Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The complex that violates the E AN: |
|
Answer» POTASSIUM ferrocyanide |
|
| 2. |
The complex showing a spin-only magnetic moment of 2,82 B.M. is |
|
Answer» `Ni(CO)_(4)` `Ni(28) = 3d^(8) 4s^(2)` `Ni^(+2) = 3d^(8) `  No. of unpaired electrons =2 MAGNETIC moment `MU = 2.82 BM` |
|
| 3. |
The complex [Pt(NH_3)_6]Cl_4 furnishes: |
|
Answer» 5 ions |
|
| 4. |
The complex, [Pt(py)(NH_3)BrCl]will have how many geometrical isomers ? |
|
Answer» 2 |
|
| 5. |
The complex [Pt(py)(H_(2)O)(NH_(3))(NO_(2))ClBr] has : |
|
Answer» 10 geometrical isomers, each ONE is OPTICALLY active |
|
| 6. |
The complex, [Pt(Py)(NH_(3))BrCl] will have how many geometrical isomers ? |
|
Answer» 3 |
|
| 7. |
The complex [Pt(NH_(3))_(4)]^(2+) has structure :- |
|
Answer» SQUARE planer |
|
| 8. |
The complex [Pt(NH_(3))_(4)]^(2+) has ……….structure. |
|
Answer» Square planar `PT^(2+)=[Xe]4f^(14)5d^(8)` 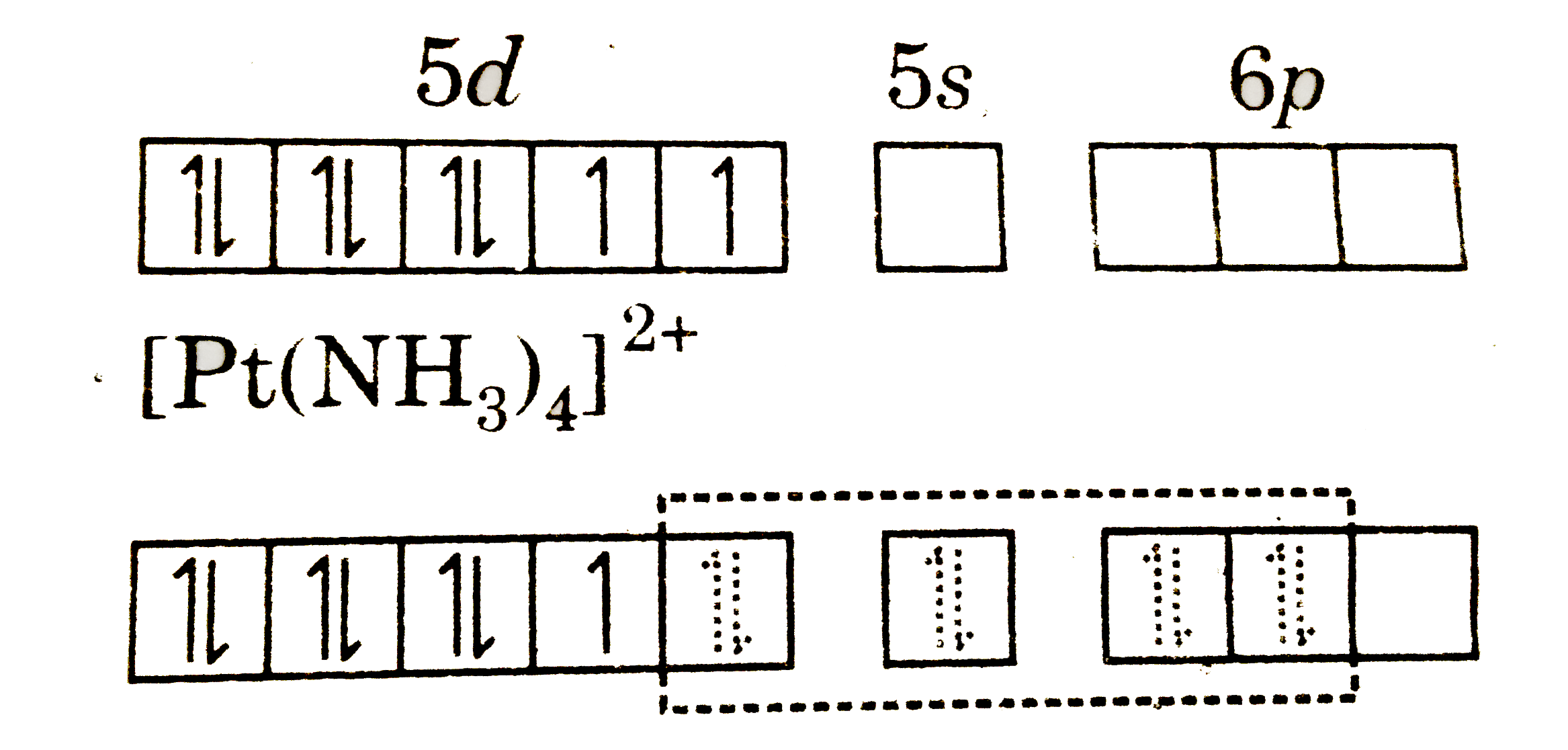
|
|
| 9. |
The complex [Pt(NH_(3))_(4)]^(2+) has ……….structure : |
