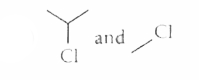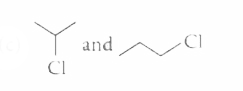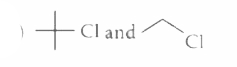Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The amine which reacts with NaNO_2 and dil. HCl to give yellow oily compound is___________. |
|
Answer» ETHYLAMINE |
|
| 2. |
The amine which can react with C_(6)H_(5)-SO_(2)-Cl to form a product insoluble in alkali shall be |
|
Answer» Primary AMINE |
|
| 3. |
The amine which liberate nitrogen on reaction with nitrous acid is |
|
Answer» ISOPROPYL amine |
|
| 4. |
The amine that does not form hydrogen bonds is |
|
Answer» Isopropyl amine |
|
| 5. |
The amine 'A' when treated with nitrous acid give yellow only substance. The amine A is |
|
Answer» triethylmine `CH_(3)NHC_(6)H_(5) overset(HNO_(2))underset("cold") to CH_(3)-underset(C_(6)H_(5))underset(|)(N)-N=0 + H_(2)O` |
|
| 6. |
The amide contains |
|
Answer» DATIVE bond |
|
| 7. |
The amide contains : |
|
Answer» SIGMA BOND only |
|
| 8. |
The alpha-particle is identical with |
|
Answer» HELIUM nucleus |
|
| 9. |
The alpha-hydrogen atom attached to the carbonyl group is acidic in nature because the carbanion which is left gets resonance stabilished. As a result, these compounds take part in the aldol condensation reactions. The carbanion acts as a nucleophile in these reactions. The compounds in which alpha-hydrogen is not acidic take part in the Cannizzaro's reaction. In this reaction, one molecule of the carbonyl compound under consideration is reduced to primary