Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The alkaline earth metal which imparts apple green colour to bunsen flame when introduced in it in the form of its chloride is |
|
Answer» calcium |
|
| 2. |
The alkaline earth metal that imparts apple green colour to the bunsen flame when introduced in it in the form of its chloride is |
|
Answer» Barium |
|
| 3. |
The alkali not suitable for volumettric determination of HCl, using phenolphthalein as an indicator is: |
| Answer» ANSWER :D | |
| 4. |
The alkaline earth metal ion present in chlorophyll is among the metal |
|
Answer» Ca |
|
| 5. |
The alkali metals (Li, Na, K etc.) and the alkaline earth metals (Mg and Ca. together with Zn) are good reducing agents, the former being stronger than the latter. Sodium, for example, reduced elemental chlorine to chloride anion (sodium is oxidized to its cation), as do the other metals under varying conditions. In a similar fashion these same metals reduce the carbon-halogen si converted to halide anion, and the carbon bonds to the metal (the carbon has carbanionic character). Halide reactivity increases in the order: Cl lt Br lt I. These reactions are obviously substitution reactions, but they cannot be classified as nucleophilic substitution, as were the earlier reactions of alkyl halides. Because the functional carbon atoms has been reduced, the polarity of the resulting functional group is inverted (an originally electrophilic carbon become nycleophilic). Reaction carbon in these compounds. The nucleophilic carbon of these reagents also bonds readily with electrophiles such as iodine and carbon dioxide. The polarity of the carbon-oxygen double bonds of CO_(2) makes the carbon atom electrophilic, shown by the formula in the shaded box, so the nucleophilic carbon of the Frignard reagent bonds of this site. ltbr. Product formed is |
|
Answer»
|
|
| 6. |
The alkali metals (Li, Na, K etc.) and the alkaline earth metals (Mg and Ca. together with Zn) are good reducing agents, the former being stronger than the latter. Sodium, for example, reduced elemental chlorine to chloride anion (sodium is oxidized to its cation), as do the other metals under varying conditions. In a similar fashion these same metals reduce the carbon-halogen si converted to halide anion, and the carbon bonds to the metal (the carbon has carbanionic character). Halide reactivity increases in the order: Cl lt Br lt I. These reactions are obviously substitution reactions, but they cannot be classified as nucleophilic substitution, as were the earlier reactions of alkyl halides. Because the functional carbon atoms has been reduced, the polarity of the resulting functional group is inverted (an originally electrophilic carbon become nycleophilic). Reaction carbon in these compounds. The nucleophilic carbon of these reagents also bonds readily with electrophiles such as iodine and carbon dioxide. The polarity of the carbon-oxygen double bonds of CO_(2) makes the carbon atom electrophilic, shown by the formula in the shaded box, so the nucleophilic carbon of the Frignard reagent bonds of this site. What is the product (B) of the following reaction sequence ? Hydrolysis of B gives |