|
Answer» SQUARE PLANAR |
|
| 10. |
The complex [Ni(CN)_(4)]^(2-) is diamagnetic and it involves the following hybridisation of nickel : |
|
Answer» `SP^(3)` |
|
| 11. |
The complex K_(4)[Zn(CN)_(4)(O_(2))_(2)] is oxidised into K_(2)[Zn(CN)_(4)(O_(2))_(2)], then which of the following is correct ? |
|
Answer» Zn (II) is oxidised into Zn (IV) `{:(""O_(2)^(-),to,""O_(2)^(-)),("(Paramagnetic)",,"(Paramagnetic)"),(" "B.O.=1.5,," "B.O.=2.0),(""(2u.P.e^(-)),,""(2u.P.e^(-))):}` |
|
| 12. |
The complex iron [Co(H_(2)O)_(5) (ONO)]^(2+) and [Co(H_(2)O)_(5) NO_(2)]^(2+) are called |
|
Answer» LINKAGE isomers |
|
| 13. |
The complex ions [Fe(CN)_(6)]^(3-) and [Fe(CN)_(6)]^(4-) : |
|
Answer» Are both octahedral and PARAMAGNETIC |
|
| 14. |
The complex K_3[Fe(CN)_6] should have a spin only magnetic moment of |
|
Answer» `SQRT3` B.M. |
|
| 15. |
The complex ion [Ni(NH_(3))_(4)]^(2+) on treatment with conc. HCl gives two compounds (A) and (B) having the formula [Ni(NH_(3))_(2)Cl_(2)]. Compound (A) can