Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The chloride of a metal 'M' contains 47.23% of the metal. 1 g of this metal displaced from a compound 0.88 g of another metal 'N'. Find the equivalent weight of 'M' and 'N' respectively. |
| Answer» SOLUTION :31.77, 27.96 | |
| 2. |
The chloride of a solid metallic element contains 57.89% by mass of the element. The specific heat of the element is 0.0324 cal "deg"^(-1)g^(-1). Calculate the exact atomic mass of the element. |
|
Answer» Solution :Equivalent mass of the ELEMENT `=(6.4)/(42.11)xx35.5=48.8` APPROXIMATE ATOMIC mass `=(6.4)/(0.0324)=200` Valency `=(200)/(48.8)=4` Exact atomic mass `=48.8xx4=195.2` |
|
| 3. |
The chloride of a metal has the formula MCl_(3). The formula its phosphate is: |
|
Answer» `M_(2)PO_(4)` |
|
| 4. |
The chloride ion is isoelectronic with potassium. The size of chloride ion is: |
|
Answer» LARGER than `K^+` ion |
|
| 5. |
The chloride ion is isoelectric with potassium. The size of chloride ion is |
|
Answer» Same as that of `K^(+)` ion |
|
| 6. |
The chloric acid and chlorates are: |
|
Answer» GOOD OXIDISING agents |
|
| 7. |
The chlorate ion can disproportinate in basic according to reaction 2ClO_3^(-) hArr ClO_2^(-) +ClO_4^(-) calculate the value of equilibrium constant. Given E_(ClO_4^(-)//ClO_3^(-))^@=0.36 V and E_(ClO_3^(-)//ClO_2^(-))^@ =0.33 V at 298 K : - |
|
Answer» 0.1 =0.33-0.36=-0.03 V `E_"cell"^@=0.06/n ` LOG K -0.03 = `0.06/2` log K `K=10^(-1)` = 0.1 M |
|
| 8. |
The chloric acid and chlorates are : |
|
Answer» Good oxidizing agents |
|
| 9. |
The chlorate ion can disproportinate in basic accordin to reaction 2ClO_(3)^(-)hArr ClO_(2)^(-)+ClO_(4)^(-) calculate the value of equilibrium constant. Given E_(clO_(4)^(-)//ClO_(3)^(-))^(O)=0.36v and E_(ClO_(3)^(-)//ClO_(2)^(-))^(O)=0.33V at298K |
|
Answer» SOLUTION :`E_("CELL")^(O)=E_(ClO_(3)^(-)//Clo_(3)^(-))^(O)+E_(ClO_(3)^(-)//ClO_(4)^('))^(O)` `=0.33-0.36=-0.03V` `E_("cell")^(O)=0.06/n log K` `=-0.03=0.06/2log K` `K=10^(-1)=0.1M` |
|
| 10. |
The chirality of the compound |
|
Answer» R |
|
| 11. |
The chiral alkane of the lowest molecular mass not containing a ring and isotopes is |
|
Answer» `CH_3- cH_2 - UNDERSET(CH_3) underset(|)(CH) -CH_2 -CH_2 -CH_3` |
|
| 12. |
The chief source of vitamin D is |
| Answer» Answer :A | |
| 13. |
The chief reaction(s) occurring in blast furnace during extraction of iron from haematite is / are : |
|
Answer» `Fe_(2)O_(3)+ 3CO to2Fe+3CO_(2)` |
|
| 14. |
The chief source of iodine in which it is present as sodium iodate is |
|
Answer» Sea weeds |
|
| 16. |
The chief impurity present in bauxite is |
|
Answer» `SiO_(2)` |
|
| 17. |
The chief constituents of biological membranes are: |
|
Answer» Proteins |
|
| 18. |
The chief constituent s of German Silver alloy are : |
| Answer» Answer :A | |
| 20. |
The chief component of cement that has property of setting quickly and acquiring considerable strength within a few days is |
|
Answer» Tricalcium SILICATE, `3CaO.siO_(2)` |
|
| 21. |
The chemistry of zero group elements is little known because |
|
Answer» They are LESS abundant |
|
| 22. |
