)is : |
|
Answer» `O_(2)ltH+_(2)O_(2)lt_(3)` |
|
| 13. |
The correct order in which the first ionisation potential increases is |
|
Answer» K, Be, Na `._(4)Be-1s^(2)2s^(2)` `._(11)Na-1s^(2)2s^(2)2p^(6)3s^(1)` `._(19)K-1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(1)` The FIRST ionisation energy of Be is maximum because ELECTRON is to be drawn from stable (fully FILLED) orbital. The `I^(st)` ionisation energy of Na is greater than K because size of the K is bigger than Na which facilitates easy REMOVAL of electron from its outermost shell. So, the sequence is `KltNaltBe`. |
|
| 14. |
The correct order in which the first ionization potential increases is |
|
Answer» K , Be , Na `._(4)Be-1s^(2)2s^(2)` `._(11)Na-1s^(2)2s^(2)2p^(6)3s^(1)` `._(19)K-1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)4s^(1)` The first IONIZATION energy of Be is maximum because electron is to be drawn from stable (fully filled) orbital. The `1^(st)`ionization energy of Na is greater than K because size of the K is bigger than Na which facilitates easy removal of electron from its outermost shell. So, the sequence is `KltNaltBe`. |
|
| 15. |
The correct order for the CFSE (numerical value) for the following complexes is : {:("Complex", P , Q , R , S),("Formula" ,[Co(CN)_(6)]^(3-) , [Co(NH_(3))_(6)]^(3+) , [CoF_(6)]^(3+) , [Co(H_(2)O)_(6)]^(3+)):} |
|
Answer» `PgtQgtRgtS` |
|
