Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The activity of transition elements is related to their - |
|
Answer» variable OXIDATION states |
|
| 2. |
The activity of the hair of an Egyptian mummy is 7 disintegrations minute^(-1). Find the age of mummy. Given t_(1//2) of .^(14)C is 5770 yearand disintegration rate offresh sample of .^(14)C is 14 disintegrations minute^(-1). |
| Answer» SOLUTION :`5770` YEARS | |
| 3. |
The activity of the radioactive sample drops to(1/64)^(th)of its original value in 2 hr find the decay constant (lambda). |
|
Answer» |
|
| 4. |
The activity of the enzymes pepsin is maximum at the pH value of |
|
Answer» 1.5 |
|
| 5. |
The activity of radioisotope changes with: |
|
Answer» temperature |
|
| 6. |
The activity of radioactive isotope decreases with: |
|
Answer» temperature |
|
| 7. |
The activity of carbon -14 in a piece of an ancient wood is only 12.5 %. If the half-life period of carbon -14 is 5760 years, the age of the piece of wood will be (log 2 = 0.3010) |
|
Answer» `17.281 xx 10^(2)` years Now, `t = (2.303)/(lamda) "log" (.^(14)C " original")/(.^(14)C "after time" t)` `= (2.303 xx 5760)/(0.693) "log" (100)/(12.5) = (2.303 xx 5760 xx 0.9030)/(0.693)` ` = 17281 = 172.81 xx 10^(2)` years |
|
| 8. |
The activity of an old piece of wood is just one fourth of a fresh piece of wood. If half life period of ""^(14)C is about 6000 years, the age of old piece of wood is: |
|
Answer» 6000 years |
|
| 9. |
The activity of alkaline earth metalsas reducing agents: |
|
Answer» INCREASES from Be to CA and DECREASES from Ca to BA |
|
| 10. |
The activity of a radioactive nuclide (X^(100) is 6.023 curie at a certain time 't' . If its disintegration constant is 3.7 xx 10^(4)s^(-1) the mass of X after t sec is : |
| Answer» Answer :C | |
| 11. |
The activity of a radioactive sample decreases ot 1//3 of the original activity (A_(0)) in a period of 9 years. After 9 years more ,its activity is (A_(0))/(x). Find the value of X. |
|
Answer» |
|
| 12. |
The activity of a radionuclide (X^(100)) is 6.023 curie . If the disintegration constant is 3.7 xx 10^(4) sec^(-1) , the mass of the radionuclide is : |
|
Answer» `10^(-14) `g `N_(t)=6.023 xx 10^(6) ` atom = `("wt")/(GAW) = (N_(t))/(6.023 xx 10^(23)) = 10^(-15)` GMS |
|
| 13. |
The activity of a radioactive nuclide is 2 xx 10^(7) disintegrations per minute (dpm). After 23.03 minutes, its activity is reduced to 2 xx 10^(6) dpm. What is the average life (in min) of this nuclide |
|
Answer» 1000 or, `lamda = (2.303)/(t) "log" (N_(0))/(N) = (2.303)/(23.03) "log" (2XX 10^(7))/(2 xx 10^(6))` or, `lamda = 0.1 "MIN^(-1)` Now, average lige `= (1)/(lamda) = (1)/(0.1) = 10` min |
|
| 14. |