|
Answer»
|
|
| 7. |
The alkali metals have relatively low melting point . Which one of the following alkali metals is expected to have the highest melting point ? |
|
Answer» Li |
|
| 8. |
The alkali metals form salts like hydrides by the direct synthesis at elevated temperature. The thermal stability of these hydrides decreases in which of the following order ? |
|
Answer» `CsHgtRbHgtKHgtNaHgtLiH` |
|
| 9. |
The alkali metals: |
|
Answer» form SALT like ionic hybrides |
|
| 10. |
The alkali metal halides are soluble in water but LiF is insoluble because |
|
Answer» It is amphoteric |
|
| 11. |
The alignment of magnetic dipoles shown below uarr darr darr uarr darr darr represents which of the following ? |
|
Answer» dimagnetism |
|
| 12. |
The alkali halide that is soluble in pyridine is |
|
Answer» NaCl |
|
| 13. |
The algebraic sum of potentials of two electrodes of a galvanic cell is called |
|
Answer» potential difference |
|
| 14. |
The algebrac sum of potential of two electrodes of a galvanic cell is called : |
|
Answer» POTENTIAL defference |
|
| 15. |
The aldol condensation reaction is given by |
|
Answer» Acetophenone 
|
|
| 16. |
The aldol condensation of acetaldehyde results in the formation of: |
|
Answer» `CH_3COCH_2CHO` |
|
| 17. |
The aldehyde which will not reduce Fehling solution readily is |
|
Answer» Ethanal |
|
| 18. |
The aldehyde which react with NaOH to produce an alcohol and sodium salt is |
|
Answer» `HCHO` `2HCHO + NaOH rarr underset("Alcohol")(CH_(3)OH)+ underset("Sodium formate")(HCOONa)` The reaction is Cannizzaro's reaction |
|
| 20. |
The aldehyde having alpha - H atom gives Cannizzaro's reaction is |
|
Answer» `CH_(3) - CH_(2) - CHO` |
|
| 21. |
The alcohol which is most readily dehydrated is: |
|
Answer» 2-butanol |
|
| 22. |
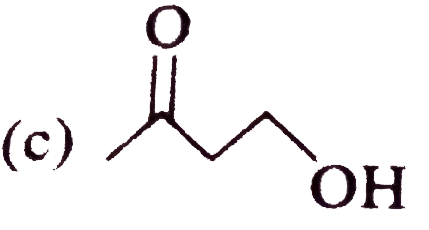
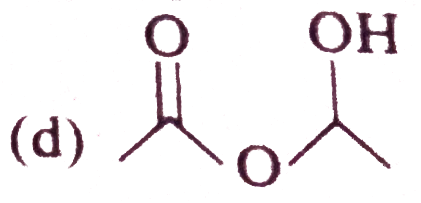
The alcohol which easily reacts with conc. HCl is |
|
Answer» `CH_(3)-CHOH-CH_(2)-CH_(3)` 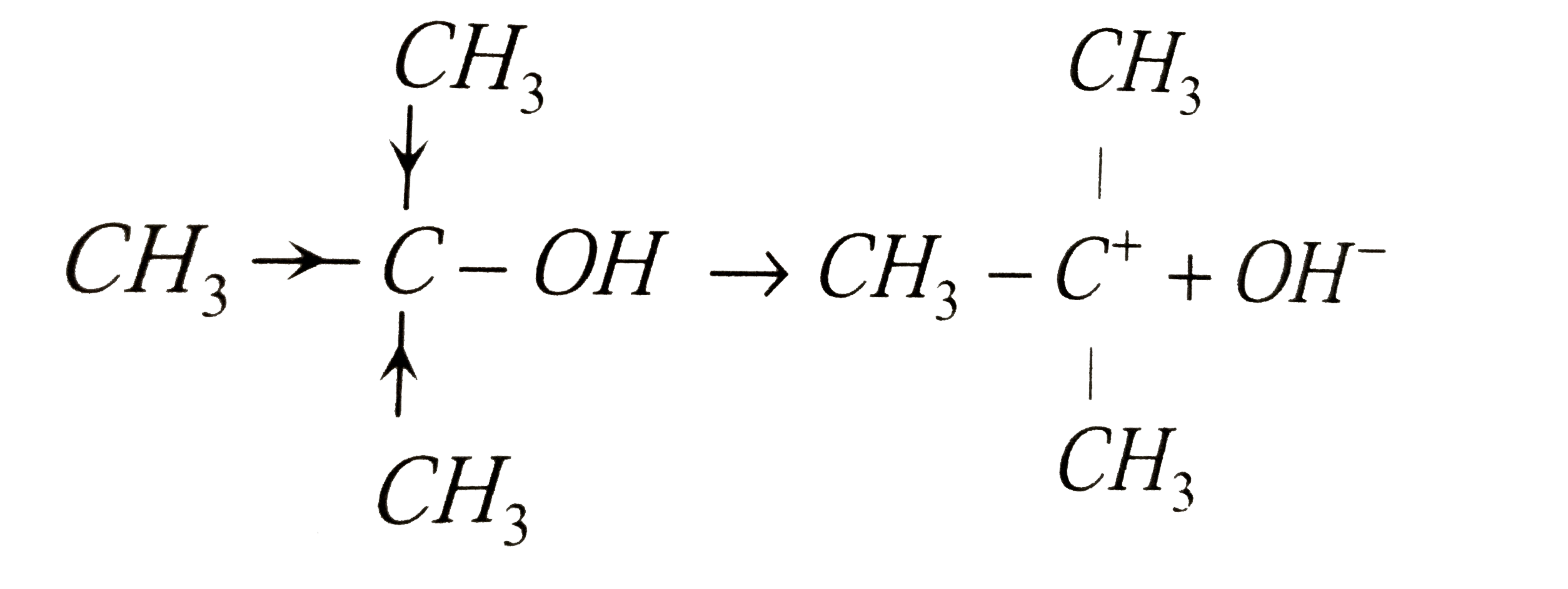
|
|
| 23. |
The aldehyde derived from vitamin B6 is _______ |
|
Answer» PYRIDOXAL |
|
| 24. |
The alcohol whose IUPAC name is 3-ethylpentanol-3 has the structural formula______. |
| Answer» SOLUTION :`(CH_(3)CH_(2))_(3)COH` | |
| 25. |
The alcohol which easily reacts with con HCl is |
|
Answer» `(CH_3)_2 - CH_2 - CH_2OH` |
|
| 26. |
The alcohol which does not give a stable compound on dehydration is |
|
Answer» ETHYL alcohol |
|
| 28. |
The alcohol that produces turbidity immediately with ZnCl_2 + Conc HCl at room temperature is ………………….. . |
|
Answer» BUTAN - 1- OL |
|
| 29. |
The alcohol that responds to Lucas reagent fastest is ….. |
|
Answer» 2-Methyl propan-2-ol |
|
| 30. |
The alcohol that produces turbidity immediately with ZnCl_(2) + conc. HCl at room temperature |
|
Answer» 1-hydroxybutane `(CH_(3))_(3)C-Cl+H_(2)O 3^(@) "reacts IMMEDIATELY" ` `(CH_(3))_(2)CH-OH + HCl overset("Anhyd." ZnCl_(2) + HCl) to ` `(CH_(3))_(2)CH-Cl + H_(2)O2^(@) ` reacts after 5 min. `CH_(3)CH_(2)CH_(2)-OH+HCl overset("Anhyd. " ZnCl_(2)+ HCl)to ` `CH_(3)CH_(2)CH_(2)-Cl+H_(2)O 1^(@)` reacts only on heating . |
