Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
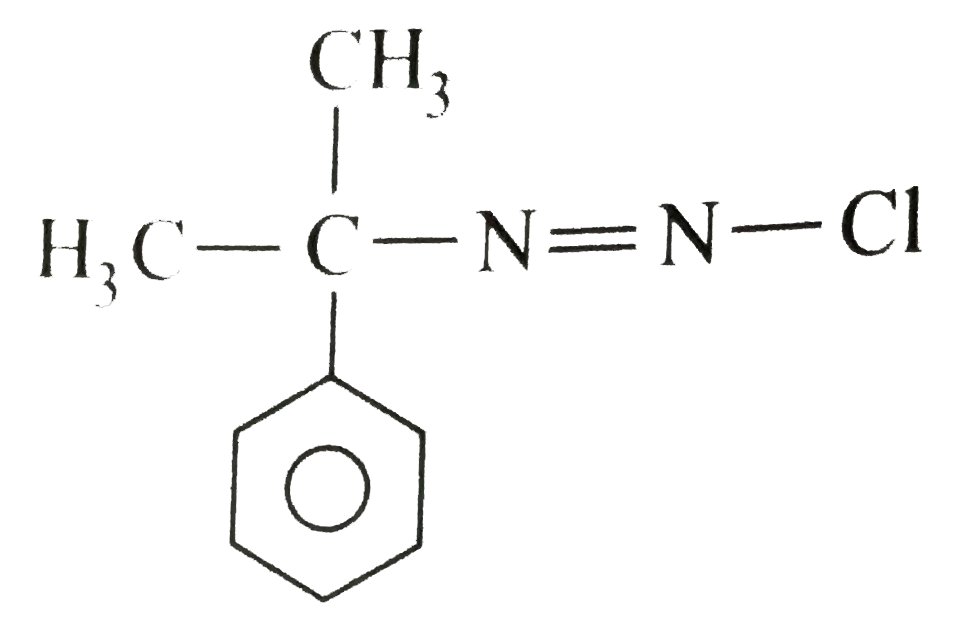
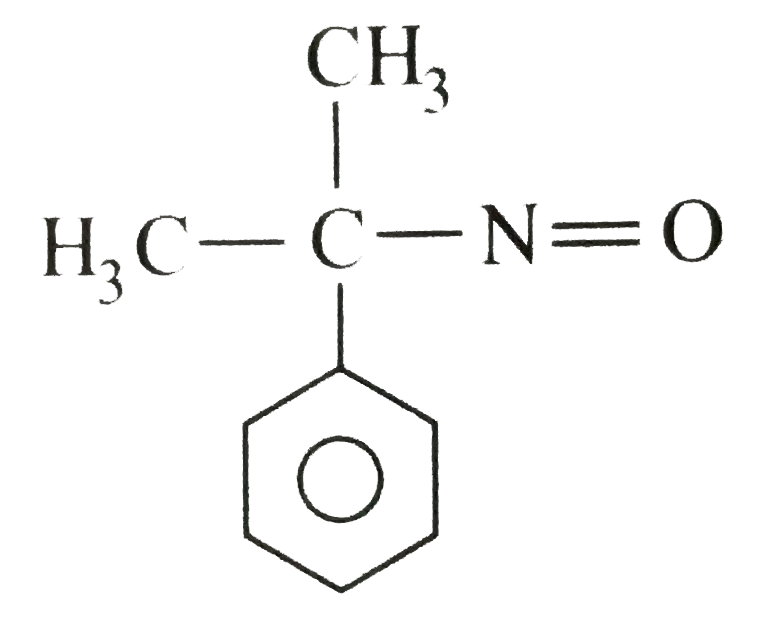
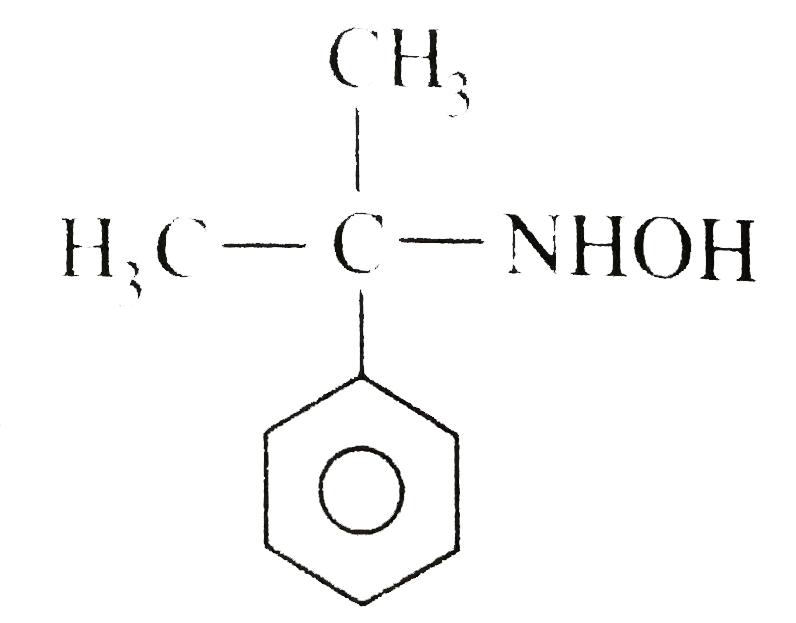
The compound(A) C_(4)H_(11)Nwhichis opticallyactive,dissolvedin conc. HClandreleasednitrogen with HNO_(2)Whatis the compound (A) ? |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)NH_(2)` |
|
| 2. |
The Compound (A) and (B) in the equation given above are |
|
Answer» `CH_(3)COOH, CH_(3)CH_(3)` |
|
| 3. |
The compound A and B are mixed in equimolar proportionto from the products, A+BhArr C+D. At equlibrium, one third of A and B are consumed. The equlibrium constant for the reaction is |
|
Answer» `0.5` So, `K=0.25` |
|
| 4. |
The compound (A) |
|
Answer» a. |
|
| 5. |
The compound 1,2-butadiene has : |
|
Answer» Only SP-HYBRIDIZED CARBON atoms |
|
| 6. |
The compound 2-Methyl-1,3 - butadiene has : |
|
Answer» onlysp hybridised CARBON ATOMS |
|
| 7. |
The compound 1-(N-ethyl -N-methyl) propanamine forms non-superimposable mirror images. But this compound does not show optical acitivity because of |
|
Answer» absence of a chiral N atom  It does not SHOW optical activity because of rapid flipping of one form into the other. |
|
| 8. |
The compostion formulae of gypsum is |
|
Answer» `(CaSO_(4))_(2).2H_(2)O` |
|
| 9. |
The composition of vapour over a binary ideal solution is determined by the composition of the liquid. If X_(A) and Y_(A) are the mole fractions of A in the liquid and vapour, respectively, find the value of X_(A) for which Y_(A)-X_(A) has a maximum. What is the value of the pressure at this composition in terms of P_(A)^(@) and P_(B)^(@). |
|
Answer» `(1)/(P_(T))=(Y_(A))/(P_(B)^(0))+((1-Y_(A)))/(P_(B)^(0))` calculate `(X_(A)-Y_(A)) &` maximise it `X_(A)=(sqrt(P_(A)^(0)P_(B)^(0))-P_(B)^(0))/((P_(A)^(0)-P_(B)^(0)))` & `P_(T)=sqrt(P_(A)^(0)P_(B)^(0))` `X_(A)=((sqrt(P_(A)^(0)P_(B)^(0))-P_(B)^(0)))/((P_(A)^(0)-P_(B)^(0))),P=sqrt(P_(A)^(0)P_(B)^(0))` |
|
| 10. |
