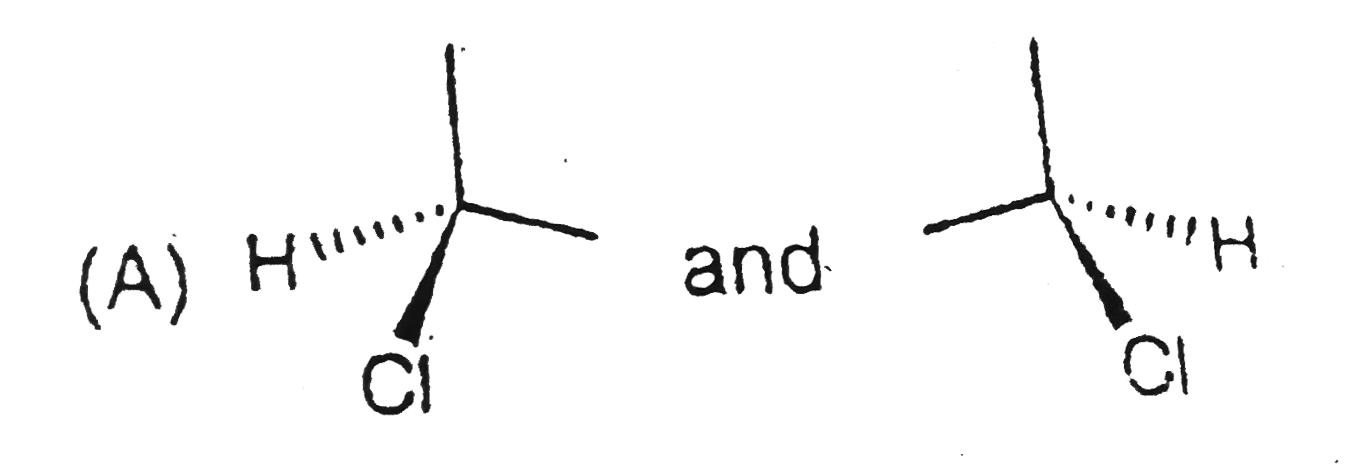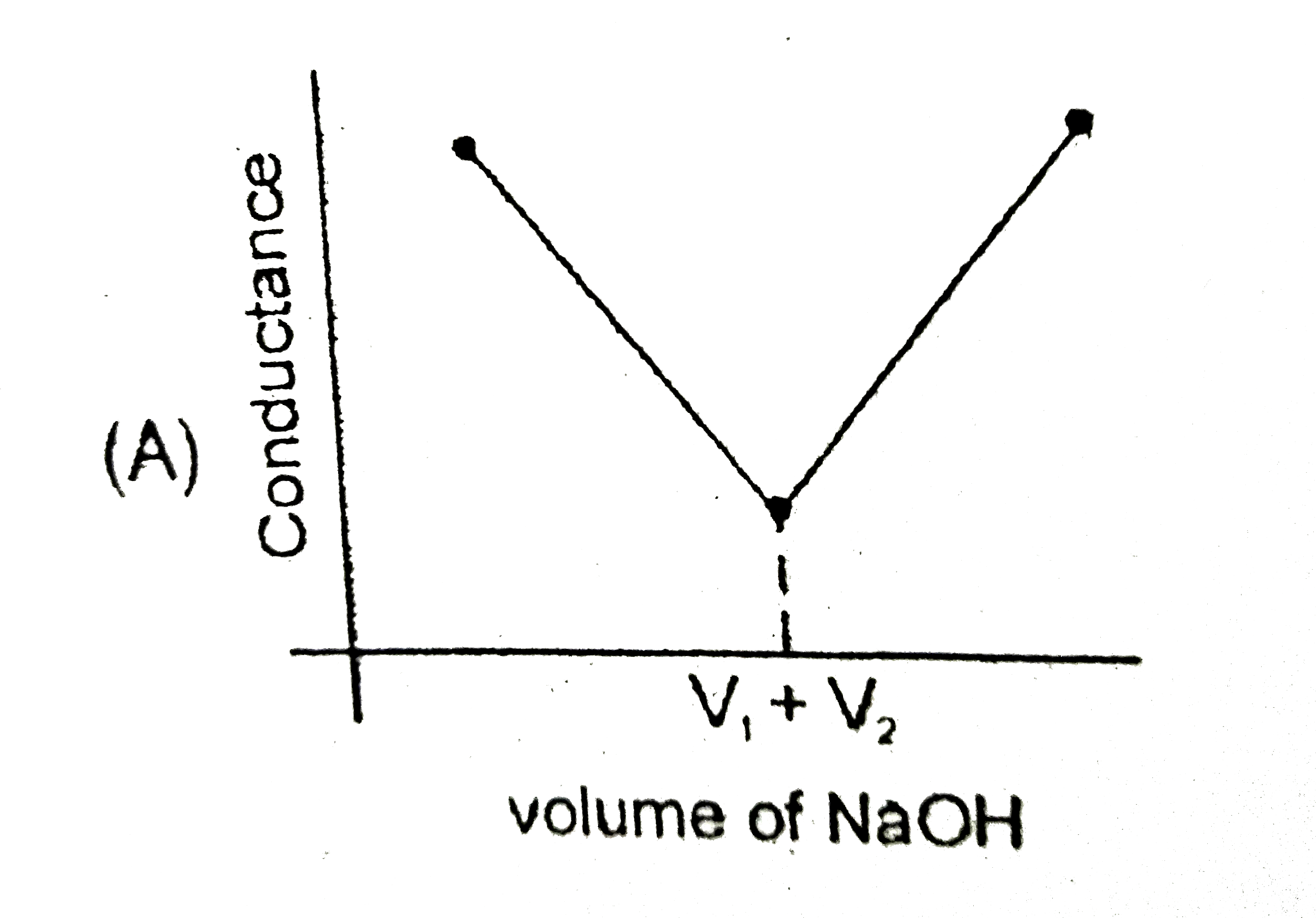Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Structurally a biodegradable detergent should contain a |
|
Answer» NORMAL ALKYL chain |
|
| 2. |
Structurally biodegradable detergent should contain |
|
Answer» UNBRANCHED ALKYL chain |
|
| 3. |
Structurally, cellulose is a linear polymer of : |
|
Answer» `BETA`-glucose MOLECULES |
|
| 4. |
Structurally a biodegradable detergent should contain a : |
|
Answer» NORMAL ALKYNES |
|
| 5. |
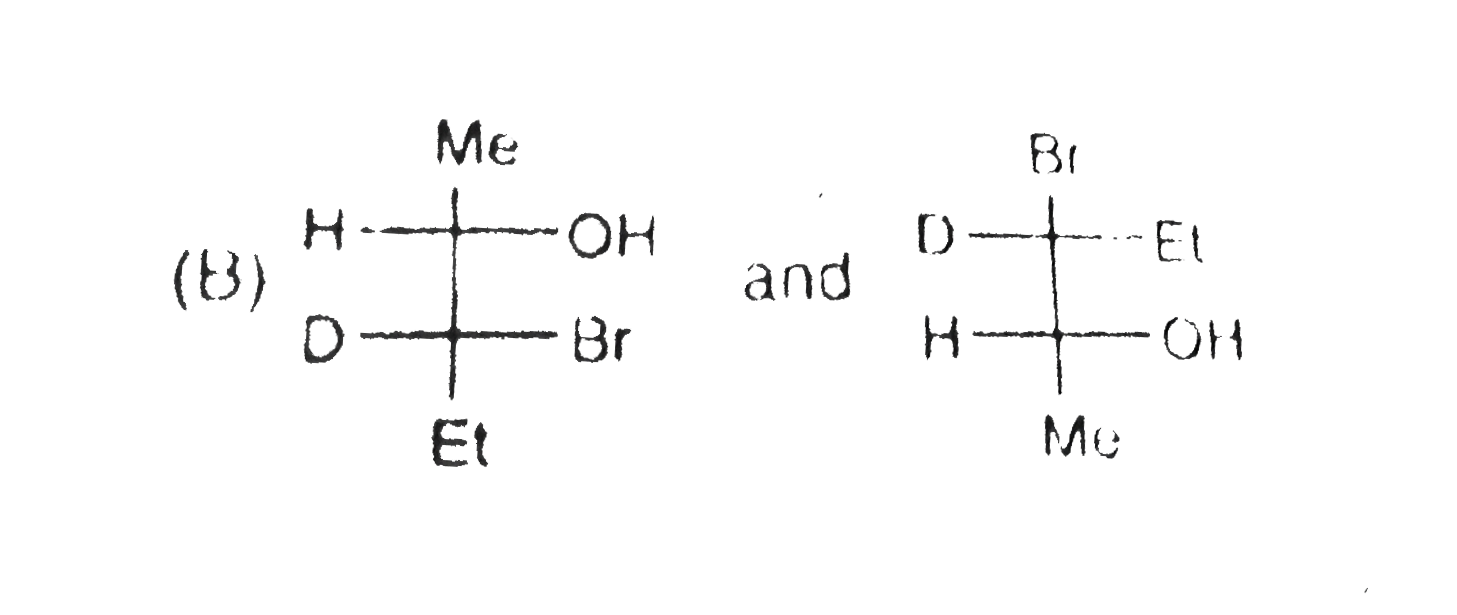
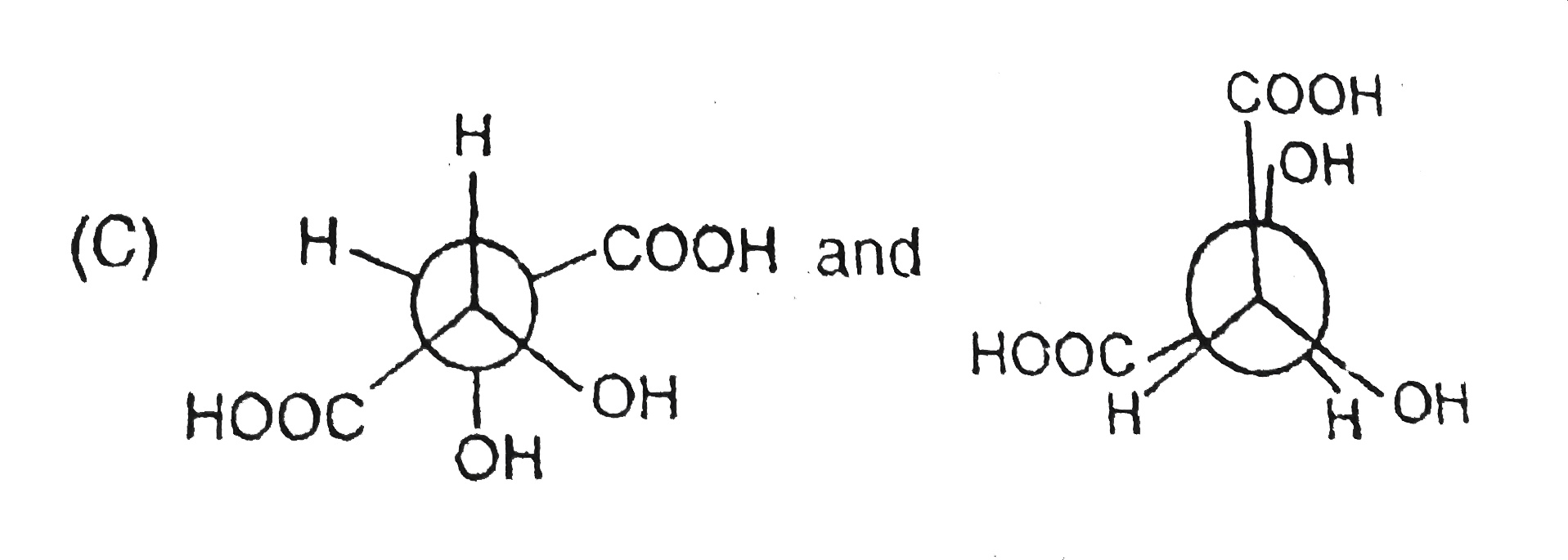
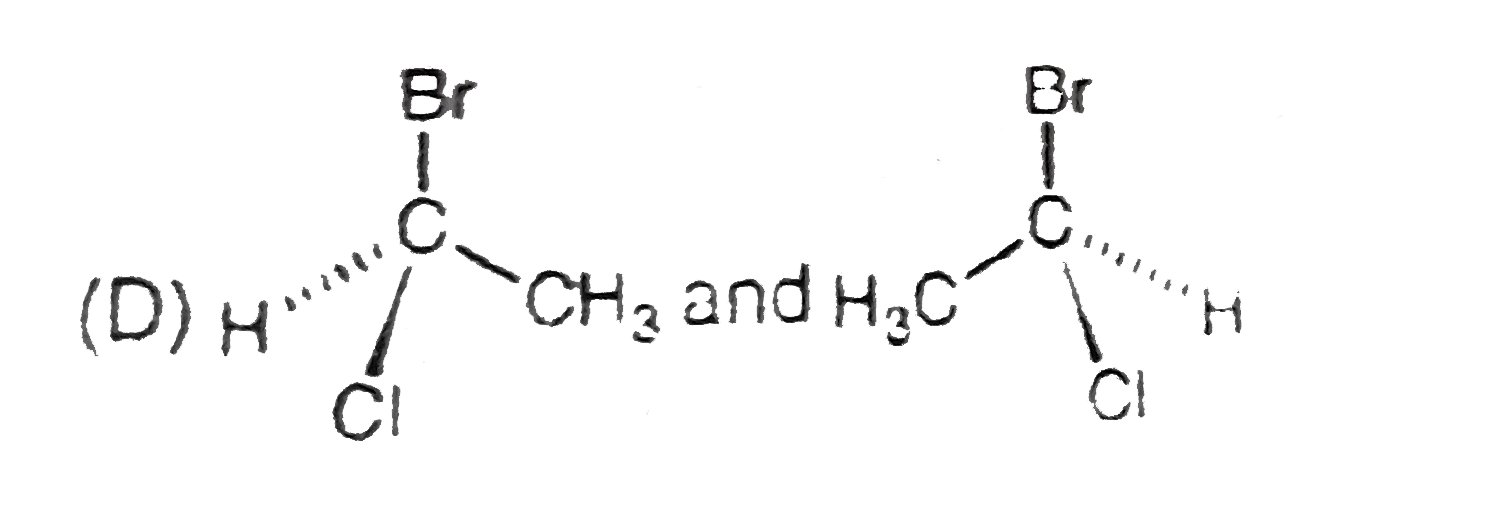
Structural isomers have different covalent linkage of atoms.Steroisomers are compounds that have same sequence of covalent bonds but differ in the relative positions of their atoms in space.Geometrical and optical isomers are the two important types of configurational isomers. The compound with double bonds or ring structure have restricated rotation.so exist in two geometrical forms.The double bonds in the larger rings (ring size 10 carbon or large) can also cause geometrical isomerism. The optical isomers rotate the plane of plane-polarised light.A sp^3-hybridised carbon atom bearing four different types of substituents is called an asymmetric centre or chiral centre.A chiral object or molecule cannot be superimposed on its mirror image.Stereoisomers that are mirror images of each other are called enantiomers.The stereoisomers that are not mirror images of each other are called diastereomers.Diastereomers have different physical properties. A racemic mixture is optically inactive and contains equal amount of both the enantiomers.Resolution refers to method of separating a racemic mixture into two pure enantiomers.A meso compound is an optically inactive stereoisomers, which is achiral due to the presence of an internal plane of symmetry within the molecule. The pair showing identifical species is |
|
Answer»
B,C,D have pairs of compounds which have CHIRAL CARBON and are NON superimposible on each other |
|
| 6. |
Structural unit of vit-B is |
|
Answer»
|
|
| 8. |
