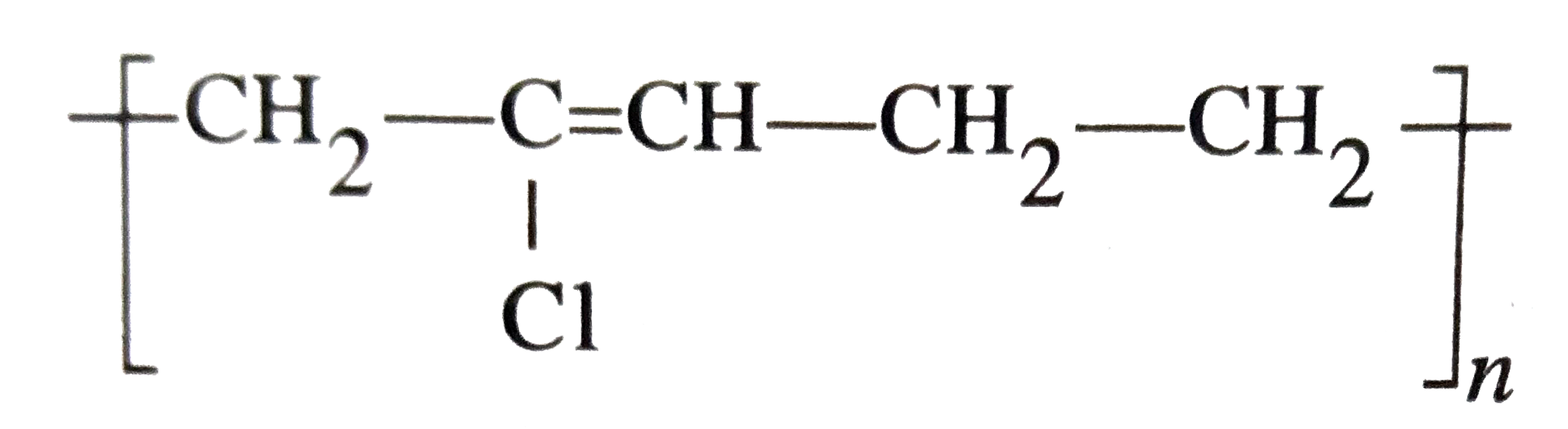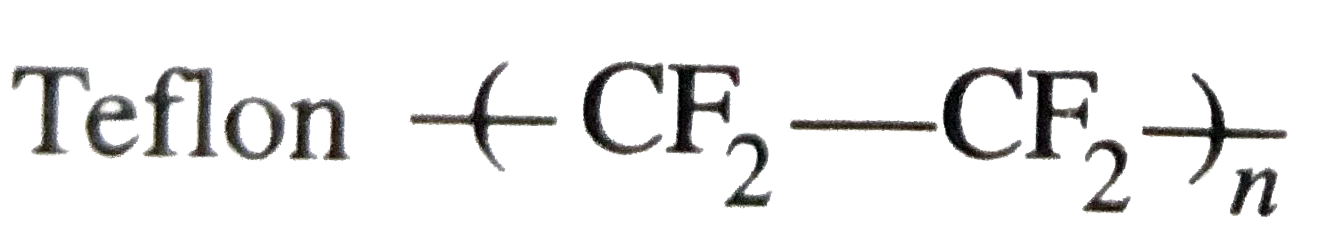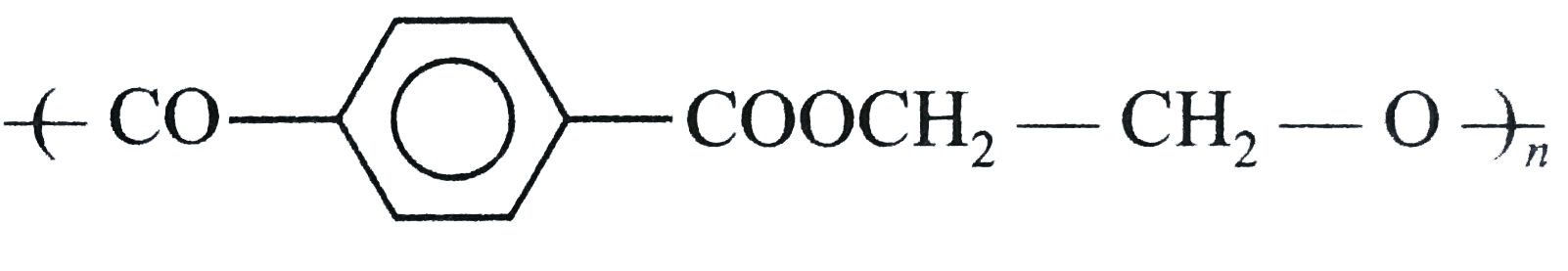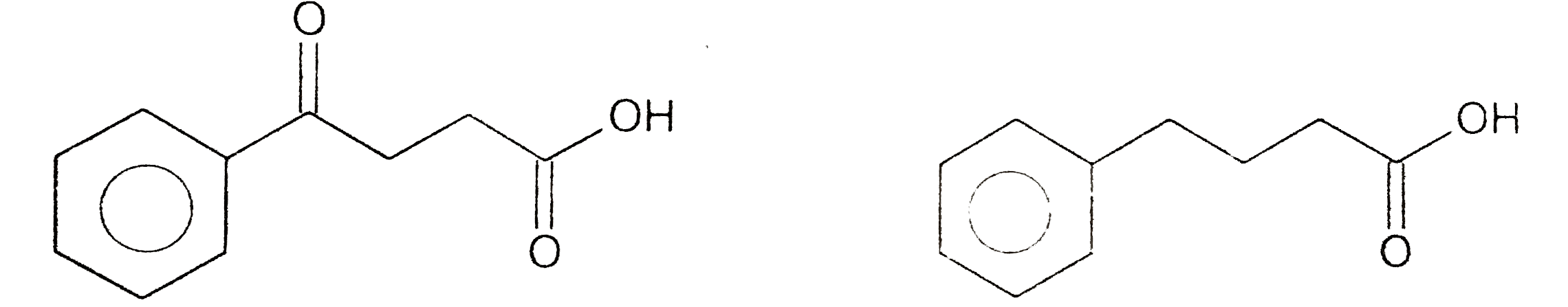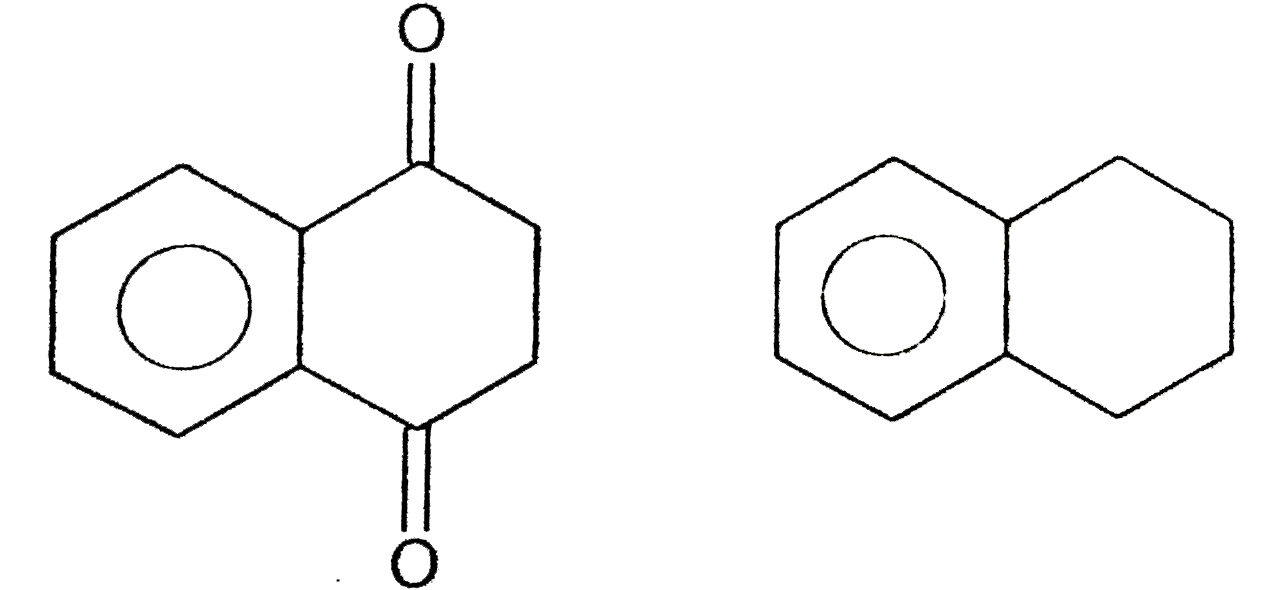Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 2. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 3. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 4. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 5. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 6. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