The composition of the equilibrium mixture for the equilibrium Cl_(2) hArr 2Cl at 1470 K may be determined by the rate of diffusion of the mixture through a pinhole. It is found that at 1470 K, the mixture diffuse 1.16 times as fast as krypton (83.8) diffuses under the same conditions. Find the degree of dissociation of Cl_(2) at equilibrium. |
|
Answer» Solution :For the EQUILIBRIUM, `{:(,Cl_(2),hArr,2Cl,,),("Initial moles : ",1,,0,,),("Moles at eqb. ",1-x,,2x,,"(x is the DEGREE of dissociation)"):}` Molecular weight of the MIXTURE of `Cl_(2)` and Cl at eqb., i.e., `M_("mix")` is calculated as `M_("mix") = ((1-x) 71 + (2x)35.5)/((1-x) + 2x) = (71)/(1+x)` Now we have, `(r_("mix"))/(r_(Kr)) = sqrt((M_(Kr))/(M_("mix"))) = sqrt((83.8)/(71(1+x))) = 1.16` `THEREFORE x = 0.14` |
|
| 11. |
The composition of the equilibrium mixture Cl_2iff 2Cl,which is attained at 1200^@C , is determined by measuring the rate of effusion through a pinhole. It is observed that at 1.80 mmHg pressure, the mixture effuses 1.16 times as fast as krypton effuses under the same conditions. Calculate the fraction of chlorine molecules dissociated into atoms. (At. wt. of Kr = 84) |
|
Answer» |
|
| 12. |
The composition of the equilibrium mixture (Cl_(2) hArr 2Cl), which is attained at 1200^(@)C, is determined by measuring the rate effusion through a pinhole. It is observed that at 1.80 mm Hg pressure, the mixture effuses 1.16 time as fast as Krypton effuses under the same conditions. Calculate the fraction of chlorine molecules dissociated into atoms (Atmic wt. Kr=84). |
|
Answer» Solution :`(r_("MIX"))/(r_("Kr"))=sqrt((M_("Kr"))/(M_("mix")))` or `1.16 =sqrt((84)/(M))` `(because` TIME of diffusion for both is same) `THEREFORE M =62.425` `Cl_(2) hArr 2Cl` For 1 0 `(1-alpha) 2alpha` TOTAL mole in mixutre `=1-alpha+2alpha=1+alpha` `therefore 71xx(1-alpha)+2alphaxx35.5=(1+alpha)xx62.425` `therefore alpha =0.137` or 13.7% |
|
| 13. |
The composition of sample of wustite is Fe_(0.93).O_(1.00). Which of the following statement is incorrect related to this compound? |
|
Answer» The PERCENTAGE of FE(III) is APPROXIMATELY 15% |
|
| 14. |
The composition of rust is |