alcohol while the other is oxidised simutaneously to the carboxylic acid. A mixture of cinnamaldehyde and crotonaldehyde is treated with concentrated alkali. C_(6)H_(5)overset(beta)(C)H=overset(alpha)(C)HCHO+overset(gamma)(C)H_(3)overset(beta)(C)H=overset(alpha)(C)HCHOoverset(OH^(-))rarr Which statement is true about the above reaction ? |
|
Answer» Aldol condensation takes place and `alpha`-carbon atom of crotonaldehyde provides the CARBANION 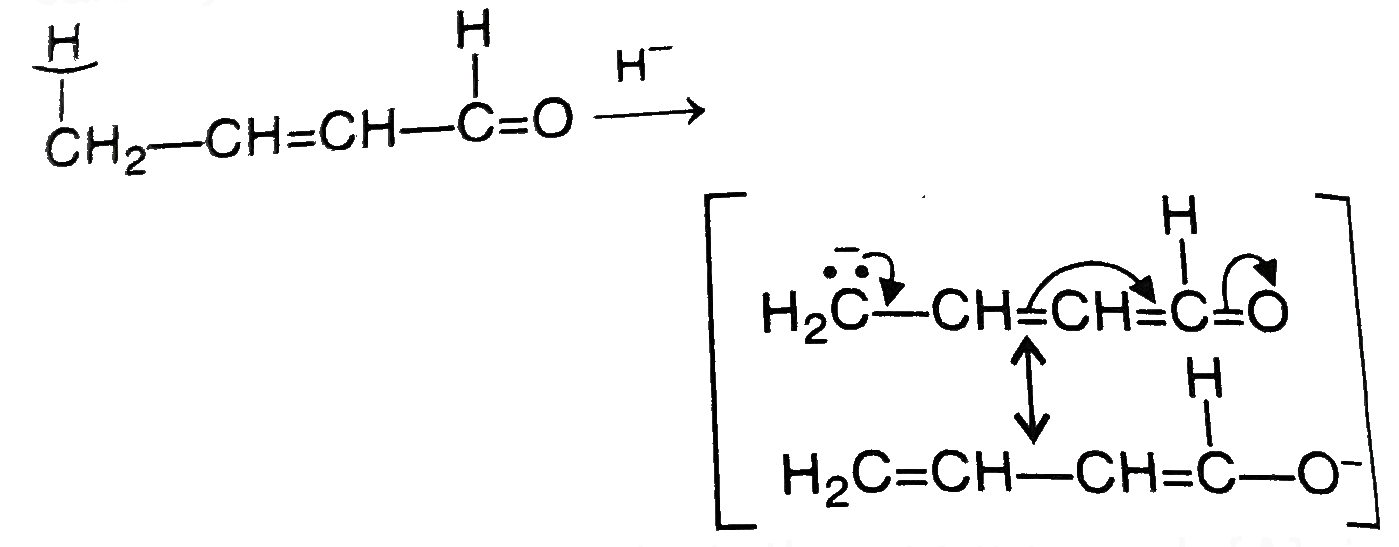
|
|
| 10. |
The alpha-hydrogen atom attached to the carbonyl group is acidic in nature because the carbanion which is left gets resonance stabilished. As a result, these compounds take part in the aldol condensation reactions. The carbanion acts as a nucleophile in these reactions. The compounds in which alpha-hydrogen is not acidic take part in the Cannizzaro's reaction. In this reaction, one molecule of the carbonyl compound under consideration is reduced to primary alcohol while the other is oxidised simutaneously to the carboxylic acid. Compound A (molecular formula C_(3)H_(8)O) is treated with acidified dichromate to form a product B (molecular formula C_(3)H_(6)O). Bforms shining silver mirror on warning with ammoniacl silver nitrate. B when treated with an aqueous solution of H_(2)NCOHNH_(2)HCl and sodium acetate gives a product C. The structure of C is : |
|
Answer» `CH_(3)CH_(2)CH = N NHCONH_(2)` 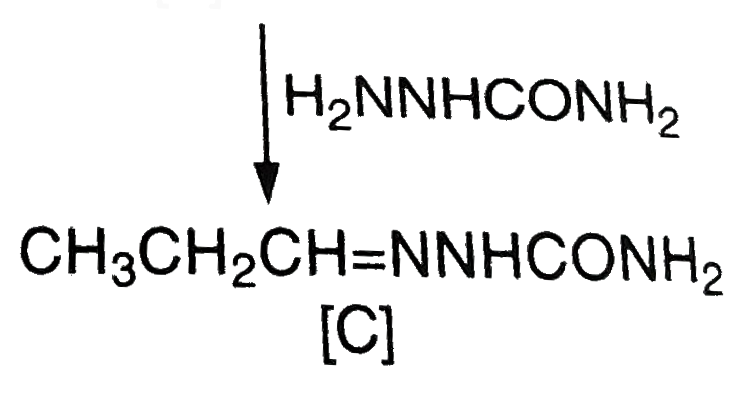
|
|
| 11. |