Structural isomers have different covalent linkage of atoms.Steroisomers are compounds that have same sequence of covalent bonds but differ in the relative positions of their atoms in space.Geometrical and optical isomers are the two important types of configurational isomers. The compound with double bonds or ring structure have restricated rotation.so exist in two geometrical forms.The double bonds in the larger rings (ring size 10 carbon or large) can also cause geometrical isomerism. The optical isomers rotate the plane of plane-polarised light.A sp^3-hybridised carbon atom bearing four different types of substituents is called an asymmetric centre or chiral centre.A chiral object or molecule cannot be superimposed on its mirror image.Stereoisomers that are mirror images of each other are called enantiomers.The stereoisomers that are not mirror images of each other are called diastereomers.Diastereomers have different physical properties. A racemic mixture is optically inactive and contains equal amount of both the enantiomers.Resolution refers to method of separating a racemic mixture into two pure enantiomers.A meso compound is an optically inactive stereoisomers, which is achiral due to the presence of an internal plane of symmetry within the molecule. Observe the following reaction Which statement is not correct about the above observation. |
|
Answer» The PRODUCT mixture of step -1 is optically active |
|
| 9. |
Structuralformulafor methylaminomethaneis |
|
Answer» `CH_(3)- NH_(2)` |
|
| 10. |
Structural differences are main in the following isomers (A) ionisation isomerism, (B) ligand isomerism ,(C) hydrate isomerism, ( D) geometrical isomerism |
|
Answer» A,B,C |
|
| 11. |
Strontium is chemically similar to calcicum and can replace calcium in bones. The radiation from Sr^(90) can damage bone narrow marrow where blood cells are produced, and lead to serious health problems. The time taken for 99.99% of a sample of Sr^(90) is lambda "for" Sr^(90)=0.23yr^(-1): |
|
Answer» 10yr |
|
| 12. |
Structur of ammonia is |
|
Answer» TRIGONAL bipyramidal |
|
| 13. |
Strongest reducing agent is |
|
Answer» K |
|
| 14. |
Strongly acidified solution of barium give a white precipitatewith ……which did not dissolve even after large addition of water |
|
Answer» SODIUM phosphate |
|
| 16. |
Strongest reducing agent among alkali metals and alkaline earth metals are respectively :- |
|
Answer» Solution :order of REDUCING strength (i) `IA - Li gt K gt Rb gt Cs gt Na` (II) `IIA - Ba gt Sr gt Ca gt Mg gt Be` |