|
Answer» `Fe_2O_3 . xH_2O` |
|
| 15. |
The composition of mica is : |
|
Answer» `NaAlSiO_4. 3H_2O` |
|
| 16. |
The composition of 'Golden spangles' is |
|
Answer» `PbCrO_(4)` |
|
| 17. |
The composition of golden spangles is: |
|
Answer» `PbCrO_(4)` |
|
| 18. |
The compositionof 'coppermatte' is |
|
Answer» ` Cu_2S+FES` |
|
| 20. |
The composition of a Vapour over a binary ideal solution is determined by the compostion of the liquid . If X_(A) and Y_(A) are the mole fractions of A in the liquid and Vapourrespectively, findthe value of X_(A) for which Y_(A)-X_(A) has maximum . What is the value of the pressure at this composition in terms of P_(A)^(0) and P_(B)^(0) |
|
Answer» `X_(A)=(P-P_(B)^(0))/(P_(A)^(0)-P_(B)^(0)),P=SQRT(P_(A)^(0)-P_(B)^(0))` |
|
| 21. |
The composition of a sample of wustite is Fe_(0.93) O_(1.00) . What percentage of iron is present in the form of Fe (III)? |
|
Answer» Solution : Let the MOLES of Fe in FEO and `Fe_2O_3`be `n_1`, and `n_2`RESPECTIVELY. THUS, moles of Fe/moles of O `= (n_1 +n_2)/(n_1 + (3n_2)/(2)) = 0.93` 15.05% |
|
| 22. |
The composition of a sample of wurtzite is Fe_(0.93)O_(1.00) what % of Iron present in theform of Fe^(3+)? |
|
Answer» `16.05%` The number of `Fe^(3+)` ions in the crystal be y Total number of `Fe^(2+) and Fe^(3+)` ions is `x+y`. Given that `x+y=0.93` The total charge `=0x(2+)+(0.93-x)(+3)-2=0` `2x+2.97-3x-2=0x=0.79` `"Percentage of "Fe^(3+)=(((0.93-0.79))/(0.93))100=15.05%` |
|
| 23. |
The component which forms a dative bond with ammonia |
|
Answer» `C Cl_(4)` |
|
| 26. |
The component present in greater proportion in water gas is : |
|
Answer» `H_2` |
|
| 27. |
The compoiund (A0 is xylene. On sulphonation, it gives onlyone product (B) which onalkaine fusion followed by acidification yields (C), a phenol. On treatmentwith chlorform and alkali, (C) givesC_(9)H_(1) O_(2)(D). Idenifuy the various organic compounds(A) to (D). |
Answer» Solution :`(A)` is xylene, it can be at `o,m`, and `p`. But it gives only ONE SUBSTITUDE PRODUCT, so `(A)` is `p-` xylene. `(OMP = 231)`. 