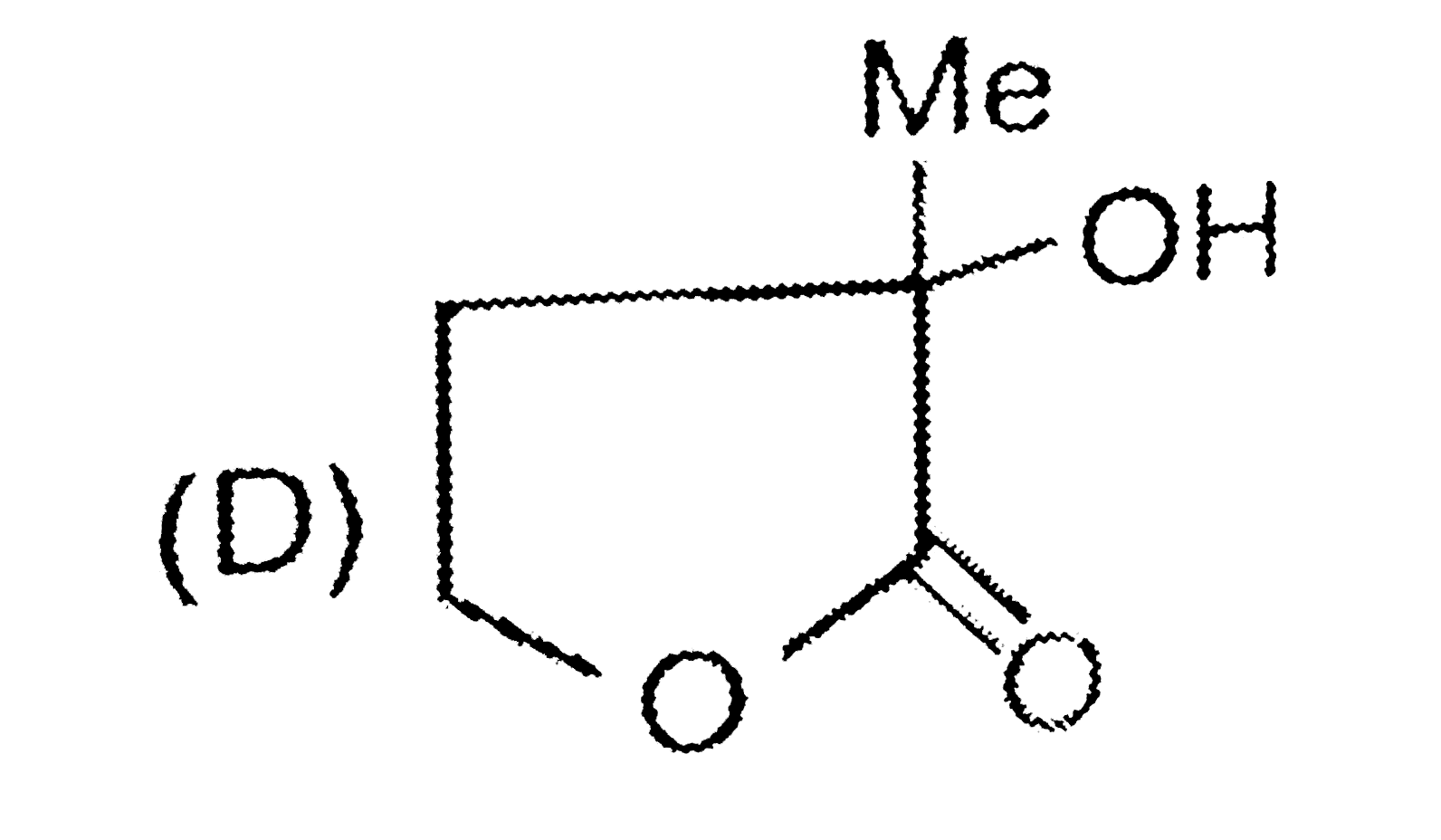
| 7. |
Strutures of products , Starting materials or reagents |
| Answer» SOLUTION :`UNDERSET("ADIPIC ACID")("HEXANE 1,6-dioic acid")` | |
| 8. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 9. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 10. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 11. |
Structures of some common polymers are given. Which one is not correctly represented ? |