The activity of a catalyst has been explained in terms of spontaneous adsorption of reactant moleculse on the surface of catlyst and ther by forming an activated adsorbed complex along with evolution of heat . The adorbed activated complex undergoes to decompositio to regencerate catlyst and give products . adorpton is of two types : (i) Physical adsorptio due to van der Waal'sforces of attraction between reactant and catalyst molecules , weak , less exothermic, mulitlay and non-dirctional . (ii) Chemical dasorpton due to free valencies , strong, more exothermic, unilayer. The extent of adormpotion ( x//m) vs temperature at constant pressure gives isobars. Freundilish studied influence of pressure on the phusical adorptons and reported that log x/m log K + 1/n log P. Select the correct statements . |
|
Answer» I, II,III |
|
| 15. |
The activity of a catalyst has been explained in terms of spontaneous adsorption of reactant moleculse on the surface of catlyst and ther by forming an activated adsorbed complex along with evolution of heat . The adorbed activated complex undergoes to decompositio to regencerate catlyst and give products . adorpton is of two types : (i) Physical adsorptio due to van der Waal'sforces of attraction between reactant and catalyst molecules , weak , less exothermic, mulitlay and non-dirctional . (ii) Chemical dasorpton due to free valencies , strong, more exothermic, unilayer. The extent of adormpotion ( x//m) vs temperature at constant pressure gives isobars. Freundilish studied influence of pressure on the phusical adorptons and reported that log x/m log K + 1/n log P. Select the correct satements . |
|
Answer» I, III, IV |
|
| 16. |
The activity ofa radioactive isotope falls to 12.5% in 90 days. Calculate the half life and decay constant. |
|
Answer» Solution :`N_(0)=100 N=12.5 t=90 days` DECAYCONSTANT `LAMBDA=(2.303)/(t) log (N_(0))/(N)` `=0.02558 log 8` `=2.311xx10^(-2) days^(-1)` `t^(1//2)=(0.693)/(lambda)=(0.963)/(2.311xx10^(-2))=29.99 days` |
|
| 19. |
The activity and selectivity of zeolites as catalyst is based on : |
|
Answer» Their PORE size |
|
| 20. |
The activation energy ofr a reaction can be determined by measuring the reaction rate at different: |
|
Answer» temperature |
|
| 21. |
The active component of dynamite is |
|
Answer» keiselghur |
|
| 22. |
The activation energy of reaction is equal to |
|
Answer» THRESHOLD ENERGY for the reaction =Threshold energy - Energy of the reacants |
|
| 23. |
The activation energy of the reaction is 75.2 kJ mol^(-1) in the absence of a catalyst and 50.14" kJ mol"^(-1) with a catalyst. How many times will the rate of reaction grow in the presence of the catalyst if the reaction proceeds at 25^(@)C? (R=8.314" JK"^(-1)" mol"^(-1)). |
|
Answer» |
|
| 24. |
The activation energy of reactant molecules in a reaction depends upon |
|
Answer» Temperature |
|
| 25. |
The activation energy of a reaction is zero . The rate constant of the reaction : |
|