|
| 31. |
The alcohol, that is used as a beverage, is |
|
Answer» PROPANOL |
|
| 32. |
The alcohol obtained by the hydrolysis of oils and fats is |
|
Answer» propanol `{:(CH_(2)- OOCR'),(|),(CH-OOCR'),(|),(CH_(2)-OOCR'):} overset(H_(2)O)rarr {:(CH_(2)-OH),(|),(CH-OH),(|),(CH_(2)- OH),("Glycerol"):} + 3R'COOH` |
|
| 33. |
The alcohol that forms fats with fatty acids is : |
|
Answer» Glycerol |
|
| 34. |
The alcohol having least solubility in water is |
|
Answer» Ethanol |
|
| 35. |
The air pollutant NO is produced in automobile engines from the high temperature reaction N_(2)(g)+O_(2)(g)hArr2NO(g),K_(c)=1.7xx10^(-3) at 2300 K. If the initial concentrations of N_(2) and O_(2) at 2300 K are both 1.40 M, what are the concentrations of N_(O), N_(2) and O_(2) when the reaction mixture reaches equilibrium ? |
| Answer» SOLUTION :[NO] = 0.056 M, `[N_(2)] = [O_(2)]` = 1.37 M | |
| 36. |
The air is a mixture of a number of gases. The major components are oxygen and nitrogen with approximate proportion of 20% is to 79 % by volume at 298 K. The water is in equilibrium with air at a pressure of 10 atm. At 298 K if the Henry's law constants for oxygen and nbitrogen are 3.30xx10^(7) and 6.51xx10^(7) respectively, calculate the composition of these gases in water. |
|
Answer» Solution :Percentage of oxygen `(O_(2))` in air = 20% Percentage of NITROGEN `(N_(2))` in air= 79% Also, it is GIVEN that water is in equilibrium with air at a total PRESSURE of 10 atm, that is `(10xx760)` mm Hg = 7600 mm Hg Therefore, Partial pressure of oxygen, `P_(O_(2))=(20)/(100)xx7600` mm Hg = 1520 mm Hg Partial pressure of nitrogen, `P_(N_(2))=(79)/(100)xx7600` mm Hg = 6004 mm Hg 79 `= 79xx760` mm Now, according to Henry.s law : `p=K_(H)x` for oxygen : `p_(O_(2))=KH.x_(O_(2))` `x_(O_(2))=(P_(O_(2)))/(K_(H))` `= ("1520 mm Hg")/(3.30xx10^(7)mm Hg)` (Given `K_(H)=330xx10^(7)` mm Hg) `= 461xx10^(-5)` For nitrogen : `P_(N_(2))=KH.x_(N_(2))` `x_(N_(2))=(P_(N_(2)))/(K_(H))` `= ("6004 mm Hg")/(6.51xx10^(7)mm Hg)` `= 9.22xx10^(-5)` Hence, the mole fractions of oxygen and nitrogen in water are `4.61xx10^(-5)` and `9.22xx10^(-5)`. |
|
| 37. |
The alcohol, C_4H_9OH, when shaken with a mixture of anhydrous ZnCl_2 and conc. HCl give an immediate oil layer product. The alcohol is a |
|
Answer» `H_3C-(CH_2)_3-OH` |
|
| 38. |
The air is a mixture of a number of gases. The major components are oxygen and nitrogen with approximate proportion of 20% is to 79% by volume at 298 K. The water is in equilibrium with air at a pressure of 10 atm. At 298 K, if the Henry's law constants for oxygen and nitrogen at 298 K are 3.30 x 10' mm and 6.51 xx 10^7mm, respectively, calculate the composition of these gases in water. |
|
Answer» <P> Solution : Total pressure of air in equilibrium with water = 10 atmAir contains 20% oxygen and 79% NITROGEN by volume. According to Raoult.s LAW, PARTIAL pressure of oxygen `(P_(O_2)) = 20/100 xx 10 atm = 2 atm = 2 xx 760 mm = 1520mm` Partial pressure of nitrogen `(P_(N_2)) =79/100 xx 10 atm = 7.9 atm = 7.9 xx 760 mm= 6004mm` `K_H(O_2)= 3.30 xx 10^7 mm , K_H (N_2) = 6.51xx 10^7 mm` Applying Henry.s law and substituting the values, we have `P_(O_2) = K_H xx x_(O_2) " or " x_(O_2) = (P_(O_2))/(K_H) = (1520mm)/(3.30 xx 10^7) = 4.61 xx 10^(-5)` `P_(N_2) = K_H xx x_(N_2) " or " x_(N_2) = (P_(N_2))/(K_H) = (6004 mm)/(6.51 xx 10^7) = 9.22 xx 10^(-5) ` |
