Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The additive used to make PVC, a flexible plastic is called |
|
Answer» ANTIOXIDANT |
|
| 2. |
The addition product is obtained by reaction between alkene and diborane. This reaction is called .. |
|
Answer» ACID catalysed hydration |
|
| 3. |
'The addition of unsymmertial reagent to unsymmetrical alknes occurs in such a way that the negative part of the addendum goes to that carbon atom of the double bond which carreis lesser number of hydrogen atoms'' is called by |
|
Answer» Saytzeff rule I `HX hArrH^(+)+,X^(-)`  `)
|
|
| 4. |
The addition of the salt CH_(3)COONa to a solution of CH_(3)COOH result in a decrease of the initial |
|
Answer» `[OH^(-)]` |
|
| 5. |
The addition of pure solid sodium carbonate to pure water causes……… |
|
Answer» an increase in HYDRONIUM ion concentration |
|
| 6. |
The addition of solid sodium carbonate to pure water causes : |
|
Answer» INCREASE in `H^(+)` ion concentration |
|
| 7. |
The addition of pure solid sodium carbonate to pure water causes ………….. . |
|
Answer» an increase in HYDRONIUM ion concentration |
|
| 8. |
The addition of one of the reactants in a reaction at equilibrium |
|
Answer» ALWAYS SHIFTS the equilibrium towards product at constant volume but not always at constant PRESSURE |
|
| 9. |
The addition of metal like Cr, Mn W and Ni to ordinary steel makes it |
|
Answer» More useful |
|
| 10. |
The addition of K_(2)CO_(3) (aq) to the following solution is expected to produce a precipitate in every case but that one which does not produce precipitate is: |
|
Answer» `BaCl_(2) (aq)` |
|
| 11. |
The addition of HI in the presence of peroxide does not show anti-Markovnikov behavior because |
|
Answer» the HI band is too strong not to be broken homolytically |
|
| 12. |
The addition of HOCl with unsymmetrical alkenes produces the product X as R-CH =CH_2 + HOClrarrX. Therefore ,the product X is : |
|
Answer» `R - CH(OH) - CH_2Cl` |
|
| 13. |
The addition of HCN to carbonyl compounds is an example of .............. reaction. |
|
Answer» NUCLEOPHILIC substitution |
|
| 14. |