Answer» a) INCREASES with INCREASE in TEMPERATURE |
|
| 26. |
The activation energy of a reaction is zero. The rate constant of the reaction: |
|
Answer» Increases with INCREASE of temperature |
|
| 27. |
The activation energy of a reaction is 94.14" kJ mol"^(-1) and the value of rate constant at 313 K is 1.8xx10^(-1)sec^(-1). Calculate the frequencyfactor, A. |
|
Answer» Solution :Here, we are given that `E_(a)=94.14" kJ mol"^(-1)=94140" J mol"^(-1)` `T=313" K",k=1.8xx10^(-5)sec^(-1)` Substituting the VALUES in the EQUATION : `LOGK=(E_(a))/(2.303" RT")+log" A"` or `""log" A"=logk+(E_(a))/(2.303" RT")=log(1.8xx10^(-5)s^(-1))+(94140" J mol"^(-1))/(2.303xx8.314" JK"^(-1)mol^(-1)xx313"K")` `=(log" "1.8)-5+15.7082=0.2553-5+15.7082=10.9635` `"A"=" antilog "(10.9635)=9.194xx10^(10)s^(-1).` |
|
| 28. |
The activation energy of a reaction is 94.14 KJ/mol and the value of rate constant at 40^@ C is 1.8 xx 10^(-1) sec^(-1). Calculate the frequency factor A. |
|
Answer» SOLUTION :Given, `E_a=94.14xx10^(-3) J MOL^(-1), T=40+273 =313` K , `K=1.8xx10^(-1) "SEC"^(-1)` By using , `K=Ae^(-E_a//RT) rArr ` In K = In A -`E_a/(RT)` Or log K= log A -`E_a/(2.303RT)` Or `log(1.8xx10^(-1))+(94.19xx10^3)/(2.303xx8.314xx313)`=log A Or A = ANTILOG (10.9635) `=9.194xx10^10 "sec"^(-1)` |
|
| 29. |
The activation energy of a reaction is 75.24 kJ "mol"^(-1) in the absence of a catalyst and 50.14 "kJ mol"^(-1) with a catalyst. How many times will the rate of reaction grow in the presence of a catalyst if the reaction proceeds at 25^(@)C? |
|
Answer» SOLUTION :Using Arrhenius equation in the following form and substituting the values, we GET In `k_(1)="In A"-(E_(a_(1)))/(RT) and "In "k_(2)="In A"-(E_(a_(2)))/(RT)` ` "orIn "k_(2)-"In "k_(1)=(1)/(RT) (E_(a_(1))-E_(a_(2)))` ` "orlog"(k_(2))/(k_(1))=(1)/(2.303xxRT) (75.24"kJ mol"^(-1)-50.14"kJ mol"^(-1))` `=(1)/(2.303xxRT) (25.10" kJ mol"^(-1))` `=(1)/(2.303xx8.314JK^(-1)"mol"^(-1)xx298K) xx25100 "J mol"^(-1)=4.40` or`(k_(2))/(k_(1))="ANTILOG "4.40=2.5xx10^(4)=25000` The rate or reaction will grow by 25000 times in the presence of catalyst. The decrease in activation energy TAKES place by`25.1 "kJ mol"^(-1)`. |
|
| 30. |
The activation energy of a reaction is 58.3kJ/"mole". The ratio of the rate constants at 305 K and 300 K is about (R=8.3Jk^(-1)mol^(-1)) ("Antilog" 0.1667=1.468) |
|
Answer» `1.25` |
|
| 31. |
The activation energy of a reaction is 9 kcal/mol. The increase in the rate constant when its temperature is raised from 395 to 300K is approximately |
|
Answer» 0.1 Hence, `k_2/k_1 = 1.288` or `k_2 = 1.288 k_1, i.e., ` INCREASE = 38.8 %`~~`25% |
|
| 32. |
The activation energy of a reaction is 58.3 kJ/mole. The ratio of the rate constnat at 205 K and 300 K is about (R=8.31Jk^(-1)"mole"^(-1))(Antilog 0.1667=1.468) |
|
Answer» 1.25 |
|
| 33. |