| 16. |
The correct order for the wavelength of absorption in the 27. visible region is : |
|
Answer» `[Ni(NO_(2))_(6)]^(4-) lt [Ni(NH_(3))_(6)]^(2+) lt [Ni(H_(2)O)_(6)]^(2+)` The value of `Delta_(0)` for ligands varies as follows `H_(2)O lt NH_(3) lt NO_(2)^(-)` so, the wavelength absorbed will vary in REVERSE order `NO_(2)^(-) lt NH_(3) lt H_(2)O`. |
|
| 17. |
The correct option(s) to distinguish nitrate salts of Mn^(2+)" and Cu^(2+) taken separately is (are) |
|
Answer» `MN^(2+)` show the characteristic green colour in the flame test (b) In weakly acidic medium, only `Cu^(2+)` ions form black precipitate of CuS. (d) `E_(Cu^(2+)//Cu)^(@)(+0.34V)` is higher than that of `E_(Mn^(2+)//Mn)^(@)(-1.18V)` The other two options are not correct. (a) `Cu^(2+)" and not "Mn^(2+)` shows characteristic green colour in the flame test. (c) Both `Cu^(2+)" and "Mn^(2+)` show the formation of precipitate (CuS-Black) & MnS-Brown) by passing `H_(2)S` in faintly basic medium. |
|
| 18. |
The correct order for E_(2) reaction with alc. KOH will be :- |
|
Answer» `igtiigtiii` |
|
| 19. |
The correct order for decreasing acidic strength of oxyacids of group 15 is |
|
Answer» `HNO_(3) gt H_(3)SbO_(4) gt H_(3)AsO_(4) gt H_(3)PO_(4)` |
|
| 20. |
The correct option(s) to distinguish nitrate salts of Mn^(2+) and Cu^(2+) taken separately is (are) : - |
|
Answer» ` Mn^(2+)` SHOWS the characteristic green colour in the FLAME test |