|
| 17. |
Strongest reducing agent among the following is: |
|
Answer» K |
|
| 18. |
Strongest oxidising agent is |
| Answer» Solution :`Pb^(+VI)` is good oxidising agent | |
| 19. |
Strongest interparticle forces exist in: |
|
Answer» Elastomers |
|
| 20. |
Strongest hydrogen bond exhibited in _____. |
|
Answer» H-F……….H, |
|
| 21. |
Strongest conjugate base among the following is : |
|
Answer» `NO_3^-` |
|
| 22. |
Strongest bond is in between |
|
Answer» `CsF` |
|
| 23. |
Strongest bond is |
|
Answer» C-C |
|
| 24. |
Strongest base is |
|
Answer» `C_(6)H_(5)NH_(2)`  and `HC-=C CH_(2)NH_(2),sp^(2)` and sp carbon atoms respectively being more ELECTRONEGATIVE aattract the lone pair of electrons on N thereby reducing the ELECTRON density. in contrast, the +I-effect of `CH_(3)CH_(2)CH_(2)` group INCREASES the electron density on N. thus, `CH_(3)CH_(2)CH_(2)NH_(2)` is the strongest base. |
|
| 25. |
Strongest acid is: |
|
Answer» `CH_(3)COOH` |
|
| 26. |
Strongest acid is |
|
Answer» `HC -= CH` |
|
| 27. |
Strongest acid among the following is : |
|
Answer» `CF_(3)COOH` |
|
| 28. |
Strongest acid amon the following is |
|
Answer» o-methoxyphenol |
|
| 29. |
Stronger the oxidising agent greater is the |
|
Answer» OXIDATION potential |
|
| 30. |
Stronger the oxidising agent , greater is the : |
|
Answer» REDUCTION potential |
|
| 31. |
Stronger is oxidising agent, more is : |
|
Answer» STANDARD reduction POTENTIAL of that species |
|
| 32. |
Strong reducing behaviour of H_3PO_4 is due to.. |
|
Answer» low oxidation STATE of E |
|
| 33. |
Strong reducing behaviour of H_3PO_2is due to |
|
Answer» LOW oxidation state of phosphorus |
|
| 34. |
Strong reducing behaviour of H_(3)PO_(2) is due to: |
|
Answer» High oxidation state of phosphorus All oxy-acid of phosphorous which CONTAIN `P-H` bond act as REDUCTANT. 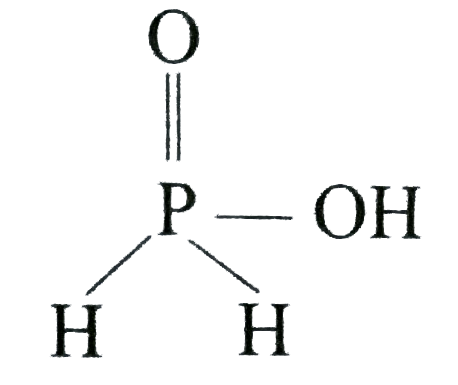 Presence of one -`OH` group and two `P-H` bonds. |
|
| 35. |
Strong reducing behaviour of H_3PO_2 is due to : |
|
Answer» High oxidation state of phosphorus. STRONG reducing behaviour of `H_3PO_2`. All oxy-acid of phosphorus which CONTAIN P-H bond act as reductant. Presence of one -OH group and two P-H bonds. 
|
|
| 36. |
Strong reducing behaviour ofH_(3)PO_(2) is due to _________ |
|
Answer» High OXIDATION STATE of phosphorus |
|
| 37. |
