Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The bond order of Cl-O in ClO_(4)^(-) is (x)/( y ) then x-y=? |
Answer»  `:. (X)/( y ) = (7 )/( 4) , x - y = 7-4 = 3` `:. (X)/( y ) = (7 )/( 4) , x - y = 7-4 = 3`
|
|
| 2. |
The bond order of C_2^+ is : |
|
Answer» 1 |
|
| 3. |
The bond strength is maximum in : |
|
Answer» `He_2^(+)` |
|
| 4. |
The bond order in peroxide ion is : |
|
Answer» LARGER than in `O_(2)` molecule |
|
| 6. |
The bond order in NO is 2.5 while that in NO^(+) is 3. Which of the following statements is true for these two species ? |
|
Answer» BOND length in `NO^(+)` is equal to that in NO. |
|
| 7. |
The bond order in O_(2)^(+) is same as in |
| Answer» Answer :A | |
| 9. |
The bond order in N_(2)^(+), O_(2) and F_(2) follows the order : |
|
Answer» 1. `F_(2)lt N_(2)^(+)lt O_(2)` |
|
| 10. |
The bond order in NO^(+) is : |
|
Answer» same as in `O_(2)^(+)` and `N_(2)^(+)` |
|
| 11. |
The bond order between C and C in acetylene molecule is |
|
Answer» 1 |
|
| 12. |
The bond lengths in the species O_(2), O_(2)^(+) and O_(2)^(-) are in the order |
|
Answer» `O_(2)^(+)gtO_(2)gtO_(2)^(-)` B.O. in `O_(2)=(10-6)//2=2.0` B.O in `O_(2)^(-)=(10-7)//2=1.5` |
|
| 13. |
The bond lengths in O_(2), O_(2)^(+) and O_(2)^(-) species follow the order : |
|
Answer» 1. `O_(2)^(-)LT O_(2)lt O_(2)^(+)` |
|
| 14. |
The bond length of the S-O bond is maximum in which of the following compounds? |
|
Answer» `SOCl_(2)` 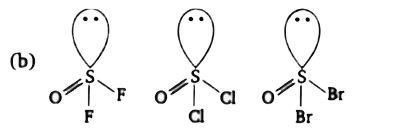 ACCORDING to Bent's Rule. |
|
| 15. |
The bond length is CO is 1.128Å what will be the bond length of CO in Fe(CO)_(5)? |
|
Answer» 1.158Å |
|
| 16. |
The bond length in LiF will be |
|
Answer» less than that of NAF |
|
| 17. |
The bond length in CO is 1.128 Å. What will be the bond length of CO in Fe(CO)_(5) ? |
|
Answer» `1.158 Å` |
|
| 18. |
The bond length between sp^3 hybridised carbon atom and other carbon atom is minimum in |
|
Answer» Propane |
|
| 19. |
The bond length in CO is 1.128 Å. What will be the bond length of CO in Fe(CO)_(5)? |
|
Answer» 1.158Å |
|
| 20. |
The bond length between sphybrid carbon and other carbon is minimum in: |
|
Answer» propane |
|
| 21. |
The bond formed by sidewise overlapping of atomic orbitals is: |
|
Answer» SIGMA BOND |
|
| 22. |
The bond in protein structure, that are not broken in denaturation is |
|
Answer» HYDROGEN bond |
|
| 23. |
The bond having the highest bond energy is : |
|
Answer» `C=C` |
|
| 24. |
The bond in a molecule of HCl is formed due to the overlapping of: |
|
Answer» s-and p-orbitals |
|
| 25. |
The bond enerhies of F_2, Cl_2, Br_2 and I_2 are 155.4, 243.6, 193.2 and 151.2 kJ mol^-1 respectively.The strongest bond is: |
|
Answer» F-F |
|
| 26. |
The bond energy of ann O-H bond is 109 kcal/mol. When 1 mole of water is formed, then |
|
Answer» 1009 kcal is RELEASED `THEREFORE`Energy absorbed in the dissociation of 1 mole of water `(H_(2)O)=2xx109=218`kcal `therefore` Energy released in the formation of 1 mole of water =218 kcal |
|
| 27. |
The bond energy of fluorine is more than that of chlorine. |
| Answer» Solution :The BOND ENERGY of FLUORINE is less than that of CHLORINE. | |
| 28. |
The bond energy (in kcal mol^(-1)) ofa C - C single bond is approximately |
|
Answer» 1 |
|
| 29. |
The bond energy (in Kcal mol^(-)) of a C-C single bond is approximately: |
| Answer» SOLUTION :`E_(C-C)~= 100` Kcal/mole. | |
| 30. |
The bond energy is the energy required to |
|
Answer» dissociate oneemole of the substance |
|
| 31. |
The bond energy for catenation next to carbon is: |
|
Answer» N |
|
| 32. |
The bond energies of C-C,C=C, H-H and C-H bonds are 350 kJ mol^-1, 600 kJmol^-1, 400 kJmol^-1 and 410 kJmol^-1 respectively. The enthalpy of hydrogenation of ethane is _______________. |
| Answer» Answer :A | |
| 33. |
The bond energies of of C - C, C = C, H - H and C - H linkages are 350, 600, 400 and 410 kJ per mol respectively. The heat of hydrogenation of ethylene is |
|
Answer» `- 170 kJ mol^(-1)` or `UNDERSET(underset(H)(|))overset(overset(H)(|))(C) = underset(underset(H)(|))overset(overset(H)(|))(C) + H - H rarr H - underset(underset(H)(|))overset(overset(H)(|))(C) - underset(underset(H)(|))overset(overset(H)(|))(C) - H` `Delta_(r) H = Sum B. E ("REACTANTS) - Sum B. E ("Products")` `= B. E. 4(C - H) + B. E. (C = C) + B. E. (H - H) - B. E. 6 (C - H) - B. E. (C - C)` `= E. (C = C) + B. E. (H - H) - 2B. E.(C - H) - B. E. (C - C)` `= 600 + 400 - 2 xx 410 - 350` `= - 170 kJ` |