|
|
| 28. |
The component of blood responsible for blood clotting is : |
|
Answer» Fibrinogen |
|
| 29. |
The component of a living cell affected by the pollutant SO_2 is |
|
Answer» CELL wall |
|
| 30. |
The component having low melting point can be separated in an impure metal by ________ |
|
Answer» WASHING with water |
|
| 31. |
The complex(es) which is /are bluein colour |
|
Answer» `Fe_(4)[FE(CN)_(6)]_(3)` (b) `Zn_(2)[Fe(CN)_(6)]`,-Zinc ferrocyanide-(Blueish white PPT) c) `Cu_(2)[Fe(CN)_(6)]`- Copper ferrocyanide -(Chocolate brown ppt) d) `Fe_(3)[Fe(CN)_(6)]_(2)`- Ferroferric CYANIDE- (Tumbull.s blue) |
|
| 32. |
The complexes [Pt(NH_(3))_(4)][PtCl_(6)]" and "[Pt(NH_(3))_(4)Cl_(2)][PtCl_(4)] are : |
|
Answer» LINKAGE ISOMERS |
|
| 33. |
The complexes [Pt(NH_(3))_(4)][PtCl_(6)]and [Pt(NH_(3))_(4)Cl_(2)] [PtCl_(4)]are:- |
|
Answer» LINKAGE isomers |
|
| 34. |
The complex(es) which is/are blue in colour: |
|
Answer» `Fe_(4)[(CN)_(6)]_(3)` |
|
| 35. |
The complexes having different CO bond length is/are : |
|