be converted into compound (B) by boiling with dilute HCl. The solution of (A) reacts with oxalic acid to form [Ni(NH_(3))_(2)(C_(2)O_(4))] while solution of (B) does not react with oxalic acid. Identify (A) and (B) and also show their geometry. |
|
Answer» Solution :`[Ni(NH_(3))_(4)]^(2+)+2HClrarrunderset((A) and (B))([Ni(NH_(3))_(2)Cl_(2)])+2 NH_(3)` COMPOUND (A) is a cis-isomer which can easily form a chelate ring with the oxalate LIGAND  Both compound (A) and (B) are square planar and compound A (cis-form) can be CONVERTED into compound B (trans-form) by boiling with dilute HCL. |
|
| 16. |
The complex ion which has no. ‘d’ electrons in the central metal atom is: |
|
Answer» `MnO_4^-` |
|
| 17. |
The complex ion which has no 'd'-electrons in the central metal atom is |
|
Answer» `[MnO_(4)]^(-)` `Mn^(7+) (Z = 25) : [Ar] 4s^(0) 3d^(0)` |
|
| 18. |
The complex ion which has no d electrons in the central metal atom is |
|
Answer» `[MnO_(4)]^(-)` |
|
| 20. |
The complex ion having minimum magnitude of triangle_circ (CFSE) in octahedral field is |