The alpha-hydrogen atom attached to the carbonyl group is acidic in nature because the carbanion which is left gets resonance stabilished. As a result, these compounds take part in the aldol condensation reactions. The carbanion acts as a nucleophile in these reactions. The compounds in which alpha-hydrogen is not acidic take part in the Cannizzaro's reaction. In this reaction, one molecule of the carbonyl compound under consideration is reduced to primary alcohol while the other is oxidised simutaneously to the carboxylic acid. An organic compound 'A' of the molecular formula C_(5)H_(10)Cl_(2) is hydrolysed to compound 'B' C_(5)H_(10)O which gives an oxime with hydroylamine and yellow precipitate with a mixture of iodine and sodium hydroxide. The compound 'A' should be : |
|
Answer» `CH_(3)CH_(2)C Cl_(2)CH_(2)CH_(3)` 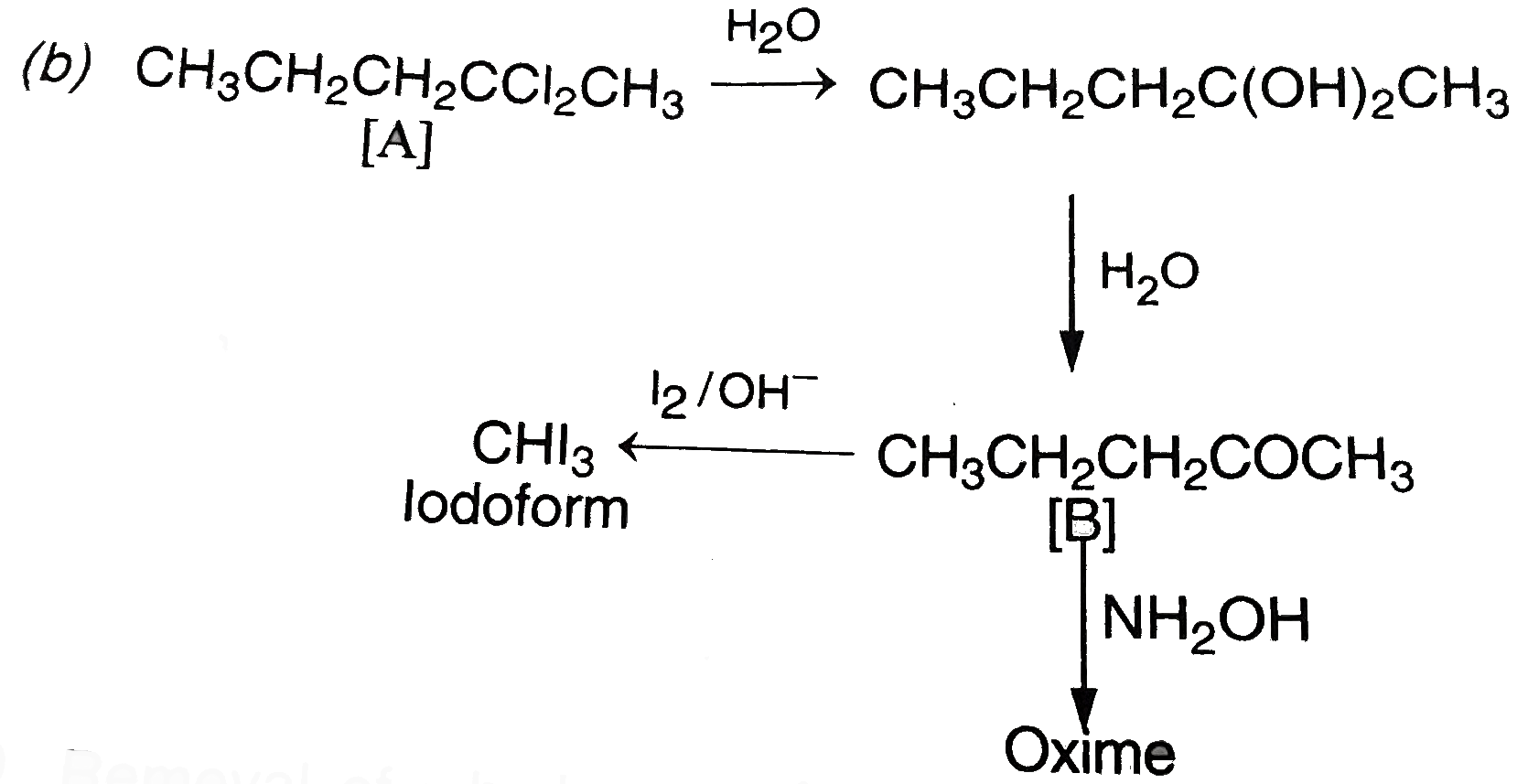
|
|
| 12. |
The alpha- and beta-forms of glucose are |
|
Answer» ISOMERS of D(+) glucose and L(-) glucose respectively |
|
| 13. |
The alpha-helical structure of protein is stabilized by |
|
Answer» dipeptide bond |
|
| 14. |
The alpha-amino acid which does not give purple colour in the ninhydrin test is |
|
Answer» proline |
|
| 16. |
The alloy used in dental filling contains |
|
Answer» AG and SN |
|
| 17. |
Which one of the following is used to remove moisture from ammonia? |
| Answer» Solution :Devarda.s metal | |
| 18. |
The alloy used in preparation of balance beam and light instruments: |
|
Answer» Copper |
|
| 19. |
The alloy of steel that is used for making automobile parts and utensils is : |