|
Answer»
i.e., `-CH_(2)-C(Cl) = CH - CH_(2)-` and not five, i.e., `-CH_(2)-C(Cl) = CH - CH_(2)-CH_(2)-`. |
|
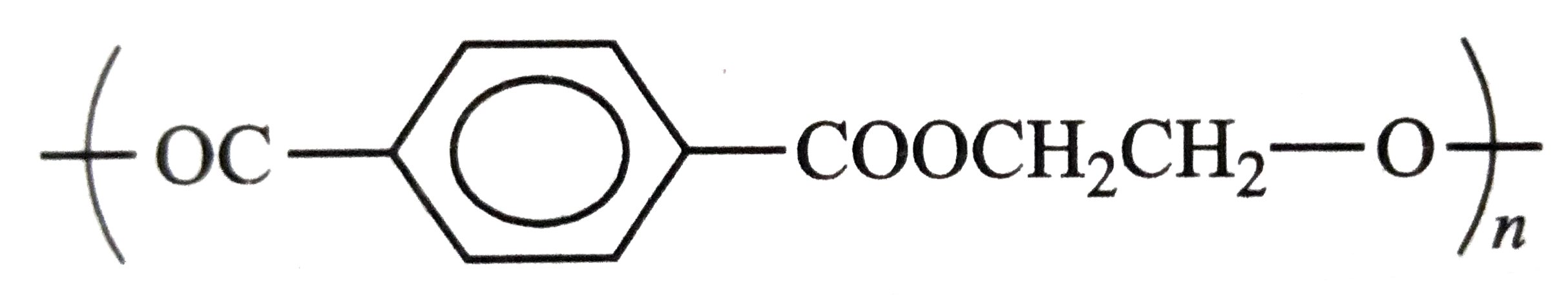
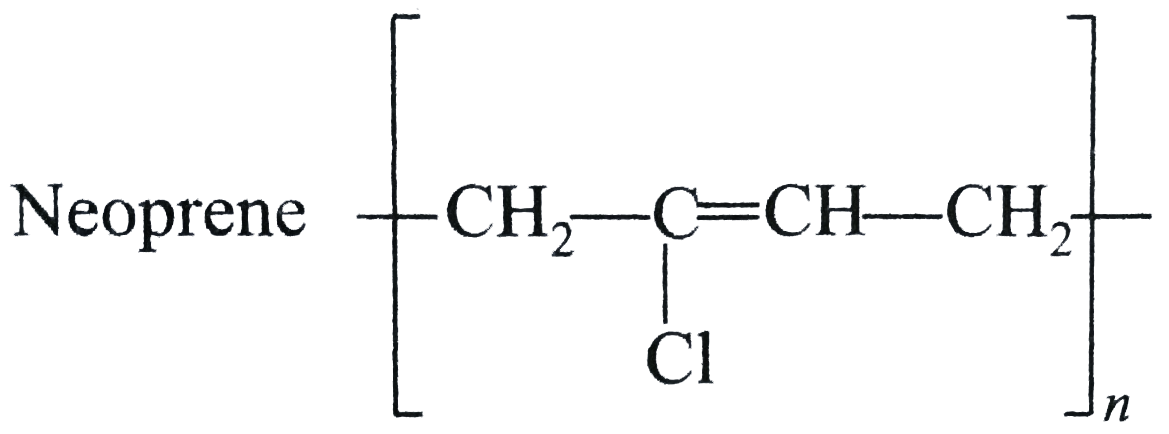
| 12. |
Structures of some common polymers are given. Which one is not correctly presented ? |
|
Answer» NYLON`-6,6-{NH(CH_(2))_(6)NHCO(CH_(2))_(4)-CO-}_(n)` 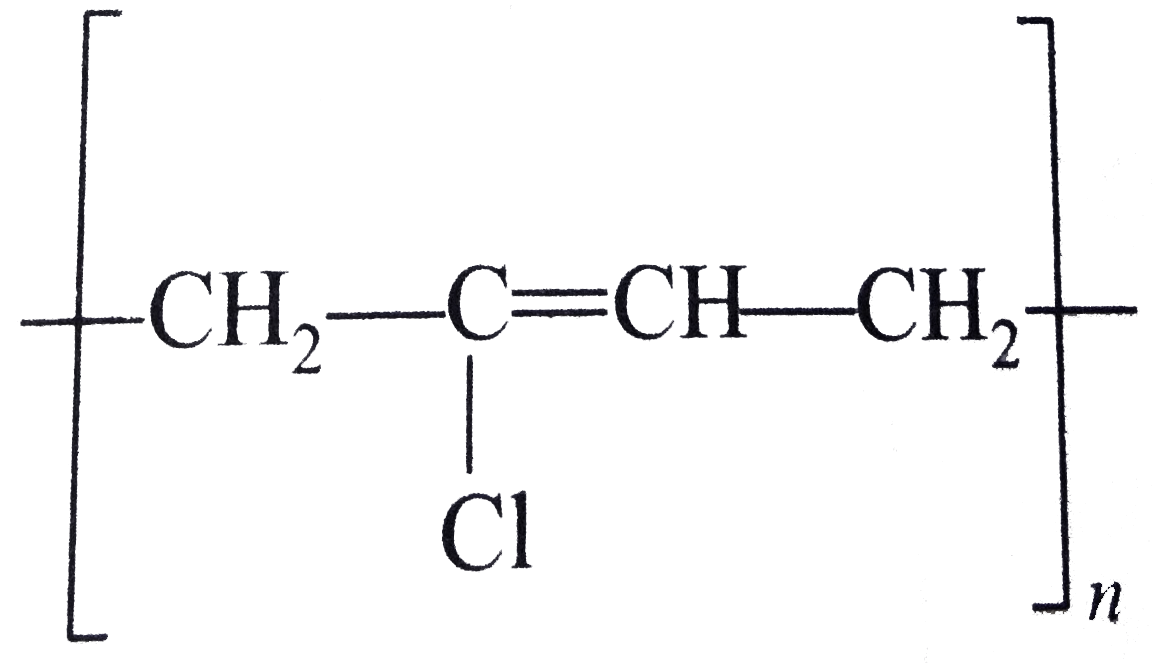 is incorrect is incorrect But neoprene  is CORRECT. is CORRECT.