|
Answer» `[CoCl_6]^(3-)` |
|
| 21. |
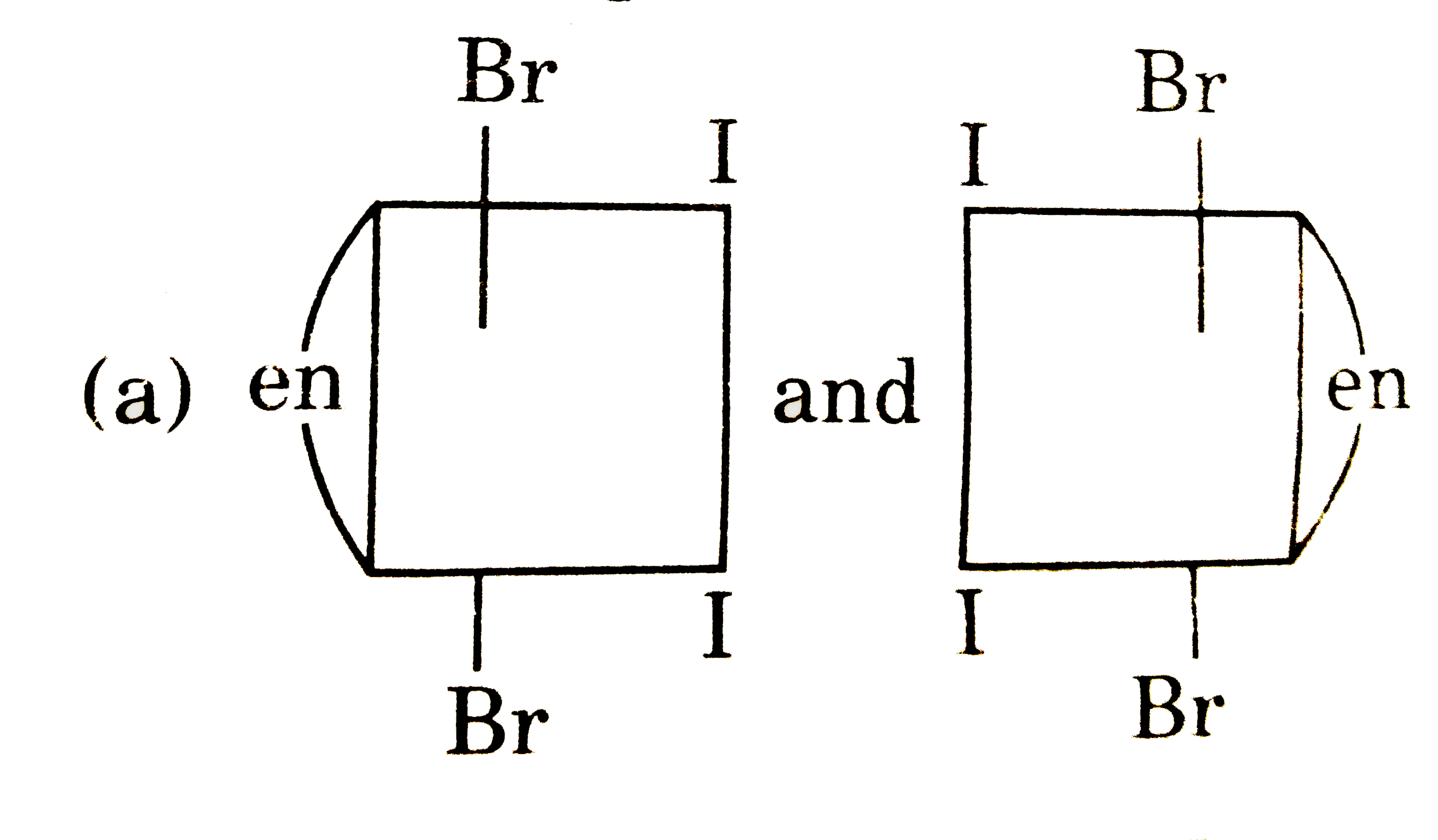
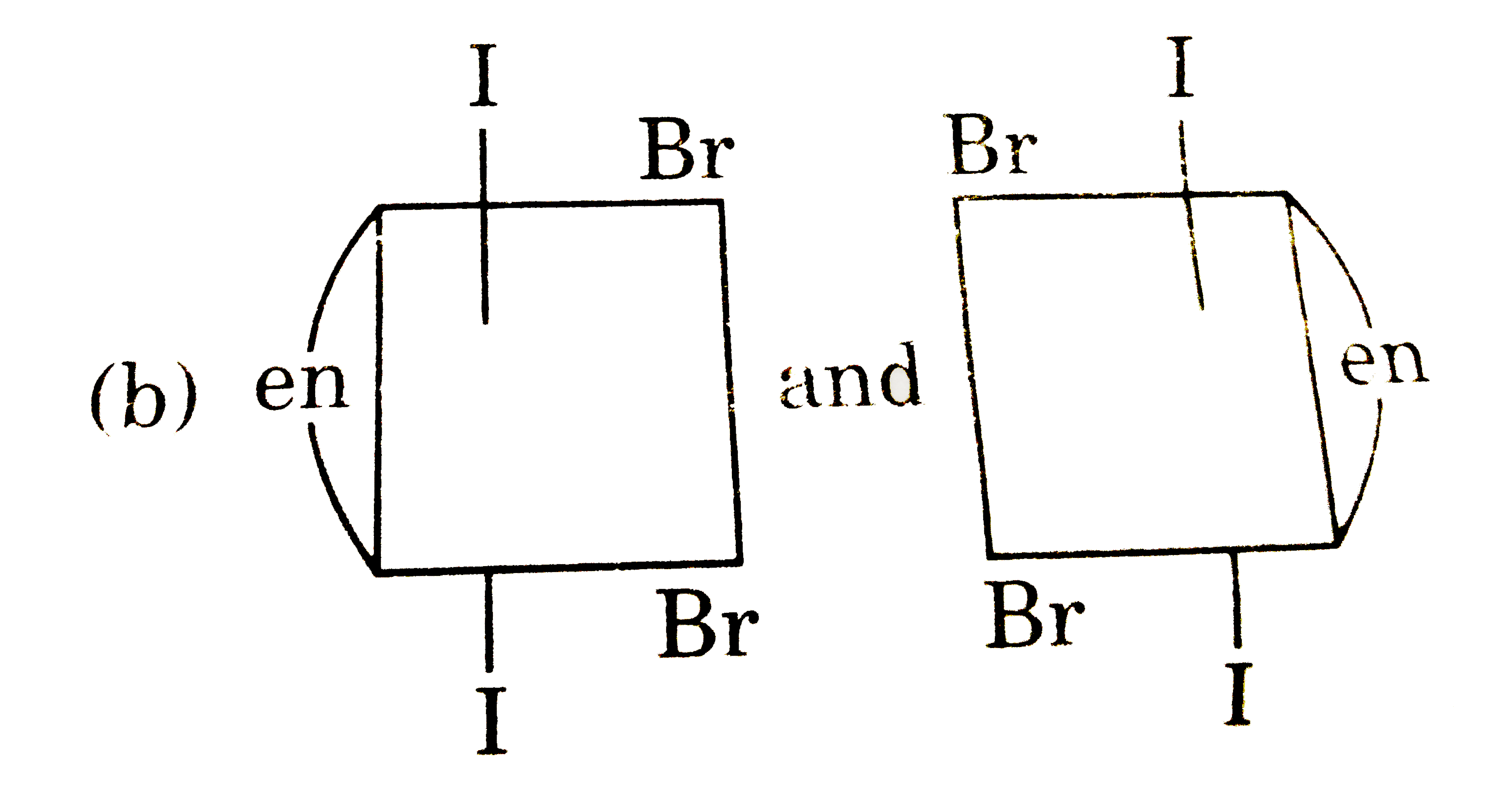
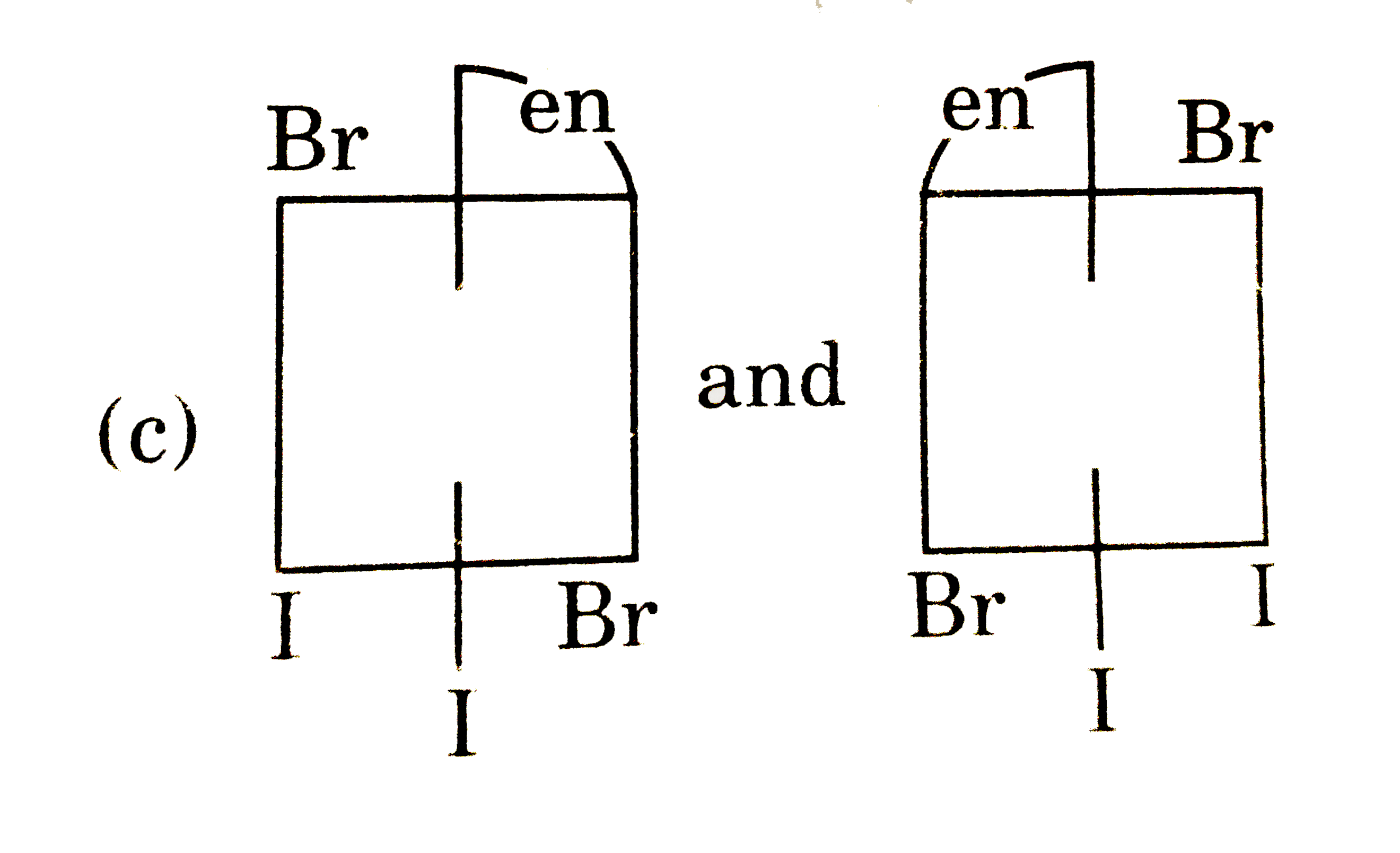
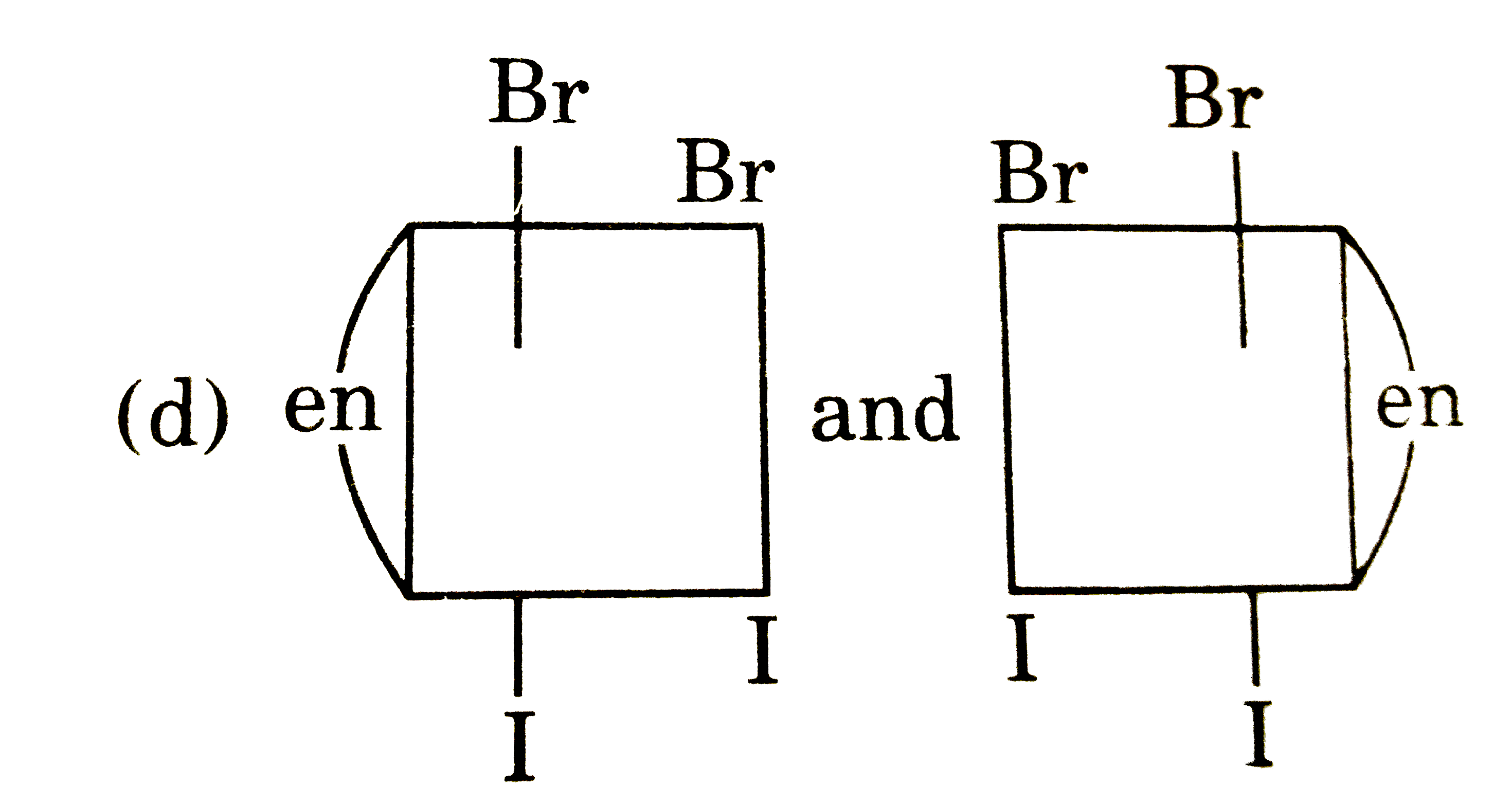
The complex ion has two optical isomers. Their correct configuration are : |
|
Answer»
|
|
| 22. |
The complex ion having minimum magnitude of Delta_o(CFSE) is |
|
Answer» `[CO(NH_(3))_(6)]^(3+)` `Cl^(-) LT H_(2)O lt NH_(3)lt CN^(-)` |
|
| 23. |
The complex ion [Fe(CN)_(6)]^(4-) contains: |
|
Answer» total of 36 electrons on `FE^(2+)` cation |
|
| 24. |
The complex ion [Cu(NH_3)_4]^(2+) has: |
|
Answer» The TETRAHEDRAL configuration with ONE UNPAIRED electron configuration |
|
| 25. |
The complex ion [Co(en)_2Cl_2]^(+)exhibits |
|
Answer» OPTICAL ISOMERISM and LINKAGE isomerism |
|
| 26. |
The complex [Hgl_(3)]^(-) |
|
Answer» is TRIGONAL planar 
|
|
| 27. |
The complex Hg[Co(CNS)_(4)] is named as : |
|
Answer» MERCURY tetrasulphocyanide COBALTATE (III) |
|
| 28. |