|
Answer» STAINLESS STEEL |
|
| 20. |
The alloy containing highest percentage composition of copper is |
| Answer» Solution :Aluminium Bronze | |
| 21. |
The allotrope of sulphur stable below 90^@ C is: |
|
Answer» ORTHO RHOMBIC sulphur |
|
| 22. |
The alkyne which will react with KMnO_(4) to give pyruvic acid is : |
|
Answer» ETHYNE |
|
| 23. |
The alkyne which gives pyruvic acid (CH_3COCOOH) on oxidation with alk KMnO_4 is: |
|
Answer» `CH-=CH` |
|
| 24. |
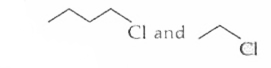
The alkyl halides required to prepare by Wurtz reaction are |
|
Answer»
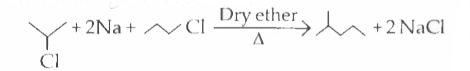
|
|
| 25. |
The alkyl halides required to prepare by wurtz reaction are |
|
Answer»

|
|
| 27. |
The halide which does not give a precipitate with AgNO_3 is: |
|
Answer» ETHYL CHLORIDE |
|
| 28. |
The alkyl halide that undergoes S_(N)I reaction more readily is : |
|
Answer» ethyl bromide `H_(3)C-underset("t-Butyl bromide")underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-BrrarrH_(3)C-underset(3^(@)" carbocation")underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C^(+))+Br^(-)` Ethyl bromide and n-propyl bromide give primary carbocation, isopropyl bromide gives SECONDARY carbocation, t-butyl bromide gives `3^(@)` carbocation. ORDER of stability of carbocation is `3^(@)gt2^(@)gt1^(@)`. HENCE t-butyl bromide is the most stable. |
|
| 29. |
The alkyl group of Grignard reagent acts as: |
|
Answer» FREE radical |
|
| 30. |