The addition of HCl will not suppress the ionization of |
|
Answer» Acetic ACID |
|
| 15. |
The addition of HCN to carbonyl compounds is an example of |
|
Answer» nucleophilic ADDITION  NUCLEOPHILE attacks at the positive C CENTRE of carbonyl group hence the addition is nucleophilic addition. |
|
| 16. |
The addition of HCl in propene proceeds via |
|
Answer» `CL^(+)` in first stpe `CH_(3)-CH=CH_(2)+Cl^(+) to CH_(3)- overset(o+)(CH)-CH_(2)Cl` |
|
| 17. |
The addition of HCl in presence of peroxide does not follow anti-Markovnikov's rule because |
|
Answer» HCL BOND is too strong to be BROKEN homolytically |
|
| 18. |
The addition of HCl does not suppresses the ionisation of : |
|
Answer» ACETIC ACID |
|
| 19. |
The addition of HBr to 2-pentene gives |
|
Answer» 2-bromopentane only |
|
| 20. |
The addition of HBr on butene-2 is an example of: |
|
Answer» NUCLEOPHILIC addition |
|
| 21. |
The addition of HBr on butene-2 in presence of peroxide follow the: |
|
Answer» ELECTROPHILIC addition |
|
| 22. |
The addition of four amine groups to a metal ion (X^(2+)) shows a stability constants of 2 * 10^4, 1.5 * 10^3, 1.2 * 10^2 and 1.4 * 10^1 respectively. Then, the overall complex dissociation equlolibrium constant for [X(NH_3)_4]^(2+) ion is |
|
Answer» 5.04 * 10^(-10)` |
|
| 23. |
The addition of ferric hydroxide solution to arseneous sulphide solution results in the precipitation ofboth. |
| Answer» Solution :This is possible only in CASE equimolar solutions (solutions with same number of MOLES) are mixed. Since the ferric hydroxide sol carries positive CHARGE and arsenic oxide sol is negatively charged on MIXING they will get their charge neutralized and will be COAGULATED. | |
| 24. |
The addition of excess of aqueous HNO_(3) to a solution containing [Cu(NH_(3))_(4)]^(2+) produces |
| Answer» Answer :B | |
| 25. |
The addition of bromine to cyclopentene provides evidence for bromonium ion intermediate in bromine addition. It follows ionic mechanism. When cyclopentene reacts with bromine in presence of. "CC"l_4 anti addition occurs and gives trans isomer. So, addition of halogen to an alkene is a stereospecific reaction. Cis alkene on addition with halogen gives racemic products and trans alkene on addition gives meso mixture. Trane 2-butene on reacting with Br_2 in CCl_4 forms mainly |