Answer» `Mn_(2)(CO)_(10)` (b) 
|
|
| 36. |
The complexes formed by Cu^(2+) ion are more stable than those formed byCd^(2+) ion because _________. |
|
Answer» the value of the stability constant for `Cd^(2+)` complexes is greater than that of |
|
| 37. |
The complexes [Co(NH_(3))_(6)]]Cr(CN)_(6)] and [Cr(NH_(3))_(6)][Co(CN)_(6)] are the example of which type of isomerism ? |
|
Answer» LINKAGE isomerism |
|
| 38. |
The complexes given below show and |
|
Answer» OPTICAL isomerism |
|
| 39. |
The complexes [Co(NH_3)_6][Cr(CN)_6]and [Cr(NH_3)_6][Co(CN)_6]are the example of which type of isomerism ? |
|
Answer» Geometrical |
|
| 40. |
The complexes [Co(NH_(3))_(6)][Cr(CN)_(6)] and [Cr(NH_(3))_(6)] [Co(CN)_(6)] are the examples of which type of isomerism? |
|
Answer» Linkage isomerism |
|
| 41. |
The complexes [Co (NH_(3))_(6)][Cr(CN)_(6)] and [Cr(NH_(3))_(6)][Co(CN)_(6)]are the example of which type of isomerism? |
|
Answer» GEOMETRICAL ISOMERISM |
|
| 42. |
The compexes [Co(NH_(3))_(6)][Cr(C_(2)O_(4))_(3)] and [Cr(NH_(3))_(6)][Co(C_(2)O_(4))_(3)] |