|
|
| 13. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|
| 14. |
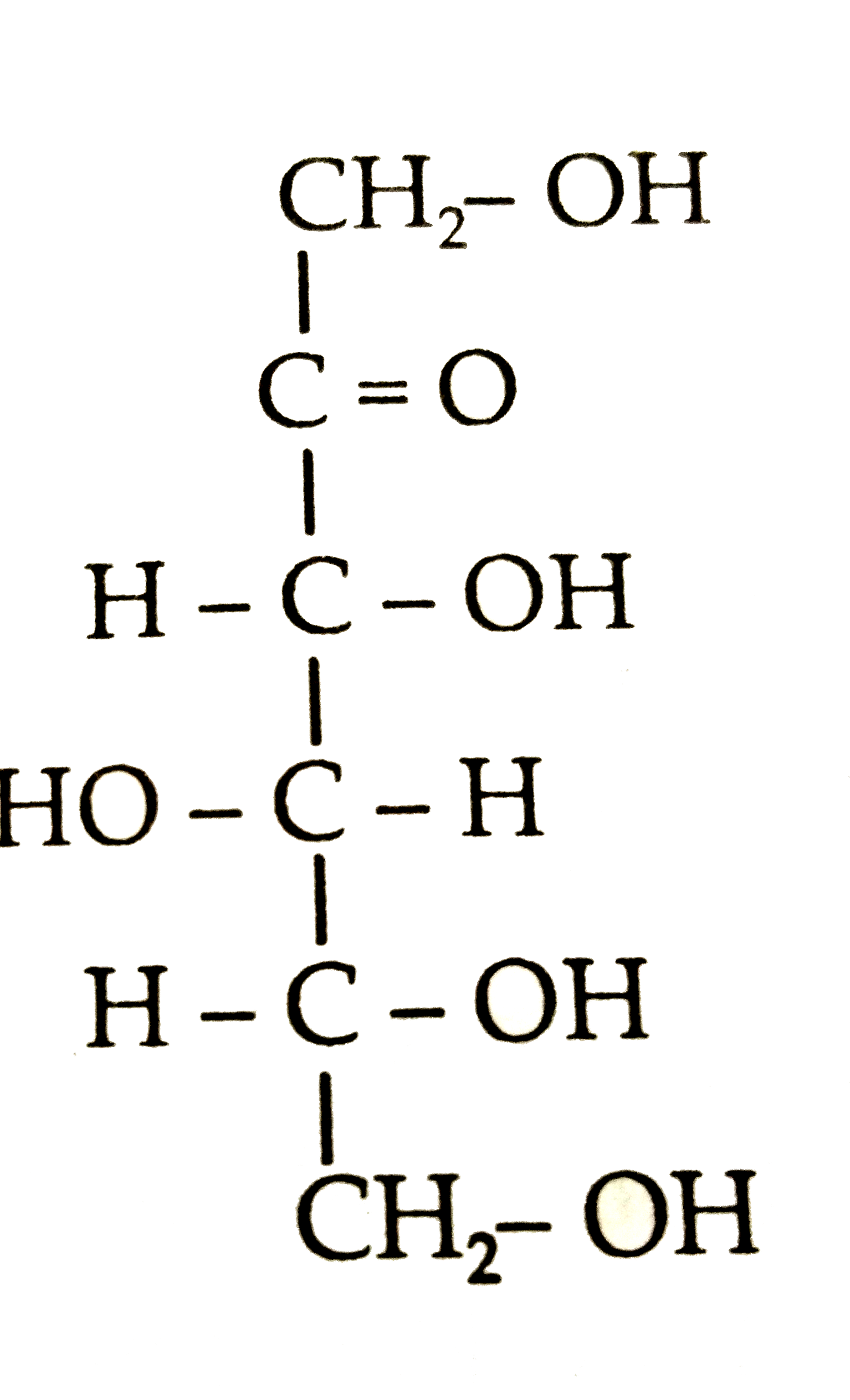
Structures of glycineandalanineare givenbelow. Show the peptidelinkagein glycylalanine . H_(2)N - underset("Glycine")(CH_(2))-COOH"" H_(2)N - underset("Alanine")underset(CH_(3))underset(|)(CH_(2)) - COOH |
Answer» SOLUTION :In glycylalanine , CARBOXYL group COMBINES with theamino group of ALANINE. 
|
|
| 15. |
Struchures of some common polymers are given. Which one is not correctly represented? |
|
Answer» Teflon `(--CF_(2)-CF_(2)--)_(n)` |
|
| 16. |
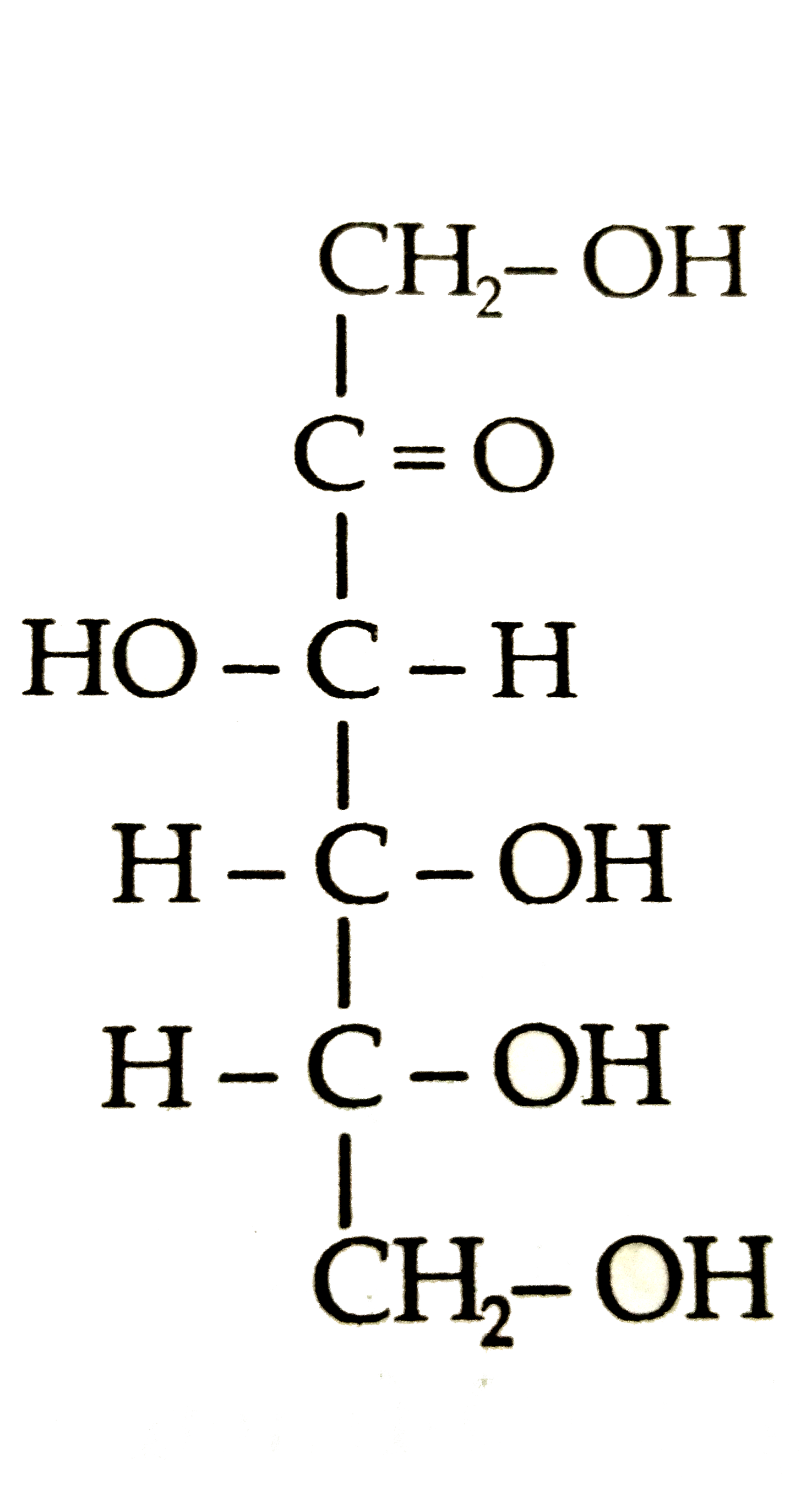