|
| 34. |
The bond energies of H - H and I - I bonds are 435 kJ mol^-1 and 150 kJ mol^-1 respectively. If DeltaH_f^o for HI is 26.5 kJ mol^-1 then bond enthalpy of H-I bond is |
|
Answer» 558.5kJ/mol |
|
| 35. |
The bond energies of C-C, C=C, H-H and C-H linkages arev350, 600, 400 and 410 kJ per mol respectively. The heat of hydrogenation of ethylene is: |
|
Answer» `-170 KJ mol^-1` |
|
| 36. |
The bond distance between H-atom in H_(2) and X-atoms in X_(2) is 74 pm and 124 pm respectively. Find distance between H and X in HX electronegativity (EN) of H is 2.1 and X is 3.1 in pauling scale. Use Stevenson-Schomaker formula. [Divide your answer by 10] |
|
Answer» Solution :`d_(H-X)=r_(H)+r_(X)-0.09|r_(X)+r_(H)|` where `r_(H) & r_(X)` PUT in `overset(@)A` unit. `d_(H-X)=37xx10^(-2)+62xx10^(-2)-0.09xx1=0.99` `0.09=0.90 overset(@)A=90pm` `=(90)/(10)=9` |
|
| 37. |
The bond energies ("in KJ mole"^(-1)) of P-H, As-H and N-H respectively ? |
|
Answer» 247, 318 and 389 |
|
| 38. |
The bond dissociation energy "off"_(2) is very low due to |
|
Answer» LOW density |
|
| 39. |
The bond dissociation energy of gaseous H_(2),Cl_(2) and HCl are 104, 58 and 103 "kcal/mole" respectively. Calculate the enthalpy of formation of HCl gas. |
|
Answer» SOLUTION :For the reaction `H_(2)(g)+Cl_(2)(g) to 2HCl(g)` For reactants Bond energy OF1 mole of H-H bond `=104` kcal Bond energy of 1 mole of Cl-Cl bond `=58` kcal For products : Energy of formation of 2 MOLES of H-Cl bond `=-2xx103` kcal Thus `DeltaH` of the above reaction `=104+58-206=-44` kcal SINCE, for `H_(2)+Cl_(2) to 2HCl` , `DeltaH=-44kcal` then for, `(1)/(2)H_(2)+(1)/(2)Cl_(2) to HCl` , `DeltaH=-22kcal` |
|
| 40. |
The bond dissociation energy of C-H in CH_4 from the equation C(g) + 4H(g) to CH_4(g), DeltaH = -397.8 kcal is : |
|
Answer» `+ 99.45 KCAL` |
|
| 41. |
The bond dissociation energy of B–F in BF_3 is 646 kJ mol^(–1) whereas that of C-F in CF_4 is 515 kJ mol^(–1). The correct reason for higher B-F bond dissociation energy ascompared to that of C-F is:- |
|
Answer» Significant `ppi-ppi` interaction between B and F in `BF_3` whereas there is no possibility of such interaction between C and F in `CF_4`. |
|
| 42. |
The bond dissociation energy needed to form benzyl radical from toluene is …. Than the formation of the methyl radical from methane |
|
Answer» Less |
|
| 43. |
The bond dissociation energy depends upon the nature of the bond and nature of the molecule. If any molecule more than 1 bonds of similar nature are present then the bond energy reported is the average bond energy. Heat evolved when 0.75 mol of molten aluminium at its melting point of 658^(@)C is solidified and cooled to 25^(@)C. The enthalpy of fusion of aluminium is 76.8 "cal" g^(-1) and C_(P)= 5.8 "cal" mol^(-1) ""^(0)C |
|
Answer» `-4.3` KCAL |
|
| 44. |
The bond dissociation energies of X_(2), Y_(2) and XY are in the ratio of 1 : 0.5 : 1, DeltaH for the formation of XY is - 200 kJ mol^(-1). The bond dissociation energy of X_(2) will be |
|
Answer» 200 KJ `mol^(-1)` `(1)/(2)x_(2)+(1)/(2)y_(2)rarrxy, DeltaH=-200 kJ mol^(-1)` `DeltaH=-200=Sigma(B.E)_("REACTANT")-Sigma(B.E)_("PRODUCT")` `=[(1)/(2)XX(x)+(1)/(2)xx(0.5x)]-[1xx(x)]` `"B.E. of "X_(2)=x=800 kJ mol^(-1)`. |
|
| 45. |
the bond dissociation energies of gasseous H_(2) , Cl_(2) and HCl are 104 .58and 103 kcal respectively . The enthaply of formation of HCl gas would be |
|
Answer» `-44 kcal` `DeltaH=[1/2RE(H_(2))+1/2BE(Cl_(2))]-BE(HCL)` `[(1/2xx104)+(1/2xx58)]-103=-22kcal` |
|
| 46. |
The bond dissociatin energy of C-H bond in CH_4 from the equation, C(g)+4H(g)rarrCH_4, triangleH=-397.8kcal is: |
|
Answer» `+397.8 KCAL` |
|
| 47. |
The bond Br-Cl is: |
|
Answer» POLAR |
|
| 48. |
The bond cleavages during esterification reaction between (A) =CH_3 COOH and (B) C_2 H_5OH |
|
Answer» `C-O " in "" B and O- H " "in " A ` |
|
| 49. |
The bond between the adsorbate and adsorbent in chemisorption is |
|
Answer» Ionic BOND |
|