Strong reducing behaviour of H_3PO_2 is due to ............... |
|
Answer» low oxidation STATE of phosphorus. 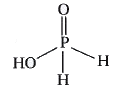
|
|
| 38. |
Strong reducing agent converts CHCl_3 into: |
|
Answer» `C_2H_2` |
|
| 39. |
Strong intermolecular attraction force in the Nylon-66 polymer is due to …………………… bond. |
|
Answer» Van-der WAALS |
|
| 40. |
Strong activatingefferctof -NH_(2) groupis reducedby using |
|
Answer» `CH_(3)COCl` |
|
| 41. |
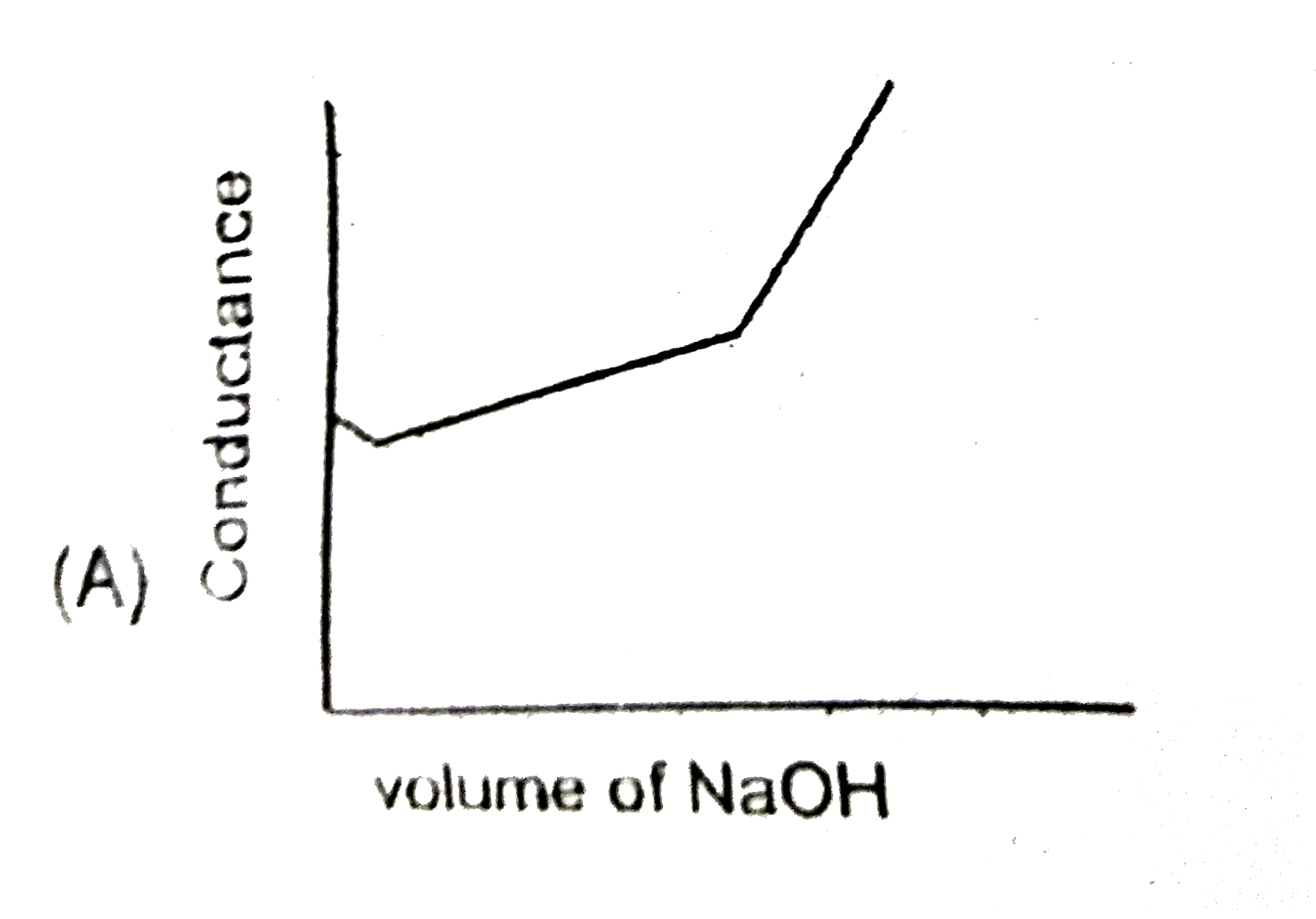
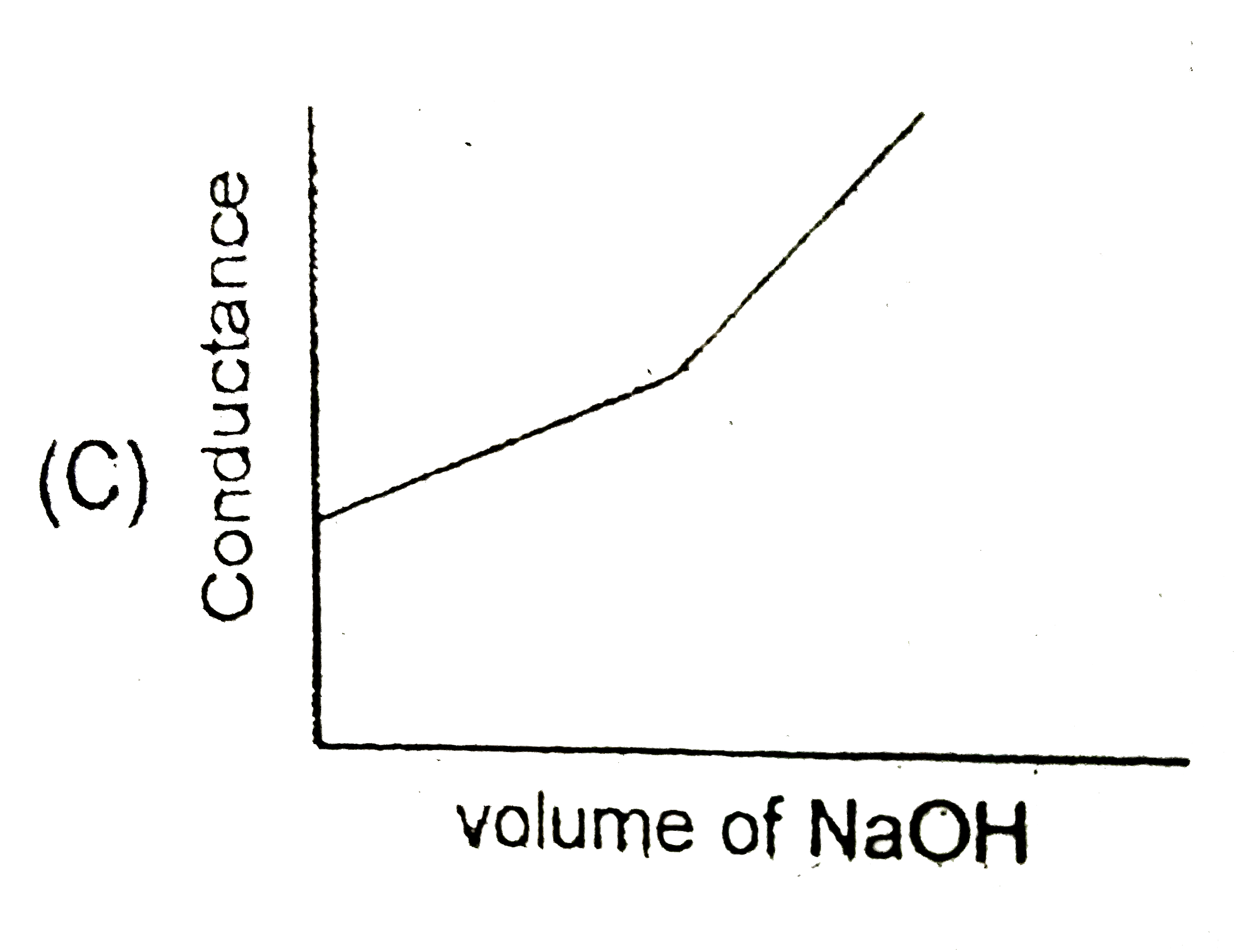
Strong acid versus strong base: The principle of conductometric titrations is based on the fact that during the titration, one of the ions is replaced by the other and invariable these two ions differ in the ionic conductivity with the result that thhe conductivity of the solution varies during the course of the titration. take, for example, the titration between a strong acid, say HCl, and a string base, say NaOH before NaOH is added, the conductance of HCl solution has a high value due to the presence of highly mobile hydrogen ions. As NaOH is added, H^(+) ions are replaced by relatively slower moving Na^(+) ions. consequently the conductance of the solution decreases and this continues right upto the equivalence point where the solution contains only NaCl. Beyond the equivalence point, if more of NaOH is added, then the solution contains a excess of the fast moving OH^(-) ions with the result that its conductance is increased ad it condinues to increase as more and more of NaOH is added. If we plot the conductance value versus the amount of NaOH added, we get a curve of the type shown in Fig. The descending portion AB represents the conductances before the equivalence point (solution contains a mixture of acid HCl and the salt NaCl) and the ascendingportion CD represents the conductances after the equivalence point (solution contains the salt NaCl and the excess of NaOH). The point E which represent the minium conductance is due to the solution containing only NaCl with no free acid or alkali and thus represents the equivalence point. this point can, however, be obtained by the extrapolation of the lines AB and DC, and therefore, one is not very particular in locating this point expermentally as it is in the case of ordinary acid-base titrations involving the acid-base indicators. Weak