Structures of glycine and alanine are give below , Show the peptide linkage in glycylalanine. H_(2)-underset("Glycine")(CH_(2))-COOH, H_(2)N-underset("Alnanie")underset(CH_3)underset(|)(CH)-COOH |
Answer» Solution :In glycylalanine , the carbonyl group of glycine COMBINES with the AMINO group of ALANINE to form a peptide BOND `(-CO-NH-)` between them as shown below : 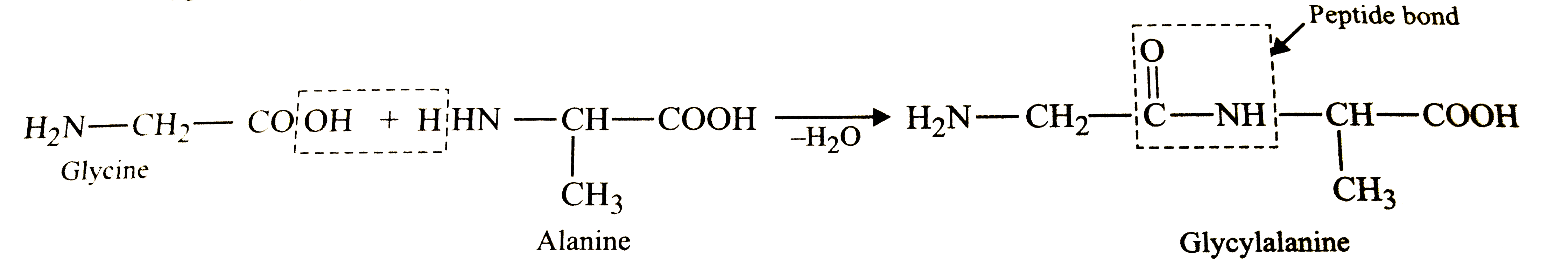
|
|
| 17. |
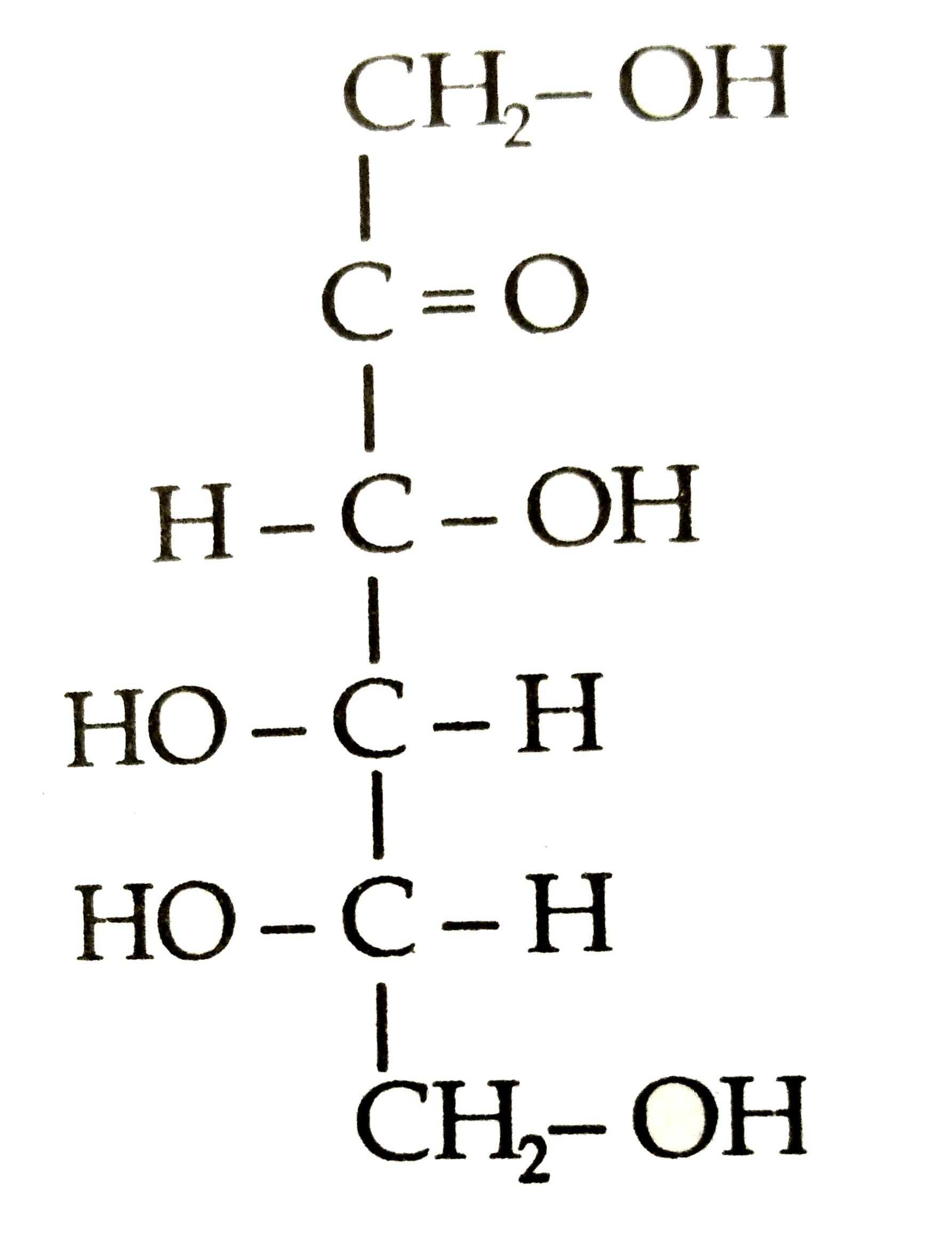
Structures of glycine and alanine are given below. Show the peptide linkage in glycylalanine. |
|
Answer» Solution :`underset("(Glycine)")(H_(2)N-CH_(2)-COOH):underset("(Alanine)")(H_(2)N-underset(CH_(3))underset("|")"CH"-COOH)` In glycylalanine, the carboxyl GROUP of glycine combines with the amino group of alanine. 