The complex Hg[Co(CNS)_4] is correctly named as: |
|
Answer» MERCURY tetrathiocyanato COBALTATE(II) |
|
| 29. |
The complex forming ability of a transition metal depends upon |
|
Answer» availability of vacant d-orbital |
|
| 30. |
The complex [Fe(H_(2)O)_(5)NO]^(2+) is formed in the brown ring test for nitrates when freshly prepared FeSO_(4) solution is added to aqueous solution of NO_(3) followed by addition of cone. H_(2)SO_(4). Select correct statement about this complex. |
|
Answer» COLOUR change DUE to charge transfer. |
|
| 31. |
The complex formed when Al_2 O_3 is leached from Bauxite using concentrated NaOH solution is, |
|
Answer» `Na[Al(OH)_(4)]` |
|
| 32. |
Thecomplex formedwhenAl_2O_3isleachedfrombauxineusingconcentratedNaOHsolutions is |
|
Answer» ` NA[AL(OH)_4] ` |
|
| 33. |
The complex [Fe(H_(2)O)_(5)NO]^(2+) is formed in the brown ring test for nitrates when freshly prepared FeSO_(4) solution is added to aq solution of NO_(3)(-) followed by addition of conc. H_(2)SO_(4). Select correct statement about this complex : |
|
Answer» colour changes due to charge transfer |
|
| 34. |
The complex [Fe(H_(2)O)_(5)NO]^(2+) is formed in the 'brown ring test' for nitrates. Choose the incorrect statement for the complex. |
|
Answer» Its magnetic moment is approximately 3.9 BM `sp^(3) d^(2) , d^(7)` config. 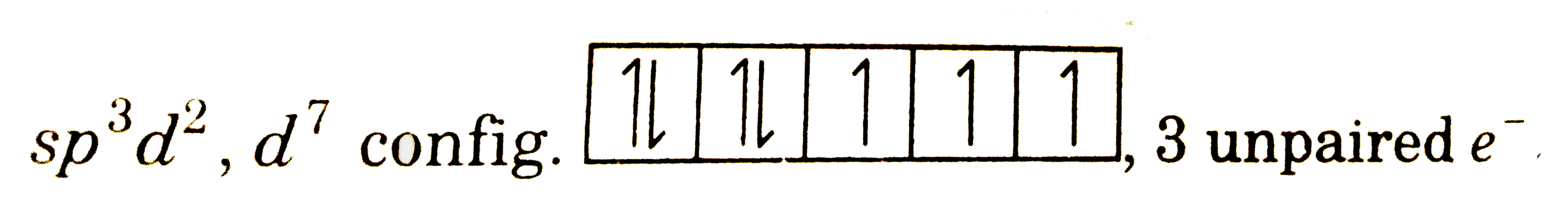 3 UNPAIRED `e^(-)` |
|
| 35. |
The complex [Fe(H_(2)O)_(5)NO]^(2+) is formed in Brown ring test for nitrates select correct statement for the complex ? |
|
Answer» Hybridisation of iron is `sp^(3)d^(2)` `Fe^(+)=3d^(6)4s^(1)` in presence of ligand `Fe^(+)=3d^(7)4s^(0)` hybridisation `=sp^(3)d^(2)`, magnetic moment.s = 3.87 B.M. |
|
| 36. |
The complexexhibits : |
|
Answer» linkage ISOMERISM, geometrical isomerism and OPTICAL isomerism b) linkage isomerism: `[Co(NH_(3))_(4)CL(NO_(2))_(2)]` and `[Co(NH_(3))_(4)(ONO)_(2)]Cl` ii) ionisation isomerism `[Co(NH_(3))_(4)(NO_(2))_(2)]Cl ltimplies[Co(NH_(3))_(4)(NO_(2))_(2)]^(+)+Cl^(-)` 
|
|
| 37. |