acid versus strong base: Let us take specific example of acetic acid being titrated against NaOH. Before the addition of alkali, the solution shows poor conductance due to feeble ionization of acetic acid. Initially the addition of alkali causes not only the replacement of H^(+) by Na^(+) but also suppresses the dissociation of acetic acid due to the common ion Ac^(-) and thus the conductance of the solution decreases in the beginning. but very soon the conductance start increasing as addition of NaOH neutralizes the undissociated HAc to Na^(+)Ac^(-) thus causing the replacement of non-conducting HAc with Strong-conducting electrolyte Na^(+)Ac^(-). the increase in conductance continuous right up to the equivalence point. Beyond this point conductance increases more rapidly with the addition of NaOH due to the highly conducting OH^(-) ions, the graph near the equivalence point is curved due to the hydrolysis of the salt NaAc. The actual equivalence point can, as usual, be obtained by the extrapolation method. In all these graphs it has been assumed that the volume change due addition of solution from burrette isnegnigible, hence volume change of the solution in beaker the conductance of which is measured is almost constant throughout the measurement. Q. If a 100 mL solution of 0.1 M HBr is titrated using a very concentrated solution of NaOH, then the conductivity (specific conductance) of this solution at the equivalence point will be (assume volume change is negligible due to addition of NaoH) report your answer after multipling it with 10 in Sm^(-1) [Given lamda_((Na^(+)))^(@)=8xx10^(-3)Sm^(2)mol^(-1),lamda_((Br^(-)))^(@)=4xx10^(-3)Sm^(2)mol^(-1)] |
| Answer» Answer :B | |
| 42. |
Strong heating of produces N, N-dimethyl hydroxylamine along with |
|
Answer» `CH_(2)=CH-CD_(3)` 
|
|
| 43. |
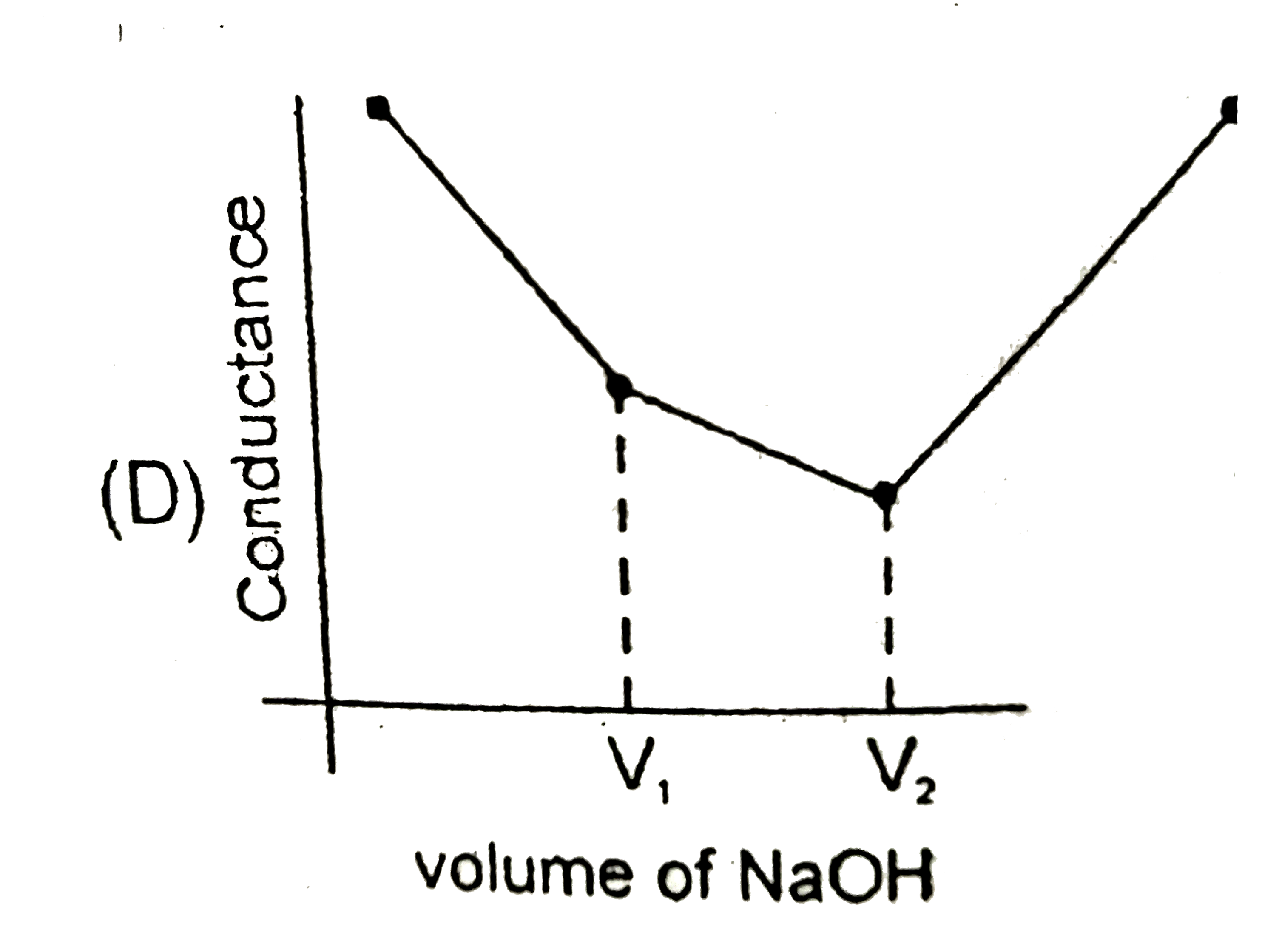
Strong acid versus strong base: The principle of conductometric titrations is based on the fact that during the titration, one of the ions is replaced by the other and invariable these two ions differ in the ionic conductivity with the result that thhe conductivity of the solution varies during the course of the titration. take, for example, the titration between a strong acid, say HCl, and a string base, say NaOH before NaOH is added, the conductance of HCl