|
|
| 18. |
Structure that gives the sequence of amino acids of a protein |
|
Answer» Primary |
|
| 19. |
Structure of Xenon fluoride cannot be explained by valence bond approach. Why ? |
| Answer» Solution :DUE to FULLY filled OCTET of Xe. | |
| 20. |
Structure of the compound whose IUPAC name is 3-ethyl-2-hydroxy-4-methyhex-3-en5-ynoic acid is |
|
Answer»
|
|
| 21. |
Structure of the compound whose IUPAC name is 3-ethyl. 2-hydroxy-4-methyl hex-3-en-5-ynoic acid is: |
|
Answer»
|
|
| 22. |
Structure of the complex with sp^3 hybridisation |
|
Answer» PLANAR triangular |
|
| 23. |
Structure of styrene butadine rubber is |
|
Answer» `(-CH_2-CH=CH-CH_2-CH_2-underset(|) CH-underset(C_(6)H_(5))(" ") )_n` |
|
| 24. |
Structure of product formod in the reaction is (AAK_MCP_37_NEET_CHE_E37_034_Q01) |
|
Answer»
|
|
| 25. |
Structure of Silicone polymer is |
|
Answer» `(-O-overset(|)overset(O)overset(|)underset(|)underset(O)underset(|)Si-O)_(N)` |
|
| 26. |
Structure of NaCl crystal is. |
|
Answer» Face CENTRED cubic |
|
| 27. |
Structure of Na_(2)B_(4)O_(7).10H_(2)O contains --- |
|
Answer» Two TRIANGULAR and two tetrahedral UNITS |
|
| 28. |
Structure of ionic compounds The bigger atom or ion will form the lattice and smaller atom/ion will occupy the voids generally the bigger ion is anion and smaller ion is cation in ionic compounds.The type of void occupied by the cation is decided by radius ratio. Find the correct set of combination for the compound whose cation has its nearest cationic neighbour at a distance of (asqrt2) where 'a' is the length of unit cell. |