|
| 21. |
The correct order for homolytic bond dissociation energies (Delta H in kcal/mol) for CH_(4)(A), C_(2)H_(6)(B) and CH_(3)Br(C) is |
|
Answer» `C GT B gt A` |
|
| 22. |
The correct option regarding size of orbitals is: |
|
Answer» `3pgt4pgt5p` |
|
| 23. |
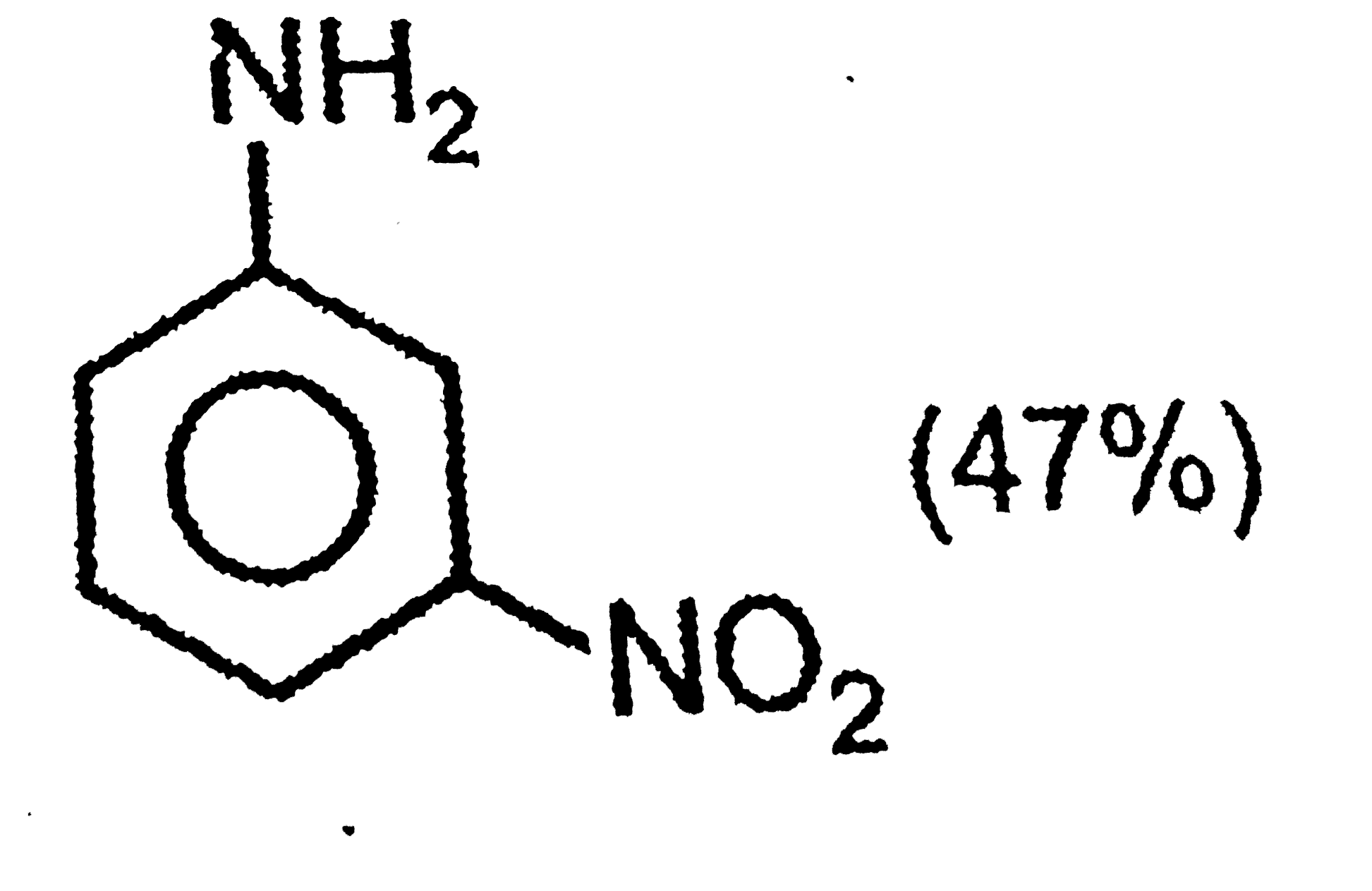
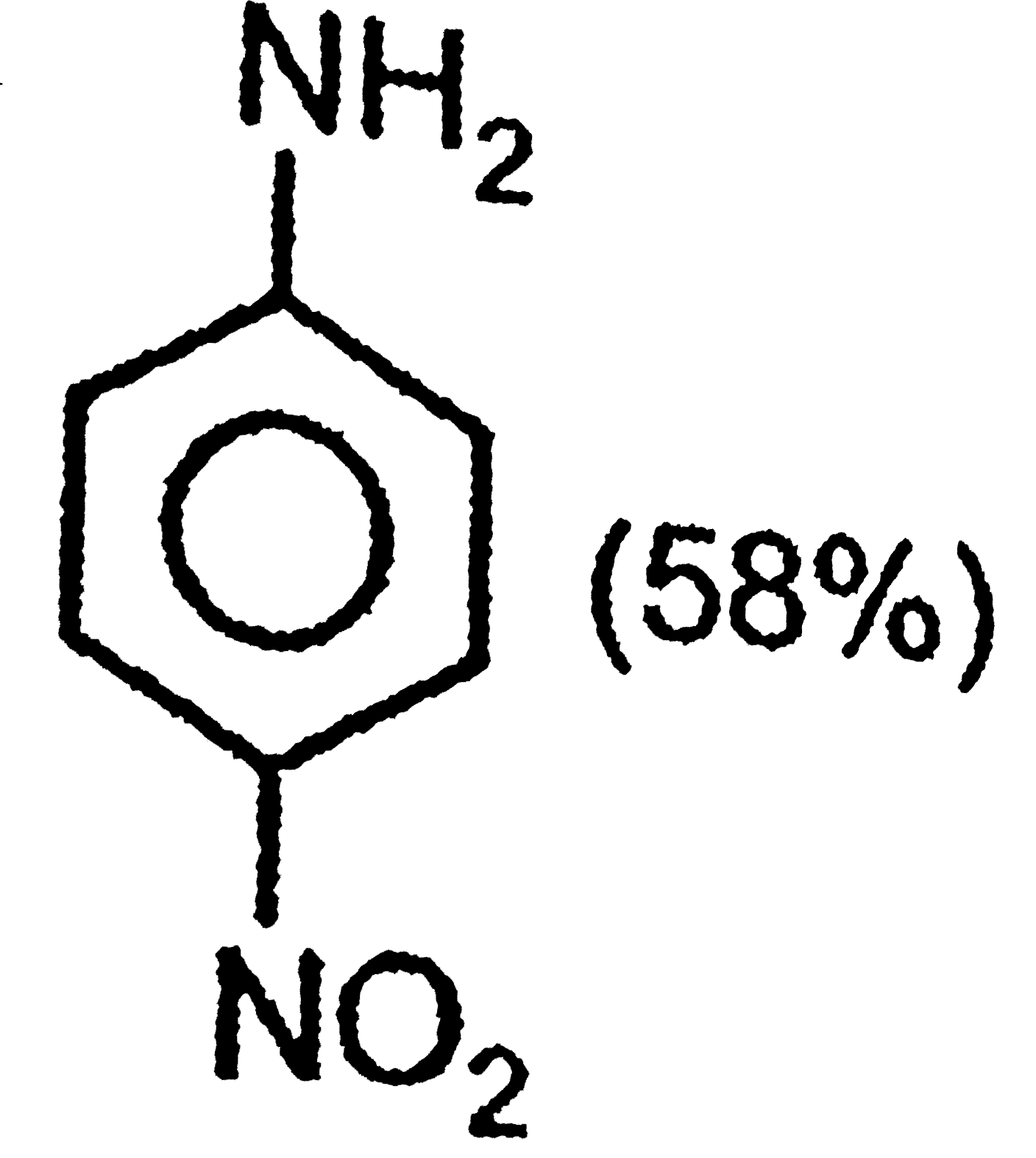
The correct option regarding the percentage of product obtained during nitration of aniline at 288K is |
|
Answer»
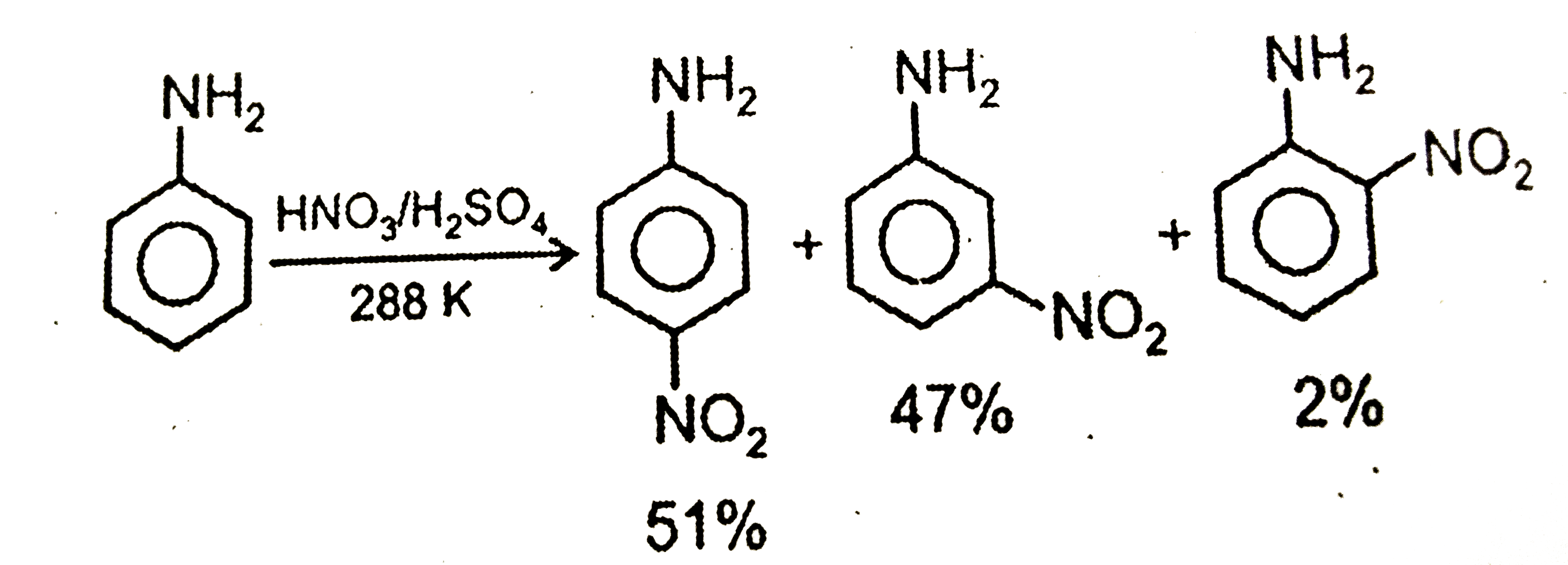
|
|
| 24. |
The correct option among the following is: |
|
Answer» Colloidal particles in lyophobic sols can be PRECIPITATED by electrophoresis. |
|
| 25. |
The correct number of significant figures in the answer of 0.00383 -0.00303 is : |
|
Answer» TWO |
|
| 26. |
Thename of the given compound [Cu(NH_3)_4](NO_3)_2 , according to IUPAC system is: |
|
Answer» CUPRAMMONIUM nitrate |
|
| 27. |
The correct name of the compound [Cu(NH_3)_4](NO_3)_2 according to IUPAC system is: |
|
Answer» CUPRAMMONIUM nitrate |
|
| 28. |
The correct name of 'sucrose' is : |
|
Answer» `ALPHA`-D-glucopyranosyl-`BETA`-D-fructofuranoside |
|
| 29. |
The correct name of |Pt(NH_(3))_(4)Cl_(2)||PtCl_(4)| is |
|
Answer» Tetraammine DICHLORO platinum (iv) TETRACHLORO platinate (II) |
|
| 30. |
The correct name of given compound is:- |
|