The activation energy of a reaction is 56 .2 kJ mole . The ratio of rate constants at 305 K and 300 K is ( R=8 J mol^(-1) K^(-1)) about : |
|
Answer» `1.25` |
|
| 34. |
The activation energy of a reaction is 225 k cal mol"^(-1)and the value of rate constant at 40^@C "is "1.8xx10^(-5)s^(-1).Calculate the frequency factor , a , . |
|
Answer» Solution :Here, we are given that `E_a = 22.5"k CAL mol"^(-1) = 22500 "cal mol"^(-1)` `T = 40^@C = 40+273=313K` `k=1.8xx10^(-5) "sec"^(-1)` Substituting the value in the equation `LOGA = log k + ((E_a)/(2.303RT))` `log A = log (1.8xx10^(-5))+((22500)/(2.303xx1.987xx313))` `logA =log (1.8)-5+(15.7089)` A = antilog (10.9642) `A = 9.208 xx10^(10) "collisions s"^(-1)` |
|
| 35. |
The activation energy of a reaction can be determined from the slope of which of the following graphs ? |
|
Answer» `1nk"VS"(1)/(T)` |
|
| 36. |
The activation energy of a reaction can be determined from the slope of which of the following graphs |
|
Answer» `(lnK)/(T)` vs. T ln K = ln `A - (Ea)/(RT) ln_(E) e ` ln K = ln `A - (Ea)/(R) xx (1)/(T) ` 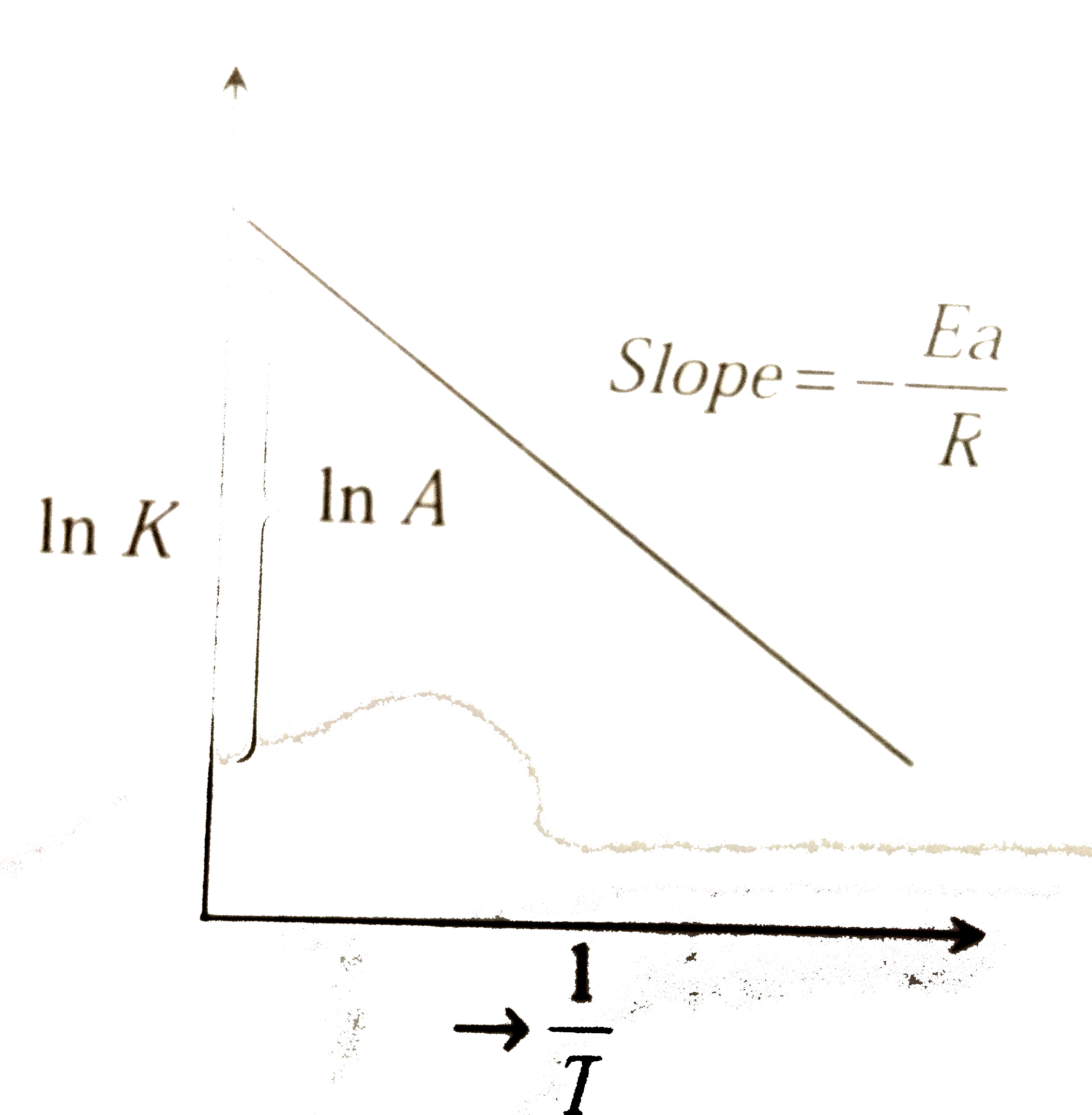
|
|
| 37. |
The energy of activation for a reaction is 100 KJ mol^(-1). The peresence of a catalyst lowers the energy of activation by 75%. What will be the effect on the rate of reaction at 20^(@)C, other things being equal? |
|
Answer» SOLUTION :`2.35xx10^13` TIMES LOG`K_2/K_1=(E_a-E_p)/(2.303RT) RARR log K_2/K_1=(75xx10^3)/(2.303xx8.314xx293)` |
|
| 38. |
The activation energy of a reaction can be determined from the slope of which of the following graphs? |
|
Answer» In `K " VS " 1/T` |
|
| 39. |
The activation energy of a certain uncatalysed reaction at 300 K is 76 k) per mole. The activation energy is lowered to 57 kJ per mole by the lise of a catalyst. By what factor is the rate of the catalysed reaction increased? |
| Answer» SOLUTION :2031 TIMES | |
| 40. |
The activation energy of a reaction can be determined by |
|
Answer» changing the concentration of REACTANTS |
|
| 41. |
The activation energy of a chemical reaction can be determined by, |
|
Answer» evaluating rate CONSTANT at two different temperatures |
|
| 42. |
The activation energy for the reaction,2HI(g)toH_2(g)+I_2(g)is 209.5kj mol^(-1)at 581 K.Calculate the fraction of molecules of reactant having energy equal to or greater than activation energy. |
|
Answer» SOLUTION :Let the fraction be X `E^(E//RT)` i.e.,x=`e^(E//RT)` In `x= -E_a/(RT)log x=-E_a/(2.303RT)=(-209.5xx10^3J)/(2.303xx8.314xx581)=-18.8323` `thereforex=Antilog` of -18.8323=Antilog of `bar19 .1677=1.471xx10^(-19)` |
|
| 43. |
The activation energy for the reaction 2HI_((g))toH_(2(g))+I_(2(g)) is 209.5 KJ mol^(-1) at 581 K.Calculate the fraction of molecules of reactants having energy equal to or freater than activation energy. |
|