|
Answer» <P>(II)(i)(R) `r_(Na^+)/r^(Cl^(-))=0.51` |
|
| 29. |
Structure of Na_(2)[B_(4)O_(5)(OH)_(4)].8H_(2)O contains |
|
Answer» TWO triangular and two tetrahedral UNITS of boron |
|
| 30. |
Structure of ionic compounds The bigger atom or ion will form the lattice and smaller atom/ion will occupy the voids generally the bigger ion is anion and smaller ion is cation in ionic compounds.The type of void occupied by the cation is decided by radius ratio. Identify the correct set of combination for the compound that involves 4:4 coordination number compound |
|
Answer» (I)(ii)(Q) `ZN^(+2)`:occupy alternate T.V. `S^2`:TERM FCC |
|
| 31. |
Structure of ionic compounds The bigger atom or ion will form the lattice and smaller atom/ion will occupy the voids generally the bigger ion is anion and smaller ion is cation in ionic compounds.The type of void occupied by the cation is decided by radius ratio. Identify the correct combination amongst the following - |
|
Answer» (I)(i)(P) NACL:-`Cl^(-)` froms FCC `Na^(+)` occupy octahedral VOIDS Effective number of `Cl^(-)` in v.c.=4 Effective number of `Na^(+)` in v.c.=4 |
|
| 32. |
structure of E is |
|
Answer»
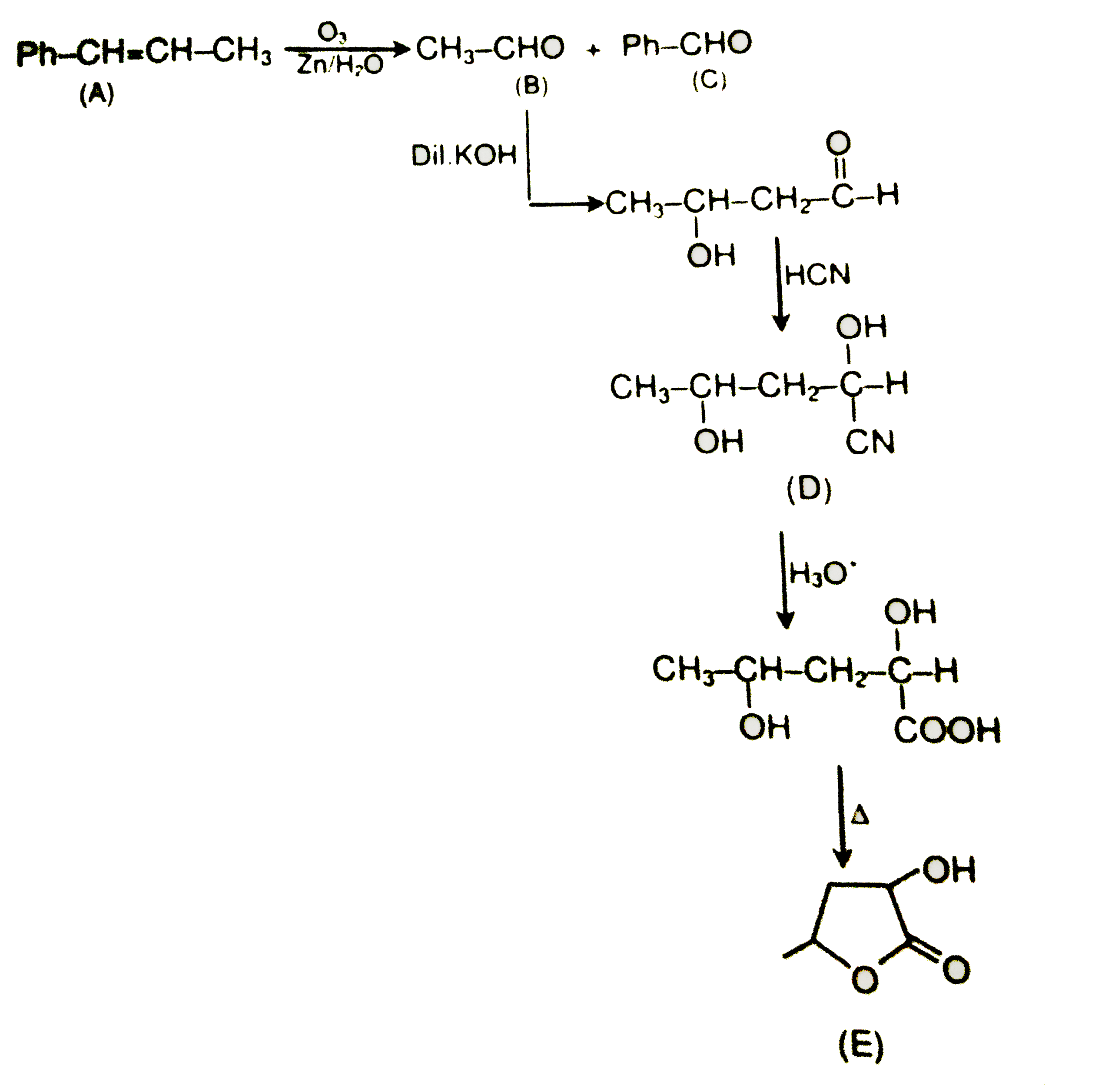
|
|
| 33. |
Structure of ethyl ethanoate is |
|
Answer» `CH_(3)COOC_(2)H_(5)` |
|
| 34. |
Structure of diethyl ether is confirmed by |
|
Answer» KOLBE synthesis |
|
| 35. |
Structure of ICl_2^-is : |
|
Answer» TRIGONAL |
|
| 36. |
Structure of diethyl ether can be confirmed by: |
|
Answer» Kolbe.s synthesis |
|
| 37. |