|
Answer» `+-2,3-`DIBROMOBUTANE |
|
| 26. |
The addition of catalyst during a chemical reaction alters which of the following quantities? |
|
Answer» Enthalpy |
|
| 27. |
The addition of bromine to cyclopentene provides evidence for bromonium ion immediate in bromine addition. It follows ionic mechanism. When cyclopentene reacts with bromine in presence of CCl_4 anti addition occurs and gives trans isomer so addition of halogen to an alkene is a stereospecific reaction. cis alkene on addition with halogen gives racemic mixture and trans álkene on addition may give meso products. trans-2-butene on reaction with Br_2 in C Cl_4 forms mainly |
|
Answer» `+-2,3-` DIBROMOBUTANE |
|
| 28. |
The addition of Br_(2) to Z-2-butane gives |
|
Answer» (R,R)-2,3-dibromo BUTANE only |
|
| 29. |
The addition of Br_2 to (E)-but-2-ene gives |
|
Answer» (R,R)-2-3-DIBROMOBUTANE 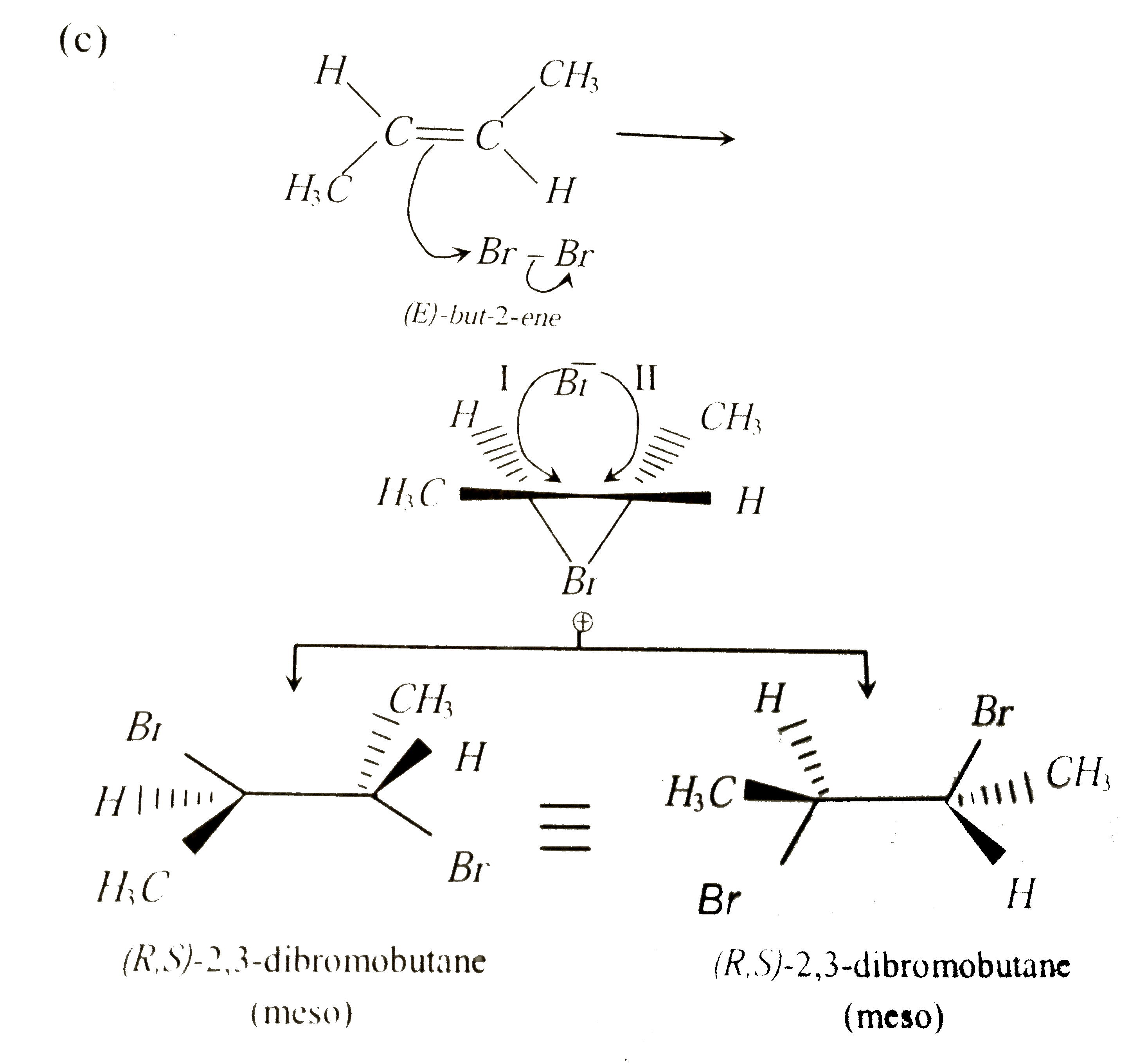
|
|
| 30. |
The addition of Br_2 to ( E)-but-2-ene gives |
|
Answer» (R,R)-2-3-DIBROMOBUTANE |
|
| 31. |
The addition of a polar solvent to a solenoid electrolyte results in |
|
Answer» Polarization |
|
| 32. |
The addition of aldehyde group to aromic molecules of phenol when it react with sodium hydroxide and chloroform is known as ...... Reaction. |
|
Answer» Kolbe-Schmitt |
|
| 33. |
The additionof ammonium chlorode to a solutioncontainingferric andmagnessiumions is easentialfor solectiveprecipitate offerric hydroxide by aqueous ammonium |
|
Answer» |
|
| 34. |
The addition of a polar solvent to a solid electrolyte |
|
Answer» POLARIZATION |
|
| 35. |
The addition of acidic K_(2)Cr_(2) O_(7) to NaCl produces a colour |
| Answer» ANSWER :C | |
| 36. |
The addition of a drop of copper sulphate solution to an alkaline solution of biuret gives: |
| Answer» Answer :B | |
| 37. |