solution has a high value due to the presence of highly mobile hydrogen ions. As NaOH is added, H^(+) ions are replaced by relatively slower moving Na^(+) ions. consequently the conductance of the solution decreases and this continues right upto the equivalence point where the solution contains only NaCl. Beyond the equivalence point, if more of NaOH is added, then the solution contains a excess of the fast moving OH^(-) ions with the result that its conductance is increased ad it condinues to increase as more and more of NaOH is added. If we plot the conductance value versus the amount of NaOH added, we get a curve of the type shown in Fig. The descending portion AB represents the conductances before the equivalence point (solution contains a mixture of acid HCl and the salt NaCl) and the ascendingportion CD represents the conductances after the equivalence point (solution contains the salt NaCl and the excess of NaOH). The point E which represent the minium conductance is due to the solution containing only NaCl with no free acid or alkali and thus represents the equivalence point. this point can, however, be obtained by the extrapolation of the lines AB and DC, and therefore, one is not very particular in locating this point expermentally as it is in the case of ordinary acid-base titrations involving the acid-base indicators. Weak acid versus strong base: Let us take specific example of acetic acid being titrated against NaOH. Before the addition of alkali, the solution shows poor conductance due to feeble ionization of acetic acid. Initially the addition of alkali causes not only the replacement of H^(+) by Na^(+) but also suppresses the dissociation of acetic acid due to the common ion Ac^(-) and thus the conductance of the solution decreases in the beginning. but very soon the conductance start increasing as addition of NaOH neutralizes the undissociated HAc to Na^(+)Ac^(-) thus causing the replacement of non-conducting HAc with Strong-conducting electrolyte Na^(+)Ac^(-). the increase in conductance continuous right up to the equivalence point. Beyond this point conductance increases more rapidly with the addition of NaOH due to the highly conducting OH^(-) ions, the graph near the equivalence point is curved due to the hydrolysis of the salt NaAc. The actual equivalence point can, as usual, be