The complex entity which shows geometrical as well as optical isomerism, is : |
|
Answer» `[Co(gly)_(3)]` |
|
| 38. |
The complex [Cr(H_(2)O)_(4)Br]Cl gives the test for : |
| Answer» Solution :N//A | |
| 39. |
The complex [Co(NH_(3))_(5)F]^(2+) reacts with water according to the equation, [Co(NH_(3))_(5)F]^(2+)+H_(2)O rarr [Co(NH_(3))_(5)H_(2)O]^(3+)+F^(-) The rate of reaction = "rate constant" xx ["complex"]^(n)xx[H^(+)]^(m)=K["complex"]^(n)[H^(+)]^(m). The reaction is acid catalyzed, i.e., [H^(+)] does not change during the reaction. Thus, rate =K' ["complex"]^(n) where K'=K[H^(+)]^(m) Calculate m and n if they are integers from the following data at 25^(@)C. {:("[complete] M",[H^(+)]M,t_(1//2) ("hour"),t_(3//4)("hour")),(0.1,0.01,1,2),(0.2,0.02,0.5,1):} t_(3//4) is time required for three fourth completion of the reaction. |
|
Answer» |
|
| 40. |
The complex [Cu(NH_(3))_(4)]^(2+) is : |
|
Answer» TETRAHEDRAL with one UNPAIRED ELECTRON |
|
| 41. |
The complex [Co(NH_(3))_(6)][Cr(CN)_(6)]" and "[Cr(NH_(3))_(6)][Co(CN)_(6)] are the examples of which type of isomerism? |