The addition of 3g of a substance to 100g C CI_(4)(M = 154 g mol^(-1)) raises the boiling point of C CI_(4) by 0.60^(@)C if K_(b)(C CI_(4)) is 5K mol^(-1) kg, calculate (a) the freezing point depression (b) the relative lowering of vapour pressure (c) the osmotic pressure at 298K and (d) the molar mass of the substance. Given: K_(f) (C CI_(4)) = 31.8 K kg mol^(-1) and rho (solution) = 1.64 g cm^(-3) |
|
Answer» `DeltaT_(f) = (31.8)/(5.03) XX 0.6 = 3.79^(@)C` (b) `m = (DeltaT_(b))/(K_(b)) = (0.6)/(5.03) = (x xx1000)/((1-x)154)` `rArr 1-x = 54.44 x` `rArr 55.44 x = 1` `x = (DeltaP)/(P^(@)) = (1)/(5.44) = 0.018` (c) `(1)/(55.44) = ((3)/(M_(1)))/((3)/(M_(1))+(100)/(154))` `rArr (100)/(154) = (166.32)/(M_(1)) M_(1) = 231.5` `M = (0.6)/(5.03)` `(M xx 1000)/(1000 xx 1.64 -M xx 251.5)` `1640 = 8634.83M` `M = 0.19` `p = n//V RT = 0.19 xx 0.0821 xx 298` `=4.65 atm` |
|
| 38. |
The addition of a catalyst during a chemical reaction alters which of the following quantities ? |
|
Answer» Activation energy |
|
| 39. |
The addition of 41.2mg of an unkown alcohol ROH to CH_(3)MgI releases 1.56mL of a gas at STP. The molar mass of the gas is |
|
Answer» 32 g `mol^(-1)` 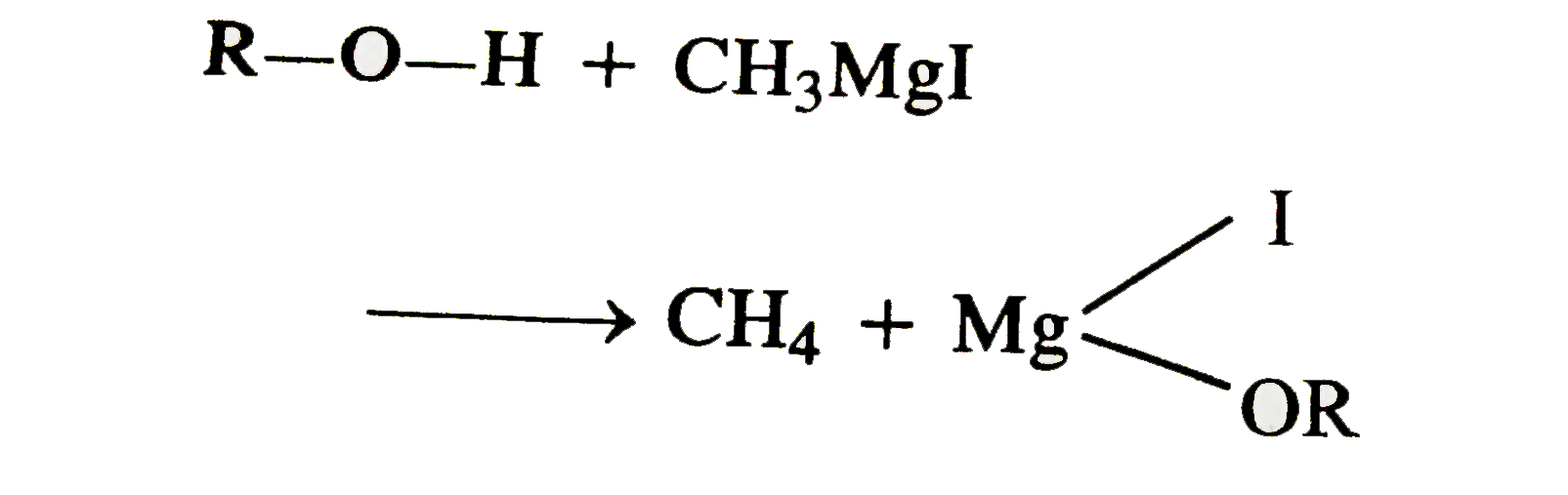
|
|
| 40. |
The addition of 3 gms of a substance to 100 gms C Cl_(4) (M.w. = 154 gm/mole) raises the boiling point of C Cl_(4) by 0.60^(@)C. If K_(b) of C Cl_(4) is 5.03 K Kg "mole"^(-1) then find out the relative lowering of vapour pressure. |
|
Answer» `0.0181` Mol. Wt. of sub `= M_(2)=?` SOLVENT `= C Cl_(4)=100 gms = W_(1)` Molal of solvent `C Cl_(4)=153" gm mole"^(-1)=M_(1)` Increase of B.P. `= Delta T_(b)=0.6^(@)C` `K_(b)` of solvent `C Cl_(4)=5.03" K kg mole"^(-1)` `M_(2)=(K_(b)xx W_(2)xx 100)/(Delta T_(b)xx W_(1))` `therefore M_(2) = (5.03xx3xx1000)/(0.6xx100)` `= 251.5"gm mol"^(-1)` Relative depression of vapour pressure `= (p^(@)-p)/(p^(@))=X_(2)` `= (n_(2))/(n_(1)+n_(2))` Where `n_(2)=` mole of solvent `= (W_(2))/(M_(2))=(3)/(251.5)` = 0.0119 mole substance `n_(1)=` mole of solvent `= (W_(1))/(M_(1))=(100)/(154)` = 0.6494 mole solvent `C Cl_(4)` TOTAL mole `= 0.6494+0.0119=0.6613` `X_(2)=(0.0119)/(0.6613)` `=0.01799 ~~0.0181` |
|
| 41. |