obtained by the extrapolation method. In all these graphs it has been assumed that the volume change due addition of solution from burrette isnegnigible, hence volume change of the solution in beaker the conductance of which is measured is almost constant throughout the measurement. Q. The most appropriate titration curve obtained when a mixture of a strong acid (say HCl) and a weak acid (say CH_(3)COOH) is titrated with a strong base (say NaOH) will be |
|
Answer»
|
|
| 44. |
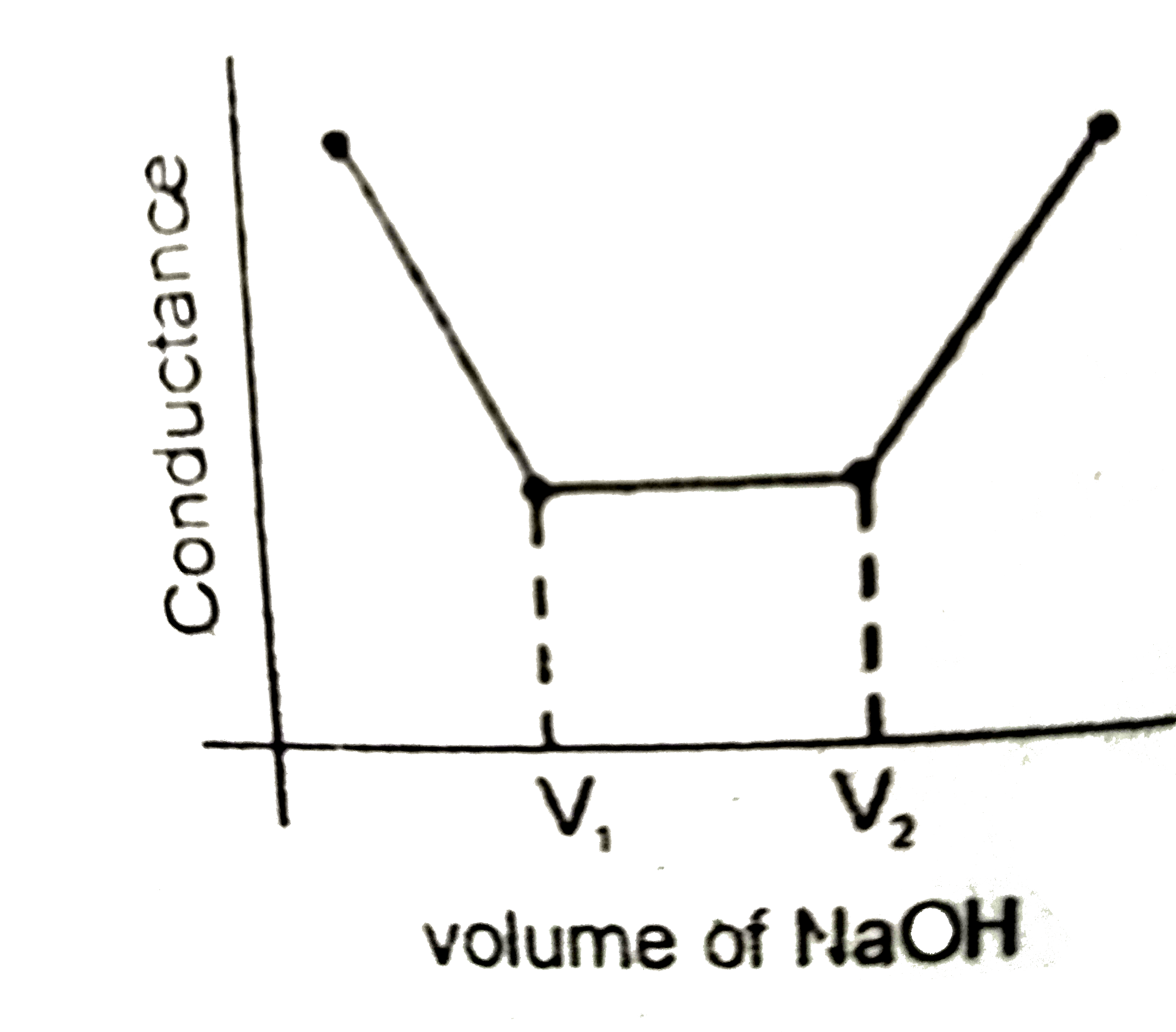
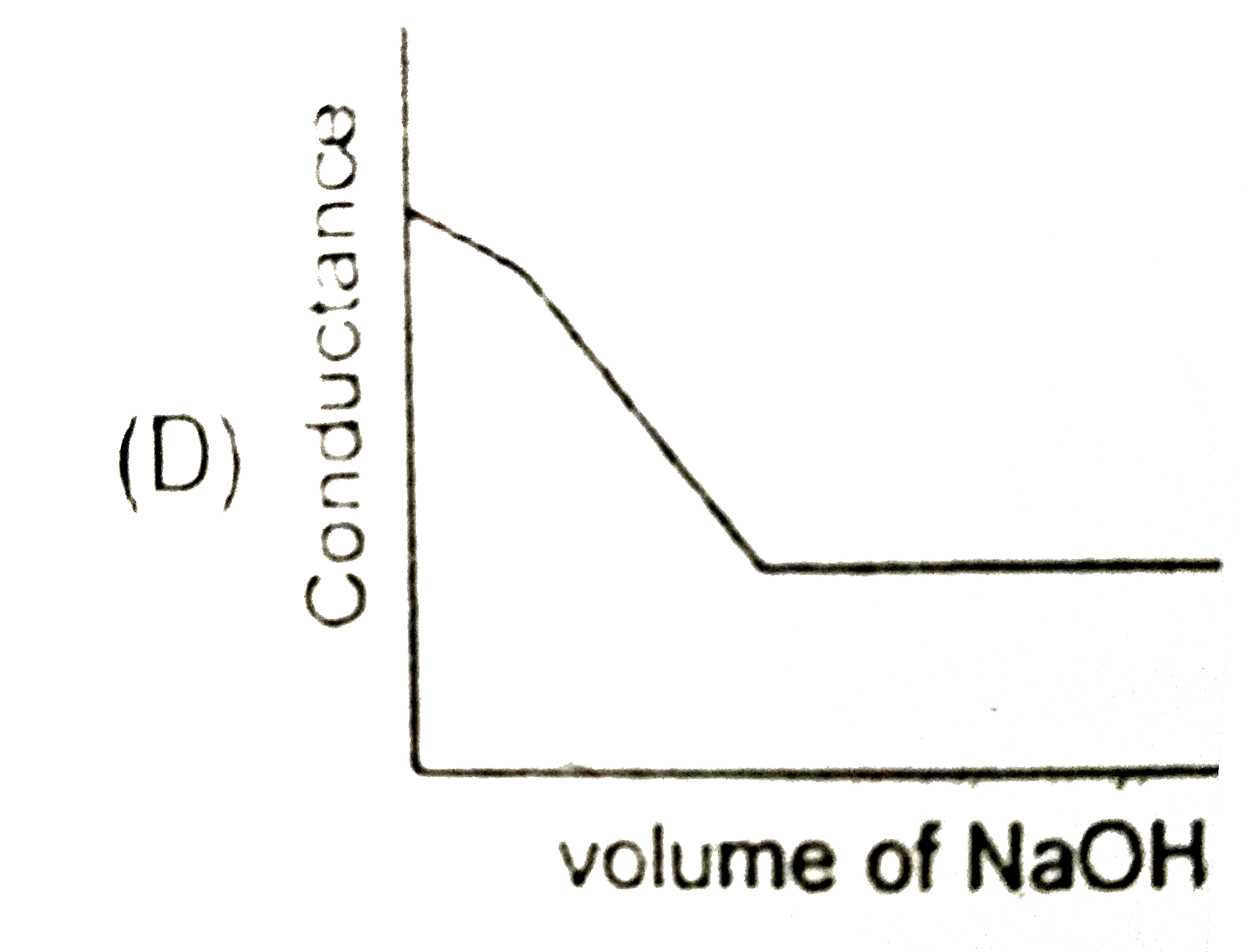
Strong acid versus strong base: The principle of conductometric titrations is based on the fact that during the titration, one of the ions is replaced by the other and invariable these two ions differ in the ionic conductivity with the result that thhe conductivity of the solution varies during the course of the titration. take, for example, the titration between a strong acid, say HCl, and a string base, say NaOH before NaOH is added, the conductance of HCl solution has a high value due to the presence of highly mobile hydrogen ions. As NaOH is added, H^(+) ions are replaced by relatively slower moving Na^(+) ions. consequently the conductance of the solution decreases and this continues right upto the equivalence point where the solution contains only NaCl. Beyond the equivalence point, if more of NaOH is added, then the solution contains a excess of the fast moving OH^(-) ions with the result that its conductance is increased ad it condinues to increase as more and more of NaOH is added. If we plot the conductance value versus the amount of NaOH added, we get a curve of the type shown in Fig. The descending portion AB represents the conductances before the equivalence point (solution contains a mixture of acid HCl and the salt NaCl) and the ascendingportion CD represents the conductances after the equivalence point (solution contains the salt NaCl and the excess of NaOH). The point E which represent the minium conductance is due to the solution containing only NaCl with no free acid or alkali and thus represents the equivalence point. this point can, however, be obtained by the extrapolation of the lines AB and DC, and therefore, one is not very particular in locating this point expermentally as it is in the case of ordinary acid-base