|
Answer» linkage |
|
| 43. |
The complex with a maximum number of stereoisomers is: |
|
Answer» `[PtCl_(3)(C_(2)H_(4))]^(-)` |
|
| 44. |
The complex which has the highest magnetic moment among the following is |
|
Answer» `[CoF_(6)]^(3-)` |
|
| 45. |
The complex which has no 'd' electrons in the central atom is (Atomic number, Cr= 24, Mn = 25, Fe = 26, Co = 27) |
|
Answer» `[MnO_(4)]^(-)` HENCE, (A) is the correct ANSWER. |
|
| 46. |
The complex which exhibits geometrical as well as optical isomerism is : |
|
Answer» `[Co(gly)_(3)]` 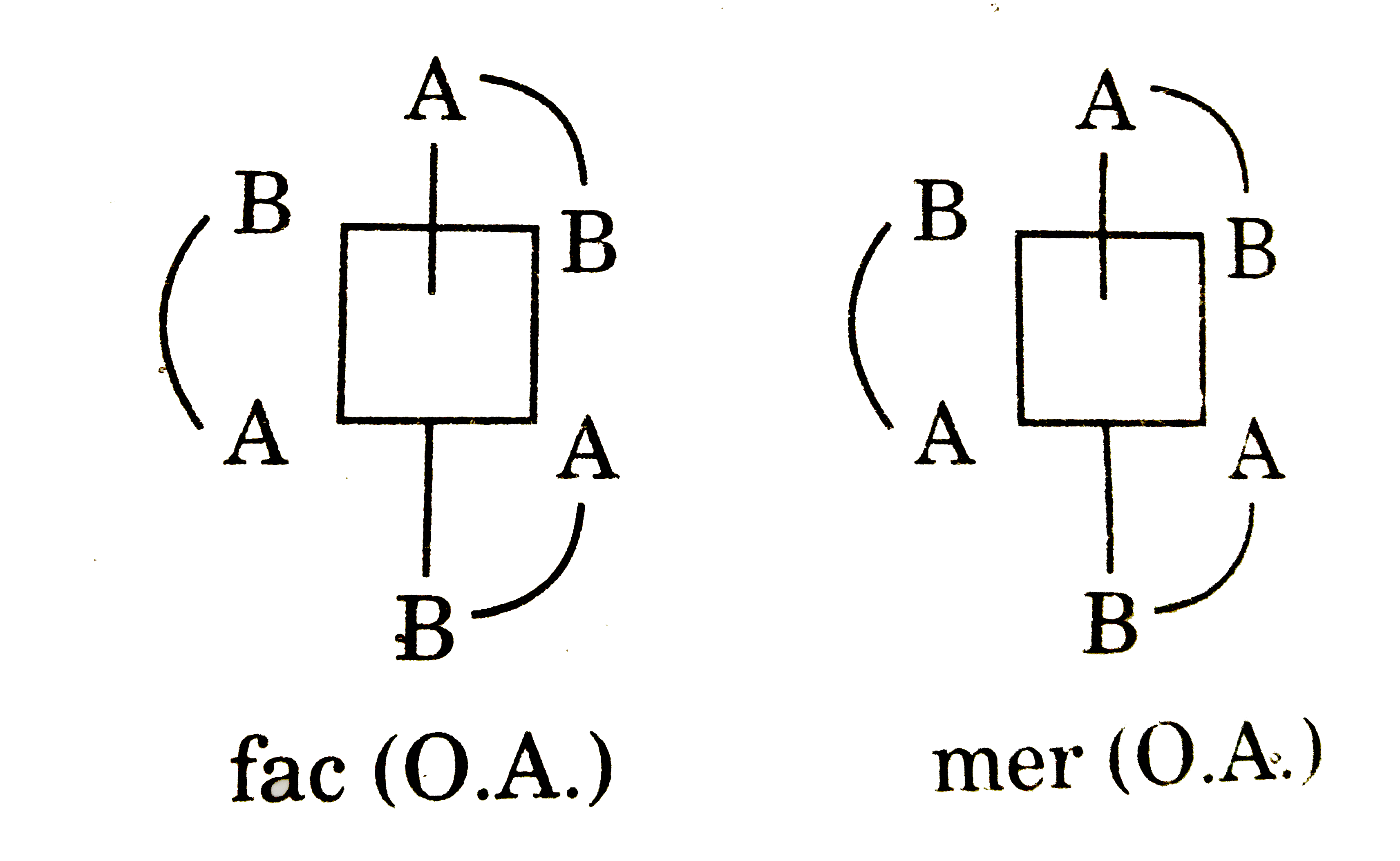
|
|
| 47. |
The complex which exhibits cis-trans isomerism as well as can be resolved into d and ? forms: |
|
Answer» `[Be(ACAC)_2]` |
|
| 48. |
The complex used as an anticancer agent is __________. |
|
Answer» MER - `[CO(NH_3)_3Cl_3]` |
|
| 49. |
The complex that shows optical activity is : |
|
Answer» TRANS-`[CoCl_(2)(en)_(2)]^(+)` 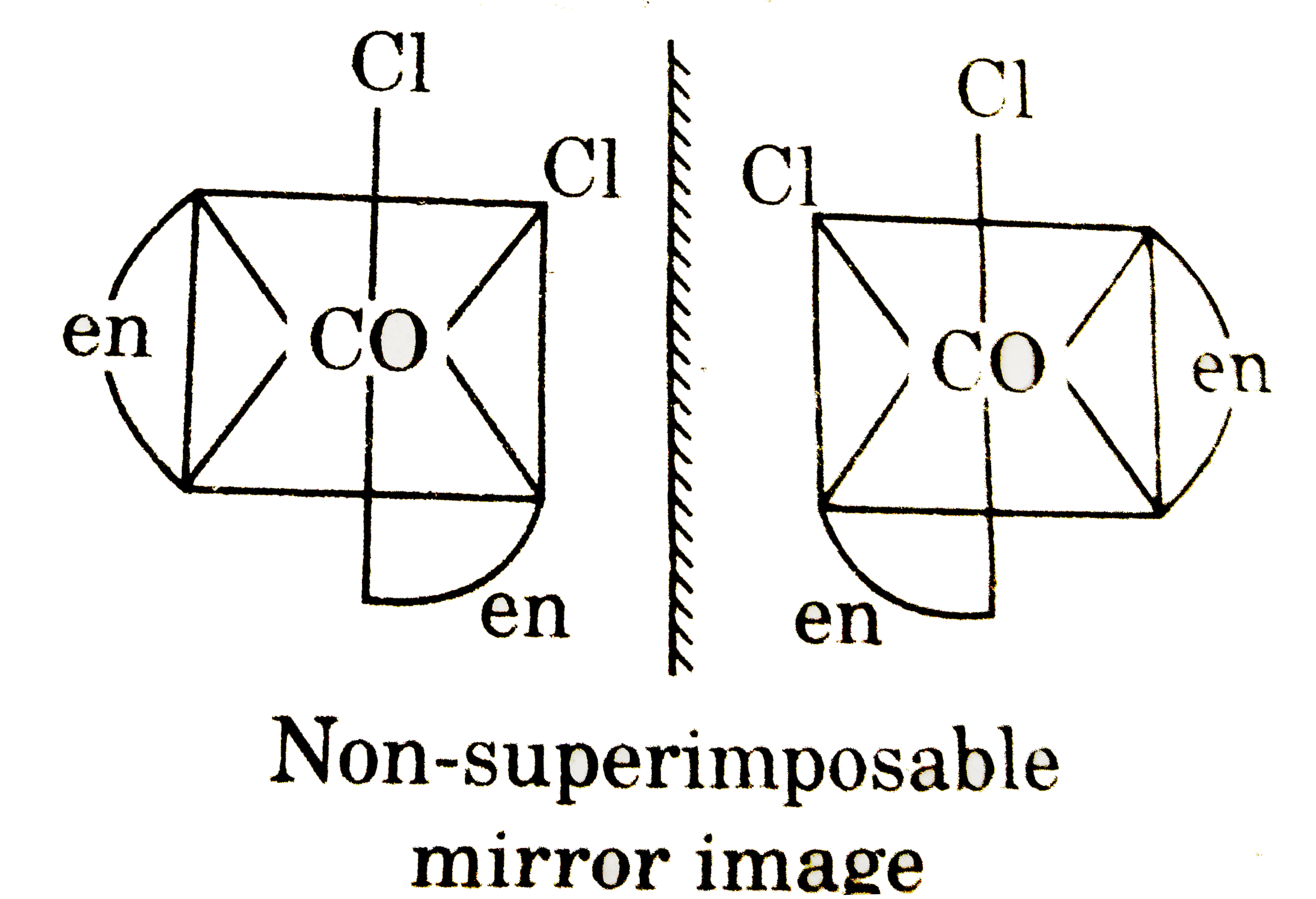 Non-superimposable MIRROR image Non-superimposable MIRROR image
|
|