The addition of 3 gm of substance to 100gm C Cl_(4) (M=154 gm "mol^(-1)) raises the boiling point of C Cl_(4) by 0.6^(@)C. K_(b)(C Cl_(4)) is5.03d kg "mol"^(-1)K, K_(f)(CCl_(4)) is31.8 kg "mol"^(-1)K and dencity of solution is 1.64 gm/ml . The molar mass of the substance is : |
|
Answer» `37.9` |
|
| 42. |
The addition of 3 gm of substance to 100gm C Cl_(4) (M=154 gm "mol^(-1)) raises the boiling point of C Cl_(4) by 0.6^(@)C. K_(b)(C Cl_(4)) is5.03d kg "mol"^(-1)K, K_(f)(CCl_(4)) is31.8 kg "mol"^(-1)K and dencity of solution is 1.64 gm/ml . Calculate the depression in freezing point |
|
Answer» `3.79^(@)C` |
|
| 43. |
The additio of HBr to but-1-ene gives a mixture of products (I),(II) and (III). . (III). CH_(3)-CH_(2)-CH_(2)-CH_(2)-Br. Mixture consists of |
|
Answer» (I) and (II) as MAJOR and (III) as MINOR PRODUCTS 
|
|
| 44. |
The addition of 3 gm of substance to 100gm C Cl_(4) (M=154 gm "mol^(-1)) raises the boiling point of C Cl_(4) by 0.6^(@)C. K_(b)(C Cl_(4)) is5.03d kg "mol"^(-1)K, K_(f)(CCl_(4)) is31.8 kg "mol"^(-1)K and dencity of solution is 1.64 gm/ml . The Osmotic pressure at 298K is : |
|
Answer» `3.79 ` ATM |
|
| 46. |
The addictive property of narcotic drugs is mainly due to their ability to produce |
|
Answer» EUPHORIA |
|
| 47. |
The actual order of reactivity ofalkyl halide is |
|
Answer» `R-CL GT R-Br gt R-I` |
|
| 48. |
The actual electron confuguration of La(Z=57) is …. |
|
Answer» `[Xe] 4F^(1)5d^(0)6s^(2)` |
|
| 49. |
The actual density of nitrogen is 1.2519 "lit"^(-1) . The density of nitrogen obtained from the atmosphere is 1.2572 g "lit"^(-1) . This is because of the fact that atmospheric nitrogen contain |
|
Answer» ARGON and other NOBLE gases |
|
| 50. |
The activity per ml of a solution of radioactive substances is x.How much water be added to 200 ml of this |
|
Answer» In VAPOUR PHASE, `x'_A=0.6,x'_B=0.4` `x'_A=p_A/P=0.6` `p_A/(p_A+p_B)=(p_A^@x_A)/(p_A^@x_A+p_B^@x_B)` `0.6=(0.3p_A^@)/(0.3p_A^@+0.7p_B^@)`…(1) `x'_B=p_B/P=p_B/(p_A+p_B)=0.4` `IMPLIES (0.7p_B^@)/(0.3p_A^@+0.7p_B^@)=0.4`...(2) DIVIDING (1) by (2) `(0.3p_A^@)/(0.7p_B^@)=0.6/0.4` `implies p_A^@/p_B^@=(0.6xx0.7)/(0.4xx0.3)=7/2=3.5` |
|