|
| 39. |
The air is a mixture of a number of gases. The major components are oxygen and nitrogen with approximate proportion of 20% is to 79% by volume at 298 K. The water is in equilibrium with air at a pressure of 10 atm. At 298 K, if the Henry's law constants for oxygen and nitrogen are 3.30xx10^(7) mm and 6.51xx10^(7) mm respectively, calculate the composition of these gases in water. |
|
Answer» Solution :Total pressure of air in equilibrium with WATER = 10 ATM As air contains `20%` oxygen and `79%` nitrogenby volume, `therefore"Partial pressure of oxygen "(p_(O_(2)))=(20)/(100)xx"10 atm = 2 atm "=2XX"760 mm = 1520 mm"` `"Partial pressure of nitrogen "(p_(N_(2)))=(79)/(100)xx"10 atm = 7.9 atm "=7.9xx"760 mm = 6004 mm"` `""K_(H)(O_(2))=3.30xx10^(7)mm, K_(H)(N_(2))=6.51xx10^(7)mm` `"Applying HENRY's law,"p_(O_(2))=K_(H)xx x_(O_(2))"or"x_(O_(2))=(p_(O_(2)))/(K_(H))=("1520 mm")/(3.30xx10^(7)" mm")=4.61xx10^(-5)` `""p_(N_(2))=K_(H)xx x_(N_(2))"or"x_(N_(2))=(p_(N_(2)))/(K_(H))=("6004 mm")/(6.51xx10^(7)" mm")=9.22xx10^(-5)`. |
|
| 40. |
The age of most ancient geological formation is estimated by |
|
Answer» Potassium- Argon METHOD |
|
| 41. |
The age of a specimen, t, is related to the daughter/parent ratio D/P by the equation |
|
Answer» <P>`t = (1)/(lamda)" ln" (D)/(P)` Where N = parent remaining = P and `N_(0)=` Parent + DAUGHTER `= P + D` `P = (P +D) e^(-lamdat) RARR (P)/(P+D) = e^(-lamda t)` `rArr ln (P + D)/(P) = lamda t rArr t = (1)/(lamda) ln (1+(D)/(P))` |
|
| 42. |
The age of minerals and rocks is estimated by: |
|
Answer» Uranium-lead method |
|
| 43. |
The advantage(s) of using O_(2) rather than air in the steel industry is(are) (I) there is a faster conversion , so a givenplant can produce more steel in a day. (II) larger quantities can be handled (III) it gives a pure product and the surface is free fromnitrides |
| Answer» Answer :D | |
| 44. |
the adsorption of solids, from a solution is called |
|
Answer» CHEMICAL adsorption |
|
| 45. |
The affinity of sodium with water is used in |
|
Answer» DRYING of alcohols |
|
| 46. |
The advantages of using carbon to reduce a number of oxides and other compounds are |
|
Answer» EASY AVAILABILITY of coke |
|
| 47. |
The advantage of manufacturing H_(2)SO_(4) by Contact process than other methods is A) The acid obfained is highly pure and concentrated B) It is comparitively cheap method. C) The impurities can be tested and the reactants can be recycled. |
|
Answer» The acid obtained is highly pure and CONCENTRATED |
|
| 48. |
The adsorption of hydrogen on palladium is called : |
|
Answer» HYDROGENATION |
|
| 50. |
The adduct of the compound 'A' obtained by the reaction with excess of isopropyl magnesium iodide , upon hydrolysis gives a tertiary alcohol. The compound 'A' is |
| Answer» Answer :A | |