The chemistry of the actionoid elements is not so smooth as that of the lanthanoids. Justify this statements by giving some examples from the oxidation state of these elements. |
| Answer» Solution :Lanthanoids SHOW a limited number of oxidation state, viz., +2, +3 and +4 (out of which +3 is the most common). This is because of a large energy GAP between 4f-, 5D- and 6s- subshells. The DOMINANT oxidation state of actinoids is also +3 but they show a number of other oxidation states also, Uranium (Z = 92) and plutonium (Z = 94), show +3, +4, +5 and +6, neptunium (Z = 93) shows +3, +4, +5 and +7, etc. This is due to small energy difference between 5-, 6d- and 7s-subshells of the actinoids. | |
| 23. |
The chemistry of the actionid elements is not so smooth as that of the lanthaoids. Justify this statements by giving some examplefrom the oxidation state of these elements. |
| Answer» Solution :Lanthanoids show limitednumber of OXIDATION state, viz., `+2, +3, ` and `+4` ( out of which `+3` is most common.) This is because of LARGE energy gap between 4f and 5 d subshells. The dominant oxidation state of ACTINOIDS is also `+3` but they show a number of ther oidation states also, e.g., uranium `(Z = 92)` and PLUTONIUM `( Z= 94)` , show `+ 3, + 4, + 5 ` and `+6` , nuptunium `(Z = 94)` shows `+ 3, +4 , +5` and `+7` etc. This is due to small energy difference between 5f , 6d and 7s subshells of the actinoids. | |
| 24. |
The chemistry of the actinoid elements is not so smooth as that of the lanthanoids. Justify this statement by giving some examples from the oxidation state of these elements. |
| Answer» Solution :Lanthanoids show (+2), (+3) and (+4) oxidation states. HOWEVER (+3) oxidation state is most dominant. Actinoid SHOWS oxidation states ranging from (+3) to (+7). The LARGE RANGE of oxidation states of actinoides as compared to lanthanoids is because of comparable energies of 5f, 6d and 7s orbitals. For example, the maximum oxidation state increases from (+4) in Th to (+5), (+6) and (+7) respectively in Pa, U and Np but decreases in succeeding ELEMENTS. However (+3) and (+4) ions tend to hydrolyze. Hence, it is unsatisfactory to review the chemistry of actinoids in terms on oxidation states. | |
| 25. |
The chemistry of corrosion of iron is essentially an electrochemical phenomenon. Explain the reactions occurring during the corrosion of iron in the atmosphere. |
Answer» Solution :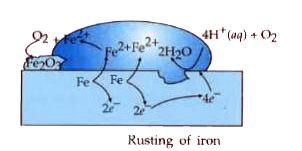 REACTIONS occuring during the corrosion: `{:("Oxidation", Fe(s) to Fe^(2+)(AQ) + 2e^(-)),("Reduction", O_(2)(g) + 4H^(+) (aq) + 4e^(-) to 2H_(2)O(l)):}` Atmospheric oxidation: `2Fe^(2+)(aq) + 2H_(2)O (l) + 1/2O_(2)(g) to Fe_(2)O_(3) (s) + 4H^(+)(aq)` |
|
| 26. |
The chemistry of lithium is very much similar to that of magnesium even though they are placed in different groups. The reason is: |
|
Answer» Both have nearly the same SIZE |
|
| 27. |
The chemicals which carry different messages in the body between neurons and muscles are called_____. |
|
Answer» transmitters |
|
| 28. |
The chemistry of benzene is characterised by whichfo the following types of reaction? |
|
Answer» Additomn |
|
| 29. |
The chemicals used for prevention of impregnation are called ......... prevention. |
|