titrations involving the acid-base indicators. Weak acid versus strong base: Let us take specific example of acetic acid being titrated against NaOH. Before the addition of alkali, the solution shows poor conductance due to feeble ionization of acetic acid. Initially the addition of alkali causes not only the replacement of H^(+) by Na^(+) but also suppresses the dissociation of acetic acid due to the common ion Ac^(-) and thus the conductance of the solution decreases in the beginning. but very soon the conductance start increasing as addition of NaOH neutralizes the undissociated HAc to Na^(+)Ac^(-) thus causing the replacement of non-conducting HAc with Strong-conducting electrolyte Na^(+)Ac^(-). the increase in conductance continuous right up to the equivalence point. Beyond this point conductance increases more rapidly with the addition of NaOH due to the highly conducting OH^(-) ions, the graph near the equivalence point is curved due to the hydrolysis of the salt NaAc. The actual equivalence point can, as usual, be obtained by the extrapolation method. In all these graphs it has been assumed that the volume change due addition of solution from burrette isnegnigible, hence volume change of the solution in beaker the conductance of which is measured is almost constant throughout the measurement. Q. The nature of curve obtained for the titration between weak acid versus strong base as described in the above passage will be: |
|
Answer»
|
|
| 45. |
Stright chain of a hydrocarbon is preferred over branched chain as a detergent. Why? |
| Answer» Solution :Branched CHAIN DETERGENTS are non-biodegradable. Unbranched hydrocarbon chain of a DETERGENT is READILY attacked by BACTERIA and hence is preferred. | |
| 46. |
Streptomycin, a well-known antibiotic, is a dervative of |
|
Answer» PEPTIDES |
|
| 47. |
Streptomycin on hum,an body. |
|
Answer» Solution :(i) ANALGIN is antipyretis and analgesics. (II) Luminal produces SLEEP and HABIT forming. (III) Seconal is an autidepressant drug. (iv) Streptomycin is an antibiotic. |
|
| 48. |
Streptomycin is used to _____. |
| Answer» SOLUTION :Ccure DISEASES CAUSED by GRAM +ve , -ve BACTERIA | |
| 49. |
Streptomycin is effective in the treatment of: |
|
Answer» Tuberculosis |
|
| 50. |
Streptomycin is effective in the treatment of ... |
|
Answer» TUBERCULOSIS |
|