Answer» SOLUTION :FRACTION of molecules =`e^(-E_(a))/(RT)` `X=e^((209.5xx1000)/(8.314xx581))` `X=e^(-43.37)` `=1.461xx10^(-19)` OR `therefore K=A e^(-(E_(a))/(RT))` `therefore (k)/(A)=e^(-(E_(a))/(RT))` [But ,`(k)/(A)`=Fraction of molecule=X] `therefore X=-e^(-(E_(a))/(2.303RT)` `therefore log X=-(209.5xx10^(3)J mol)/(2.303xx8.134 J K^(-1)mol^(-1)xx581K)` |
|
| 44. |
The activation energy for the reaction, 2HI_((g))rarr H_(2(g))+I_(2(g)) is 209.5 KJ mol^(-1) at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy. |
|
Answer» Solution :In the given case `E_(a)=209.5 "KJ mol"^(-1)=209500J mol^(-1)` T = 581 K `R = 8.314 JK^(-1) mol^(-1)` Now the FRACTION of molecules of reactants having energy equal to or greater than activation energy is given as. `x = e^(-E_(a)//RT)` `rArr log x = - E_(a//RT)` `rArr log x =-(E_(a))/(2.303 RT)` `rArr log x = (209500 "J mol"^(-1))/(2.303xx8.314 JK^(-1)mol^(-1)xx581)` `= 18.8323` x = Antilogs (18.8323) = Antilogs `BAR(19).1677` `= 1.471xx10^(-19)`. |
|
| 45. |
The activation energy for the reaction, 2" HI "(g)toH_(2)(g)+I_(2)(g), is 209.5" kJ mol"^(-1) at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy. |
|
Answer» Solution :FRACTION of molecules having energy equal to or GREATER than ACTIVATION energy `=x=(n)/(N)=e^(-E_(a)//RT)` `:.LNX=-(E_(a))/(RT)" or "logx=-(E_(a))/(2.303"RT")` or `logx=-(209.5xx10^(3)" J mol"^(-1))/(2.303xx8.314" JK"^(-1)mol^(-1)xx581" K")=-18.8323` `:.x=" Antilog"(-18.8323)="Antilog "bar(19).1677=1.471xx10^(-19)` |
|
| 46. |
The activation energy for the reaction 2HI (g) to H_(2)(g) +I_(2)(g), is 209.5 kJ "mol"^(-1) at 581 K. Calculate the fraction of molecules of reactants hving energy equal to or greater than activation energy. |
|
Answer» Solution :Fraction of molecules having energy equal to or greater than activation energy, X is GIVEN by: `x=(n)/(N)=e^(-E_(a)//RT) or "In"x= -(E_(a))/(RT) or " log"x= -(E_(a))/(2.303RT)` SUBSTITUTING the values, we get `therefore x="Antilog" (-18.8323)="Antilog" bar(19).1677=1.471xx10^(-19)`. |
|
| 47. |
The activation energy for a reaction that doubles the rate when the temperature is raised from 300 K to 310 K is (log 2 = 0.3) |
|
Answer» `50.6 KJ MOL^(-1)` |
|
| 48. |
The activation energy for most of the reaction is approximately 50 kJ mol^(-1). The rate for temperature coefficient for such reaction will be |
| Answer» Answer :A | |
| 49. |
The activation energy for a simple chemical reactionA to Bis E_(a) in forward direction . The activation energy for reverse reaction |
|
Answer» Is ALWAYS double of `E_(a)` |
|
| 50. |
The activation energy for a reaction when the temperature is raised from 300K to 310K is |
|
Answer» `50.6kJ mol^(-1)` `LOG (K_(2))/(K_(1)) = E_(a)/2.303[1/T_(1)-1/T_(2)]` `log(2K_(1))/(K_(1))=(E_(a))/(2.303R) [1/T_(1)-1/T_(2)]` `LOG2 = (E_(a))/(2.303 xx 8.314 Jk^(-1)mol^(-1))[1/(300K-1/310K)]` `0.30 = (E_(a))/(2.303 xx (8.314 J mol^(-1)) xx (9300)` `=53.6 kj mol^(-1)` 
|
|