Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The brown ring formed in the test of nitrates with FeSO_(4) solution and concentrated H_(2)SO_(4) has the formula ….. . |
| Answer» SOLUTION :`[FE(H_(2)O)_(5)NO]^(2+)` | |
| 2. |
The brown ring less test is performed for the qualitative detection of |
|
Answer» Bromides |
|
| 3. |
The brown ring complex is formulated as [Fe(H_2O)_(5)NO^(+)]SO_4The Oxidation state of Fe is X what is the value of X? |
|
Answer» |
|
| 4. |
The brown ring complex compound is formulated as [Fe(H_2O)_5(NO)]SO_4. The oxidation state of iron is: |
| Answer» ANSWER :A | |
| 5. |
The brown ring complex compound is formulated as [Fe(H_(2)O)_(5)(NO)]SO_(4). The oxidation state of iron is : |
| Answer» Answer :B | |
| 6. |
The brown ring complex compound is formulated as [Fe(H_2O)_5NO]SO-4 The oxidation states of iron is |
|
Answer» `+1` |
|
| 7. |
The brown ring complex compound is formu = lated as [Fe(H_(2)O)_(5)NO]SO_(4). Theoxidation state of iron is : |
| Answer» Answer :B | |
| 8. |
The brown righ test for NO_(2)^(-) and NO_(3)^(-) is due to the formation of complex ion with a formula |
|
Answer» `[FE(H_(2)O)_(6)]^(2+)` |
|
| 9. |
The brown compound formed in the ring test for nitrates contains the ion |
|
Answer» `[Fe(H_(2)O)_(5)NO]^(3+)` |
|
| 10. |
The brown precipitate formed in step C consists of |
|
Answer» `HG(NH_(2)I` |
|
| 11. |
The brown gas formed when HNO_(3) is reduced by metals is |
|
Answer» `N_(2)O` |
|
| 12. |
The brown complex obtained in the detection of nitrate radical is formulated as [Fe(H_2 O)_5 NO]SO_4. What is the oxidation number of Fe in this complex? |
|
Answer» `+1` `[Fe(H_2 O)_5 NO [ SO_4]or [Fe(H_2O)_5NO]^(2+) SO_(4)^(2-)` thecolourof thecomplexis dueto changetransfer. Thiscomplexformallycontains`Fe(+1)`and `NO^(+)`sinceFe (II)gainsone electron fromNOwhichchangesto `NO^(+)` |
|
| 13. |
The brown colour of ring in NO_(3)^(-) test is of |
|
Answer» `FeSO_(4)` |
|
| 14. |
The Bronsted acids in the reversible reaction are HCO_(3)^(-)(aq.) + OH^(-) (aq.) CO_(3)^(2-)(aq.) + H_(2)O |
|
Answer» `OH^(-)` and `CO_(3)^(2-)` |
|
| 15. |
The bronstad acid which gives the weakest conjugate base is : |
|
Answer» HF |
|
| 16. |
The bromination of anisole is carried out in the presence of……. |
| Answer» Solution :`CH_(3)COOH` | |
| 17. |
The bromination of aniline produces |
|
Answer» 2- bromoaniline 
|
|
| 18. |
The bromination of acetone which occurs in acid solution is represented by the equation: CH_(3)COCH_(3)(aq) + Br_(2)(aq) to CH_(3)COCH_(2)Br(aq) + H^(+)(aq) + Br^(-1)(aq) The following kinetic data was obtained for the given reaction concentration: Initial rates of disappearance of of Br_(2)(Ms^(-1) 5.7 xx 10^(-5), 5.7 xx 10^(-5) 1.2 xx 10^(-4), 3.1 xx 10^(-4) Based on these data, the rate equation is: |
|
Answer» RATE = `k[CH_(3)COCH_(3)][Br_(2)][H^(+)]^(2)` `k[CH_(3)COCH_(3)][H^(+)]` |
|
| 19. |