|
Answer» `dsp^(2)` |
|
| 42. |
The complex [Co(NH_(3))_(6)]^(3+) is an inner orbital complex whereas the [CoF_(6)]^(3-) is an outer orbital complex. The number of unpaired electrons in these two complexes are respectively : |
|
Answer» Zero and 4 |
|
| 43. |
The complex[Co(NH_3)_5BR]SO_4 will give white ppt with: |
|
Answer» `PbCI_3` |
|
| 44. |
The complex [Co(NH_(3))_(5)Br] SO_(4) will give white precipitate with |
| Answer» Answer :A | |
| 45. |
The complex, [Co(NH_(3))_(4)Cl_(2)]^(+) is known to exist in two different coloured forms. This is due to |
|
Answer» ionisation isomerism |
|
| 46. |
The complex[Co(NH_(3))_(4)Cl_(2)]Br can exhibit : |
|
Answer» LINKAGE isomerism |
|
| 47. |
The complex [Co(NH_3)_3Cl_3]is : |
|
Answer» Neutral |
|
| 48. |
The complex [Co(NH_3)_3Cl_3] when dissolved in water gives how many ions: |
|
Answer» 2 |
|
| 49. |
The complex compound which does not give precipitate with AgNO_(3) solution is |
|
Answer» `[CO(NH_3)_5Cl]Cl_2` |
|