Answer» ANTIFERTILITY drugs |
|
| 30. |
The chemical used in mental disease is |
|
Answer» ANTIPYRETICS |
|
| 32. |
The chemical used in Holme's signals are |
|
Answer» Calcium NITRIDE and calcium CARBIDE |
|
| 33. |
The chemical used for cooling in refrigeration is |
|
Answer» `CO_(2)` |
|
| 34. |
The chemical used for cooling in refrigenaration or in manufacture of ice is: |
|
Answer» `CS_2` |
|
| 35. |
The chemical that undergoes self-oxidation and self-reduction in the same reaction |
|
Answer» BENZYL alcohol |
|
| 36. |
The chemical treatment of the ore for concentration is done in the case of |
|
Answer» aluminium Al ore bauxite is leached with NAOH. Au, Ag leached with NaCN. For .Cu ore `(CuFeS_(2))` concentration done by froth FLOTATION. It is a physical method |
|
| 37. |
The chemical that initiates electrolysis in Halls process is |
|
Answer» `Na_(3)AlF_(6)` `AlF_(3) hArr Al^(3+) + 3F^(-)` `Al_(2)O_(3) + 6F_(2) to 4AlF_(3)+……………….` |
|
| 38. |
The chemical substances used to lower temperature of the body are called : |
|
Answer» ANTIBIOTICS |
|
| 39. |
The chemical substances produced by living organisms to inhibit the growth or to kill microorganisms are called |
|
Answer» ANTIBIOTICS |
|
| 40. |
The chemical substances that raise the pH value of stomach are called as _______. |
|
Answer» antacids |
|
| 41. |
The chemical substance which is used for disnfectants and presrvationof food is |
|
Answer» `SO_(2)` |
|
| 42. |
The chemical substance which are used to prevent decompsition is/are |
|
Answer» antacids |
|
| 43. |
The chemical reactivity of ethylene is due to: |
|
Answer» Shot carbon to carbon BOND distance |
|
| 44. |
The chemical reagent used for leaching of gold and silver ores is |
|
Answer» SODIUM HYDROXIDE |
|
| 45. |
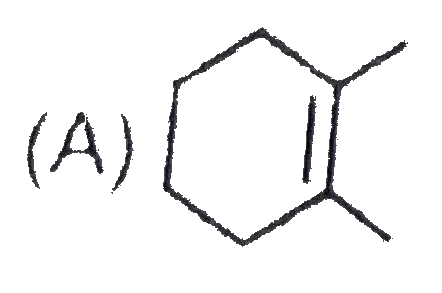
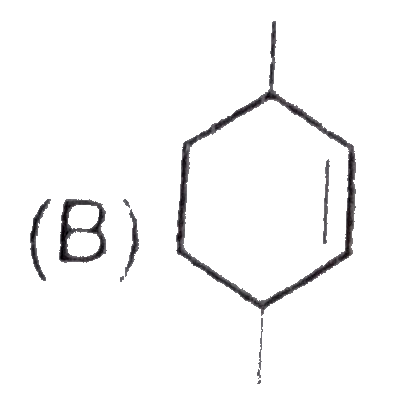
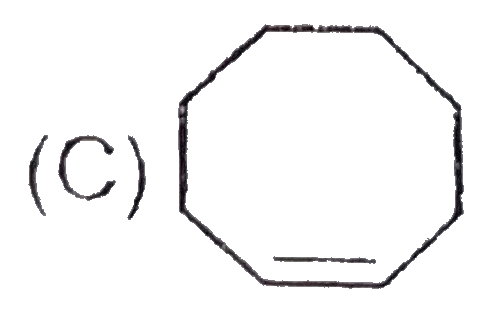
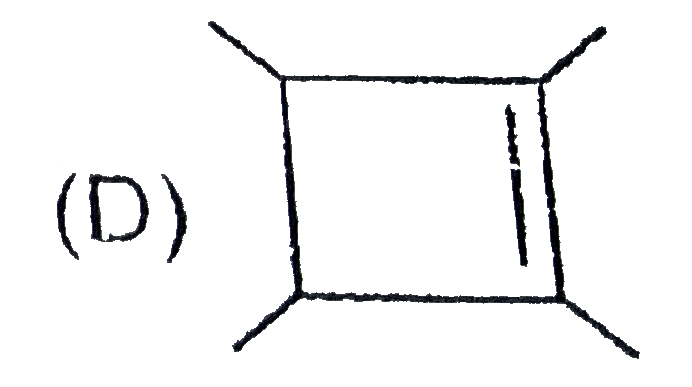
The chemical reactions of an unsaturated compound 'M' are given below. Determine the possible structural formula of 'M' |
|
Answer»
|
|
| 46. |
The chemical reactions in which reactants require high amount of activation energy are genera11y |
|
Answer» SLOW |
|
| 47. |
The chemical reactions in which the reactants require high amount of activation energy are generally |
|
Answer» slow |
|
| 48. |
The chemical reactions in which reactants high amount of activation energy are generally |
|
Answer» SLOW |
|
| 49. |
The chemical reaction of K_(2)Cr_(2) O_(7) with following: (i) H_(2)SO_(4) "" (ii) KOH "" (iii) KI. (b) What is chromly chloride test ? |
|
Answer» Solution :`2K_(2)Cr_(2)O_(7)+8H_(2)SO_(4) overset(DELTA)to2K_(2)SO_(4) +2Cr_(2)(SO_(4))_(3)+8H_(2)O+3O_(2)` `K_(2)Cr_(2)O_(7)+2KOH) to 2K_(2)CrO_(4)+H_(2)O` (III) `K_(2)Cr_(2)O_(7)+7H_(2)SO_(2)+5HI to 4K_(2)SO_(4)+cr_(2)(SO_(4))_(3)+7I_(2)+7H_(2)O` (b) When heated with CONC. HC or with a soluble chloride and conc. H2SO4 reddish brown vapour of chromyl chloride are produced. `K_(2)Cr_(2)O_(7)+4KCl+ underset((Conc))(6H_(2)SO_(4)) overset(Delta)to underset("Chromyl Chloride")(2CrO_(2)Cl_(2))+6KSHO_(4)+3H_(2)O` This reaction is used as a test for soluble chlorides and is called chromyl chloride test. |
|