The alkyl cyanides when hydrolysed to the corresponding acid, the gas evolved is |
| Answer» Answer :C | |
| 31. |
The alkene which on reductive ozonolysis yields acetone is: |
|
Answer» `CH_2 = CH_2` |
|
| 32. |
The alkene which on ozonolysis yields acetone is: |
|
Answer» `CH_2 = CH_2` |
|
| 33. |
The alkene which on oxidation with acidified KMnO_(4) gives acetic acid is : |
|
Answer» Ethylene |
|
| 34. |
The alkene that will give the same product with HBr in the absence as well as in the presence of peroxide is |
|
Answer» 2-butene |
|
| 35. |
The alkene that exhibits geometrical isomerism is: |
|
Answer» PROPENE |
|
| 36. |
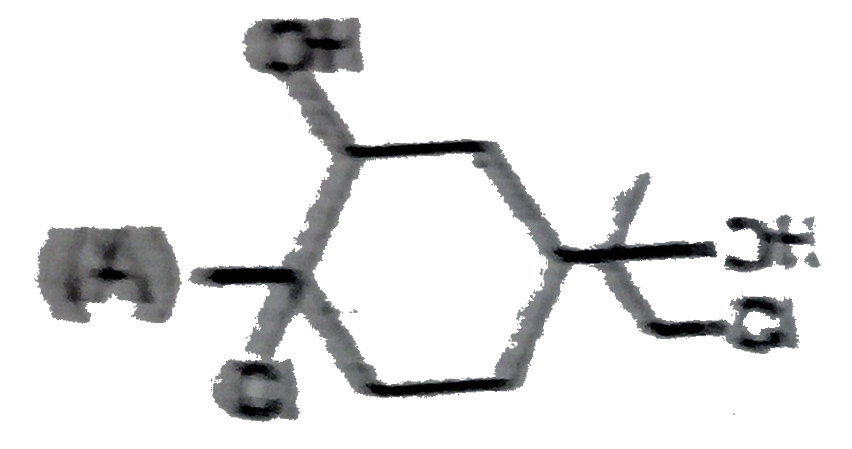
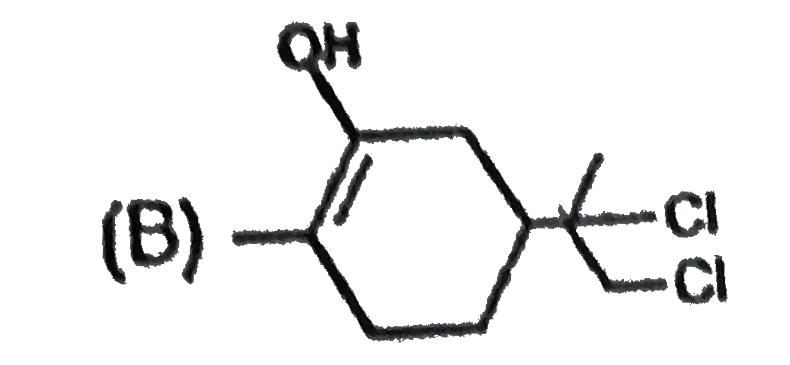
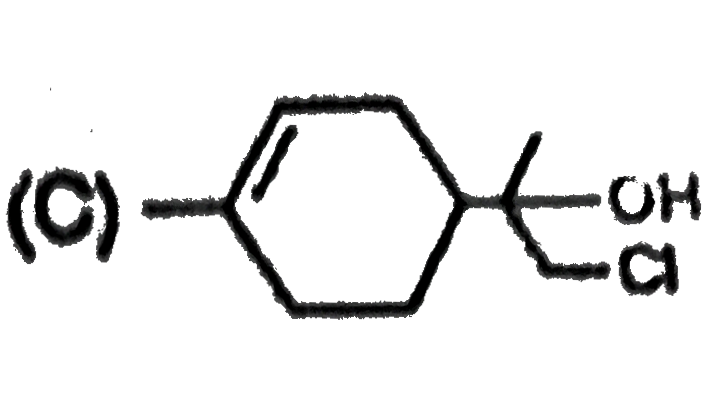
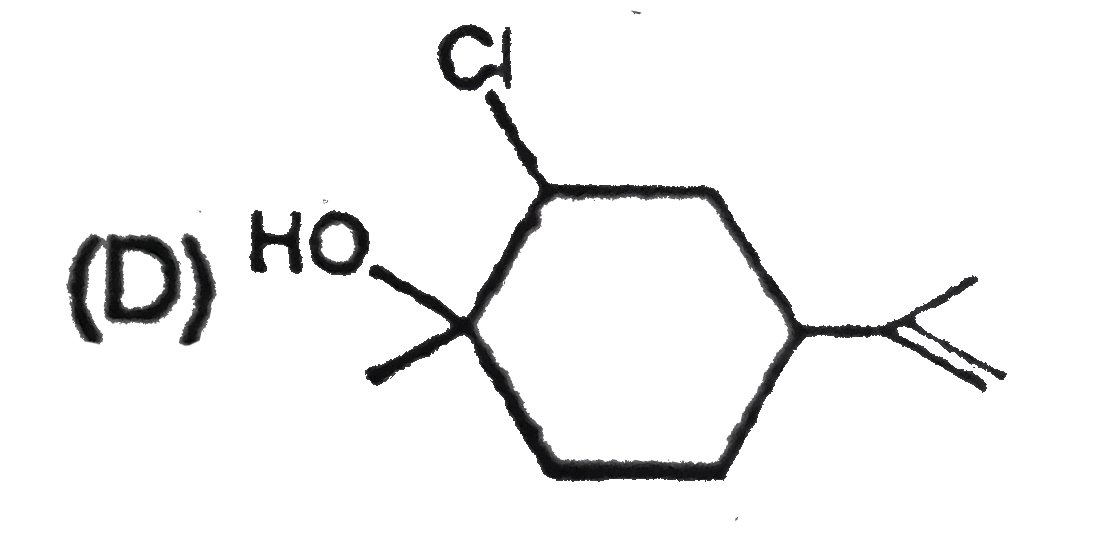
The alkene limonene has the following structure, Which product results from the reaction of limonene and 1 molar equivalent chlorine water ? |
|
Answer»
|
|
| 37. |
The alkene that exhibits geometrical isomerism is : |
|
Answer» PROPENE |
|
| 38. |
The alkene formed as a major product in the above elimination reaction is |
Answer» Solution :  During Hofmann ELIMINATION more ACIDIC `beta`-hydrogen is eliminated. |
|
| 39. |
The alkene C_6H_10 producing OHC-(CH_2)_4-CHO on ozonolysis is |
|
Answer» Hexene-1 |
|
| 40. |
The alkane,(a) C_(5)H^(12)and(B) C_(8) h_(18), on treatmentwithchorine give only one monochloride ,Givethe structuresof eachalkane and itscholride . |
|
Answer» Solution :`(a) (CH_(3))_(4)Cto (CH_(3))_(3)C-CH_(2)Cl,` `(B)(CH_(3))_(3)C-(CH_(3))_(3)to (CH_(3))_(2)CH_(2)Cl]` |