Answer» Isopropyl BENZENE  2-Phenyl propane OR isopropyl benzene & its COMMON NAME is cumene. |
|
| 31. |
The correct name of is : |
|
Answer» Tri-u-carbonyl BIS (tricarbonyl) iron(0) |
|
| 32. |
The correct name and product of given reaction are respectively (AAK_MCP_37_NEET_CHE_E37_020_Q01) |
|
Answer» KOLBE's REACTION, |
|
| 33. |
The correct name for the complex ion [CoCl(ONO)(en)_(2)]^(+) is: |
|
Answer» chlorobis(ETHYLENEDIAMINE)nitrito-O-COBALTATE (III) ion |
|
| 34. |
The correct match which is responsible for colour |
|
Answer» `{:(A,B,C,D),(4,1,2,5):}` |
|
| 35. |
The correct match is |
|
Answer» `{:(A,B,C,D),(IV,I,III,II):}` |
|
| 36. |
The correct match is |
|
Answer» a - II, B - i, c - iii, d - IV |
|
| 37. |
The correct match is |
|
Answer» `{:(A,B,C,D),(3,4,1,2):}` |
|
| 38. |
The correct match is |
|
Answer» 1-C, 2-A, 3-E, 4-B |
|
| 39. |
Thecorrect match is |
|
Answer» |
|
| 41. |
The correct match is |
|
Answer» `{:(i,II,III,iv),(D,A,B,C):}` |
|
| 42. |
The correct match is |
|
Answer» |
|
| 43. |
The correct match is |
|
Answer» A- II, B-III, C-i |
|
| 44. |
The correct match is |
|
Answer» 1-C,2-E,3-A,4-B |
|
| 45. |
The correct match is |
|
Answer» `{:(A,B,C,D),(1,4,3,2):}` |
|
| 46. |
The correct match is |
|
Answer» i-D,ii-A,iii-B,iv-C |
|
| 47. |
The correct match is |
|
Answer» A-5, B-1, C-2, D-3 |
|
| 49. |
The correct match is |
|
Answer» `{:(A,B,C,D),(1,2,3,5):}` |
|