Structure of diethyl ehter is confirmed by : |
|
Answer» KOLBE's SYNTHESIS |
|
| 39. |
Structure of Cr_(2)O_(7)^(2-) has |
|
Answer» TWO TETRAHEDRA SHARING one corner |
|
| 41. |
Structure of Ascorbic acid as represented as follows. How many of the following reagents can give positive test with ascorbic acid. {:(Cu_(2)Cl_(2)+NH_(4)OH,2","4-DNP,"Na metal",HCl+ZnCl_(2),FeCl,),((I),(II),(III),(IV),(V),),(NaOH+"Phenopthalein","dil".KMnO_(4),Br_(2)//H_(2)O,AgNO_(3)+NH_(4)OH,I_(2)+NaOH,),((VI),(VII),(VIII),(XI),(X),):} |
|
Answer» |
|
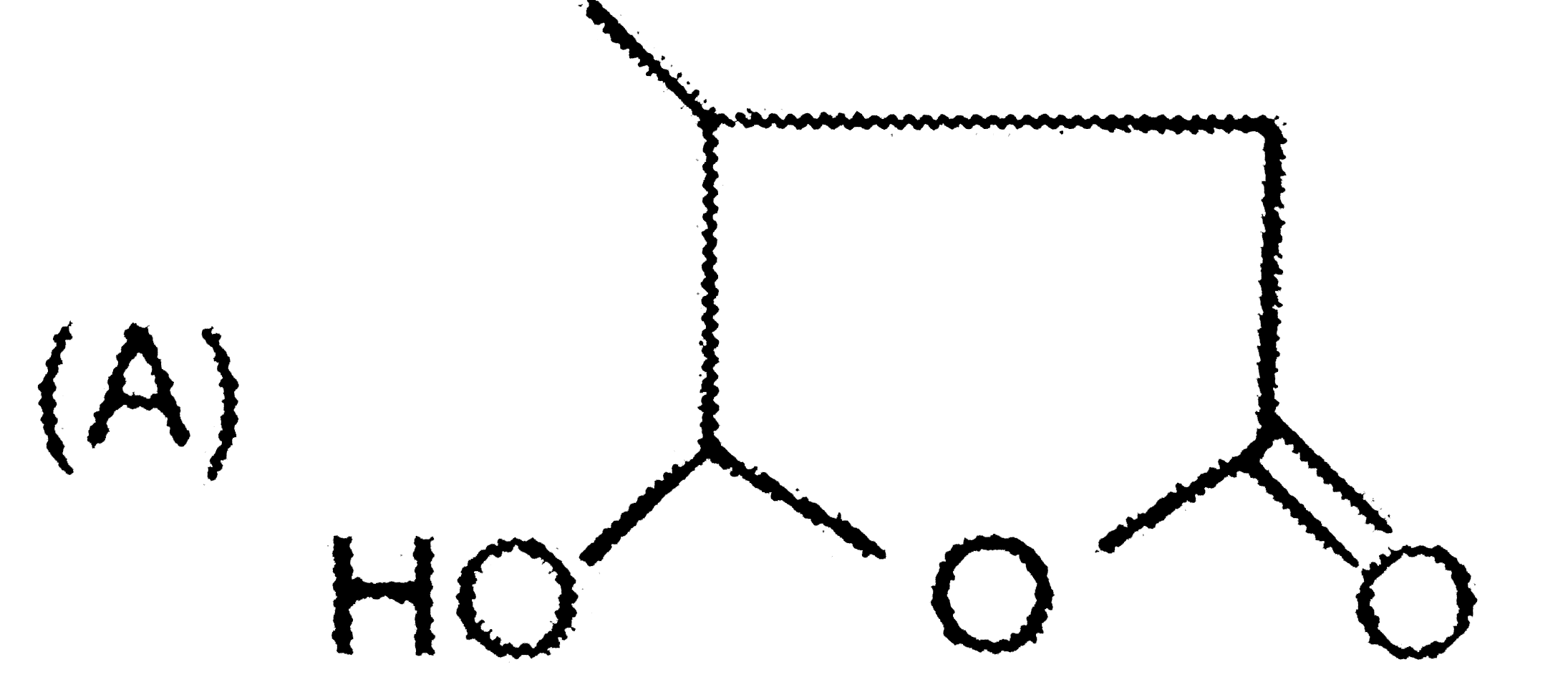
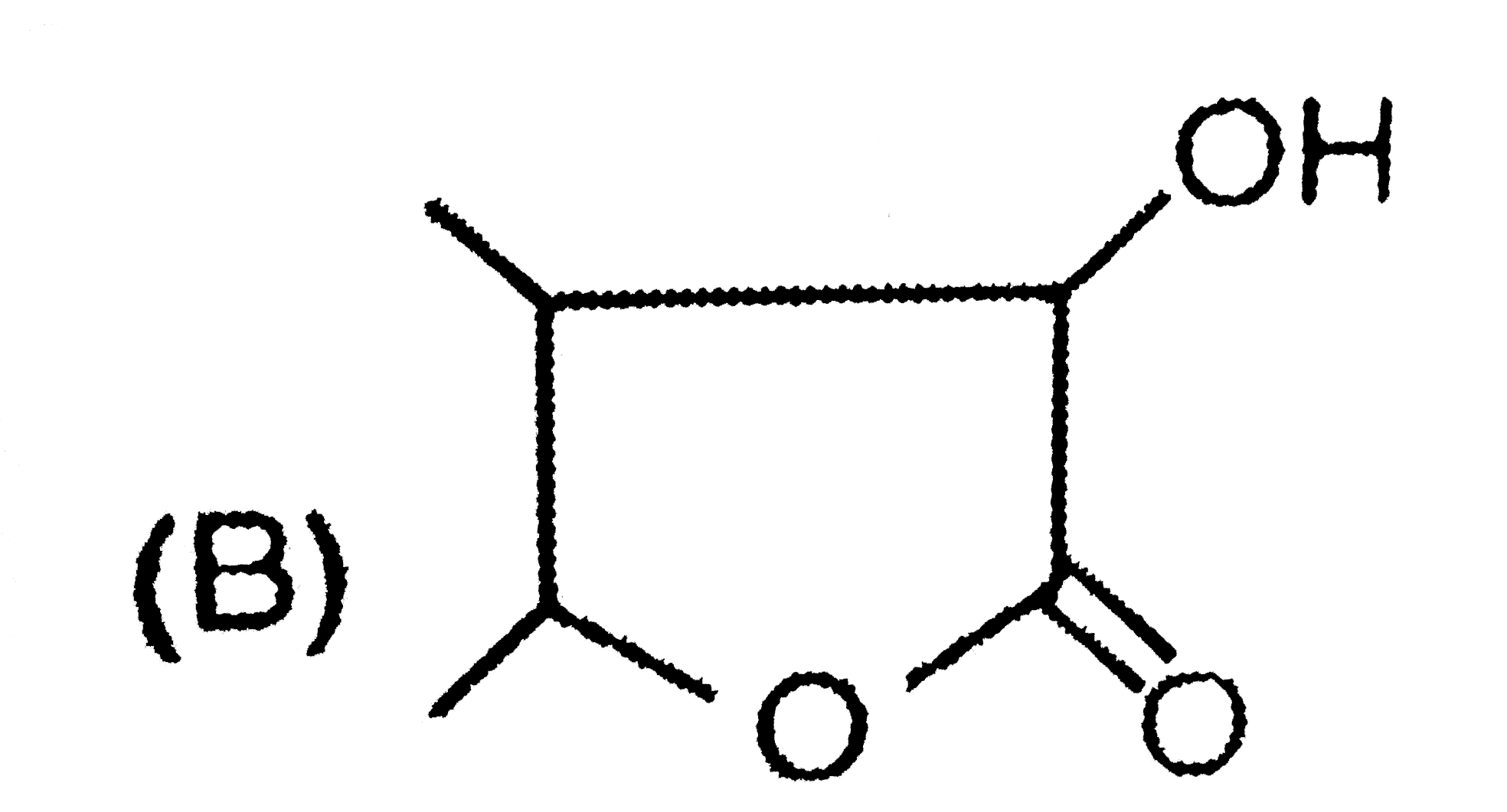
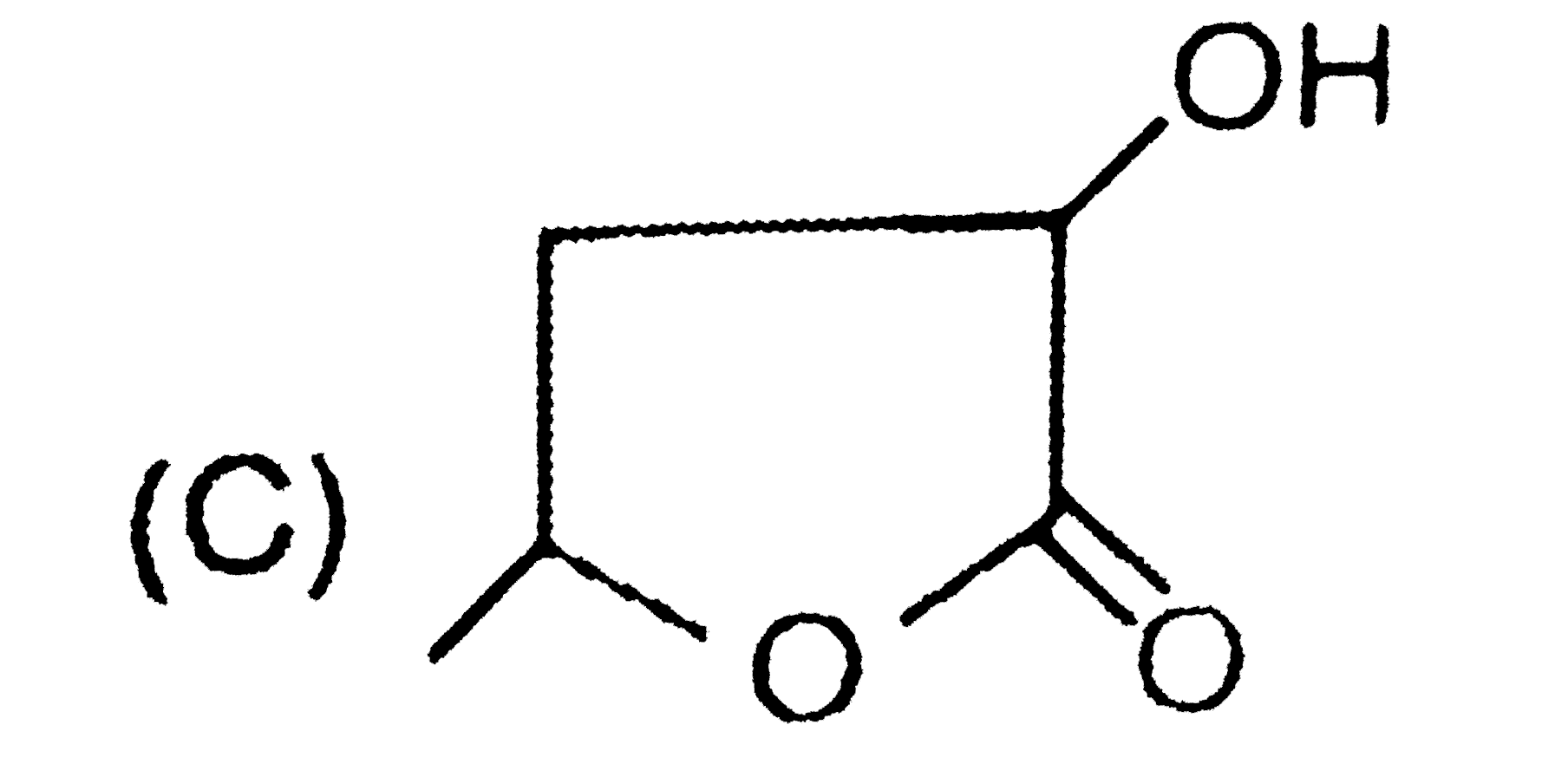
| 42. |
Structure of (B) and (C) differentiated by |
|
Answer» TOLLEN's REAGENT |
|
| 43. |
Structure of AX_(7) type is ………………. . |
| Answer» SOLUTION :PENTAGONAL bipyramidal | |
| 44. |
Structure of ammonia is: |
|
Answer» Trigonal |
|
| 45. |
Structure of a mixed oxide is cubic close-packed (ccp). The unit cell of mixed oxide is composed of oxide ions. One fourth of the tetrahedral voids are occupied by divalent metal A and the octahedral voids are occupied by a monovalent metal B. The formula of the oxide is |
|
Answer» `ABO_(2)` |
|
| 46. |
Structure C_(8)H_(8)Cl_(2) an alkaline hydrolysis gives a product which does not give iodoform test but give silver mirror test ,is |
|
Answer»
|
|
| 47. |
Structure of A is |
|
Answer» `H_(2)C=CH-CHO` |
|
| 49. |
Structure of 2-methyl 2-halo butane is |
|
Answer» `CH_(3)-CX(CH_(3))C_(2)H_(5)` |
|
| 50. |
Structure of (A) and (B) respectively are: |
|
Answer»

|
|