The bromination of acetone that occurs in acid solution is represented by this equation. CH_(3)COCH_(3)Br(aq)+Br_(2)(aq) to CH_(3)COCH_(2)Br(aq)+H^(+)(aq)+Br^(-)(aq) These kinetic data were obtained for given reaction concentrations. Initial concentrations, M {:([CH_(3)COCH_(3)],[Br_(2)],[H^(+)],"Initial rate, disap"),(,,,"pearance of " Br_(2)),(0.30,0.05,0.05,5.7xx10^(-5)),(0.30,0.10,0.05,5.7xx10^(-5)),(0.30,0.10,0.10,1.2xx10^(-4)),(0.40,0.05,0.20,3.1xx10^(-4)):} Based on these data, the rate equation is |
|
Answer» `"RATE"=k[CH_(3)COCH_(3)][Br_(2)]` |
|
| 20. |
The bromination of acetone that occurs in acid solution is represented by this equation CH_(3)COCH_(3) (aq) + Br_(2) (aq) to CH_(3) COCH_(2) Br(aq) + H^(+) (aq) + Br^(-) (aq) These kinetic data were obtained for given reaction concentrations .Initial concentrations , M {:([CH_(3)COCH_(3)] ,, [Br_(2)] ,, [H^(+)]) , (0.30 ,, 0.05 ,, 0.05) , (0.30 ,, 0.10 ,, 0.05) , (0.30 ,, 0.10 ,, 0.10) , (0.40 ,, 0.05 ,, 0.20):} Initial rate , disappearance of Br_(2), Ms^(-1) |
|
Answer» `5.7 xx 10^(-5)` |
|
| 21. |
The bromination of acetone that occurs in acid solution is represented by CH_(3)COCH_(3)(aq.)+Br_(2)(aq.)to CH_(3)COCH_(2)Br(aq.)+H^(+)(aq.)+Br^(-)(aq.) These kinetic data were obtained for given reaction concentration Initial concentration {:("Initial concentration",,,"Initial rate of"),(,,,"disappearance"),([CH_(3)COCH_(3)],[Br_(2)],[H^(+)],"of "Br_(2)),(0.30,0.05,0.05,5.0xx10^(-5)),(0.30,0.10,0.5,5.0xx10^(-5)),(0.30,0.10,0.10,1.0xx10^(-4)),(0.90,0.05,0.10,3.0xx10^(-4)):} Based on these data, order of reaction will be - |
|
Answer» `[CH_(3)COCH_(3)]` and `[H^(+)]` = constant `[Br_(2)]` = doubled RATE = UNCHANGED `therefore` Order w.r.t `Br_(2) = 0` POSSIBLE rate LAW (By option) `r = k [CH_(3)COCH_(3)][H^(+)]` |
|
| 22. |
The broad spectrum antibiotic among the following is : |
|
Answer» Chloramphenicol |
|
| 23. |
The B.P. of alcohols are much higher than the ethers of comparable molecular masses due to |
|
Answer» interamolecular H - BONDING |
|
| 24. |
The branch of chemistry which with the reaction rates and reaction mechanism is called |
|
Answer» THERMOCHEMISTRY |
|
| 25. |
The branch of chemistry which deals with the reaction rates and. reaction mechanism is called : |
|
Answer» THERMOCHEMISTRY |
|
| 26. |
The b.p. of a solution of 5g of sulphur in 100g of carbon disulphide is 0.476^(@) above that of pure solvent. Determine the molecular formula of sulphur in this solvent. The b.p. of pure carbon disulphide is 46.30^(@)C and its heat of vaporisation is 84.1 calories per gram. |
|
Answer» Solution :We have, `K_(b)=(RT^(2))/(1000l_(v))=(2xx(46.30+272)^(2))/(1000xx84.1)=2.425` `:.m=(DeltaT_(b))/(K_(b))=(0.476)/(2.425)` And also `m=("moles of solute")/("wt.of solvent in grams")xx1000` `=(5)/(M)xx(1000)/(100)=(50)/(M)` where M is the mol. wt. of SULPHUR Thus, `(50)/(M)=(0.476)/(2.425)`, `M=255` Since the atomic wt. of sulphur is 32 and its mol.wt. is `255`, therefore, number of sulphur atoms associated to form a SINGLE molecule is `(255)/(32)=7.96` , i.e., 8. Hence the formula is `S_(8)`. |