|
| 41. |
The alkane which does NOT undergo nitration is ___________. |
|
Answer» butane |
|
| 42. |
The alkaline hydrolysis of fats to give glycerol is known as …………………… . |
|
Answer» ESTERIFICATION |
|
| 43. |
The alkaline hydrolysis of fats gives glycerol and the reaction is known as …………………….. . |
| Answer» SOLUTION :SAPONIFICATION | |
| 44. |
The alkaline hydrolysis of fats to give glycerol is known as ……… . |
|
Answer» Esterification |
|
| 45. |
The alkaline hydrolysis of ethyl acetate is represented by the equation CH_(3) COOC_(2)H_(5) + NaOH to CH_(3) COONa + C_(2)H_(5) OH Experimentally it is found that for this reaction (dx)/(dt) = k [CH_(3)COOC_(2)H_(5)][NaOH] Then the reaction is |
|
Answer» Bimolecular and of FIRST ORDER |
|
| 47. |
Thealkalinehydrolysisofesteris knownas |
|
Answer» dehydrogenation ` R -overset(O) overset("||") Cunderset( "easter") (- O ) - R '+ NaOHoverset (H_ 2 O )to ` `R - overset (O )overset( "||") C underset("sodium carboxylate")( - O ^ (- ) )- Na ^(+ )+underset ( "alcohol")(R'OH )` |
|
| 48. |
The alkaline earth metals forming predominantly covalent compounds is: |
|
Answer» BARIUM |
|
| 49. |
The alkaline earth metals Ba,Sr,Ca and Mg may be arranged in the order of their decreasing first ionisation potential as |
|
Answer» Mg,CA,Sr,BA |
|
| 50. |
The alkaline earth metal with least density is |
|
Answer» MG `{:(,"Be","Mg","Ca","Sr","BA","Ra"),("Density (g/mL)",1.84,1.74,1.55,2.63,3.59,5.5):}` |
|