|
| 27. |
The Bouveault-Blanc reduction involves |
|
Answer» `C_(2)H_(5)OH//Na` |
|
| 28. |
The Born -Haber cycle for formation of rubidium chloride (RbCl) is given below (the enthalpies are in K Cal mol"^(-1) ) Find the value of X. |
|
Answer» `x=-90.75" KCAL MOL"^(-1)` |
|
| 29. |
The borax bead is chemically : |
|
Answer» `B_2O_3 +NaBO_2` |
|
| 30. |
The boron, carbon, nitrogen and oxygen have their increasing ionisation enthalpy in the order |
|
Answer» N,O,B,C |
|
| 31. |
Thebonds presentin thestatemetn is/aretrue? |
|
Answer» FOUR equivalentCr-O BONDS only.  Six equivlent Cr-O bonds and one Cr-O-Cr bond. |
|
| 32. |
The bonds present inN _2O_5 are : |
|
Answer» Only ionic |
|
| 33. |
The bonds present in the structure of dichromate ion are |
|
Answer» four equivalent CR - O bonds only 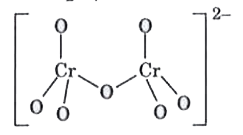 There ar six equivalent Cr-O bonds and one Cr-O-Cr bond. |
|
| 34. |
The bonds present in N_(2)O_(4) are |
|
Answer» only IONIC |
|
| 35. |
The bonds present in N_(2)O_(5) are : |
|
Answer» Only IONIC |
|
| 36. |
The bonds is K_(4)[Fe(CN)_(6)] are |
|
Answer» all IONIC |
|
| 37. |
The bonds formed by transition metals in lower oxidation states are .......... while those formed in higher oxidation states are ............ |
| Answer» SOLUTION :IONIC, covauent | |
| 38. |
The bond that determines the secondary structure of proteins is ……. . |
|
Answer» COORDINATE bond |
|
| 39. |
The bond that determines the secondary structure of a protein is |
| Answer» Solution : Hydrogen bond. | |
| 40. |
The bond stability of O_(2)^(+),O_(2),O_(2)^(-),O_(2)^(2-) varies in the order |
|
Answer» `O_(2)^(2-)gtO_(2)^(-)gtO_(2)gtO_(2)^(+)` Bond order `prop` STABILITY the bond stability of `O_(2)^(+), O_(2),O_(2)^(-), O_(2)^(2-)` varies in the following order `O_(2)^(+)gtO_(2)gtO_(2)^(-)gtO_(2)^(2-)` |
|
| 41. |
The bond that determines the secondary structure of a protein is : |
|
Answer» COORDINATE bond |
|
| 42. |
The bond (s) persent inquaternary ammonium salt is /are. |
|
Answer» covalent |
|
| 43. |
The bonds present in pernitric acid are: |
|
Answer» IONIC BONDS |
|
| 44. |
The bond order of the underlined species: NOHSO_4 is. |
|
Answer» |
|
| 45. |
The bond order order is maximum in: |
|
Answer» `H_2 ` |
|
| 46. |
The bond order of NO molecule is |
|
Answer» |
|
| 47. |
The bond order of NO is 2.5 while that of NO^(+) is 3. Which of the following statements is true for these two species? |
|
Answer» BOND LENGTH in `NO^(+)` is GREATER than in NO |
|
| 48. |
The bond order of individual carbon-carbon bonds in benzene is |
|
Answer» ONE |
|
| 49. |
The bond order of CO molecule on the basis of molecular orbital theory is : |
|
Answer» Zero |
|