Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Streptomycin, a well known antibiotic, is a derivative of |
|
Answer» Peptides |
|
| 2. |
Strength of hydrogen bond is intermediate between |
|
Answer» VANDER WAAL and COVALENT |
|
| 3. |
Strength of halogen acids, HF, HCl, HBr and HI varies as : |
|
Answer» `HF GT HCL gt HBr gt HI` |
|
| 4. |
Strength of H-bond and boiling point order is opposite for :- |
|
Answer» `HF, H_(2)O, H_(2)O_(2)` BOILING point `-HF lt H_(2)O lt H_(2)O_(2)` |
|
| 5. |
Strength of acideity is in order |
|
Answer» II GT I gt III gt IV |
|
| 6. |
Strength of 2N H2O2 solution is approximately |
|
Answer» 10 volume |
|
| 7. |
Straight chain polymer is formed by hydrolysis of [x] which is tetrasubstituted chloromethylsilane then followed by condensation polymerization. Atomicity of [x] is: |
|
Answer» |
|
| 8. |
Straight line slope of Freundlich isotherm curve indicates the value of ......... |
|
Answer» `N` |
|
| 9. |
Streling silver: |
|
Answer» Is an alloy of AG + CU |
|
| 11. |
Stoichiometric defects in an ionic solid is also called ……………………or …………………..defect. |
| Answer» SOLUTION :INTRINSIC or THERMODYNAMIC | |
| 12. |
Stoichiometric defects are of two types such as vacancy defects and interstitial defects. Which stoichiometric defect causes the decrease in density of solid? |
|
Answer» |
|
| 13. |
Stoichiometric defects are of two types such as vacancy defects and interstitial defects. Which defect is basically a vacancy defect in ionic solids? |
|
Answer» |
|
| 14. |
Stinges of bees, red ant and wasps contain : |
|
Answer» FORMALIN |
|
| 15. |
Stibene (PhCH=CHPh). Can exist in two idastereomeric forms (X) and (Y) and (X) is found to be more soluble in water than (Y). Predict which of the following statement is correct? |
|
Answer» X is trans isomer |
|
| 16. |
Stibene (PhCH=CHPh) can exist in two diastereomeric forms (X) and (Y), and (X) is found to be more soluble in water than (Y). Predict which of the following statement is correct. |
|
Answer» X is TRANS isomer 
|
|
| 17. |
Steroids are derived from |
|
Answer» HIGHLY BRANCHED glycerides |
|
| 18. |
Steroisomes have different |
|
Answer» MOLECULAR formula |
|
| 19. |
Sterol, the basic unit of vitamin D, consists of 4 rings they are |
|
Answer» Three 6-carbon RINGS ONE FIVE carbon ring |
|
| 20. |
Sterility is caused because of ……………….. |
|
Answer» VITAMIN E DEFICIENCY |
|
| 21. |
Steric factor, P, refers to the orientation of molecules which collide and contributes to __________ collision. |
| Answer» SOLUTION :EFFECTIVE | |
| 22. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. Select the correct sequence of decreasing order of stability? |
|
Answer» GAUCHE `GT` staggered `gt` partially eclipsed `gt` FULLY eclipsed |
|
| 23. |
Stericeffectof methyl groupreduces basic characterof . |
|
Answer» `3^(0)`AMINES |
|
| 24. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. The energy barrier between eclipsed and staggered forms is: |
|
Answer» 44 kJ/mol |
|
| 25. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. Which among the following conformations of cyclohexane is the most stable form? |
|
Answer» CHAIR FORM |
|
| 26. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. Dihedral angle is staggered and eclipsed conformation are: |
|
Answer» `60^(@)` and `0^(@)` |
|
| 27. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. Which of the following molecules has the highest deviation from tetrahedral bond angle? |
|
Answer» Cyclopropane |
|
| 28. |
Stereoisomers, which can be interconverted simply by rotation about sigma bonds, are confomational isomers while those, which can be converted only by breaking and remaking of bonds and not simply by rotation, are called configurational isomers. The angle between C-C and C-H bonds on adjacent carbon atoms in any conformation is called dihedral angle. The cyclic compounds most commonly found in nature containing six membered rings can exist in a conflormation that is almost completely free of strain. the most stable conformation of cyclohexane is chair form. According to Bayer strain theory, the greater deviation from the normal tetrahedral angle, greater is the angle strain or torsional strain and hence lesser is the stability of the cycloalkane. Dihedral angle between two methyl groups of n-butane in the gauche and anti forms are: |
|
Answer» `60^(@),0^(@)` |
|
| 29. |
Stereoisomers may be of different types. Stereosiomers that are radialy interconvertible by rotation around a sigma-bond are known as_______isomers |
|
Answer» geometrical |
|
| 30. |
Stereoisomers have different : |
|
Answer» MOLECULAR FORMULA |
|
| 31. |
Stereoisomers have different: |
|
Answer» MOLECULAR formula |
|
| 32. |
Stereoisomerism occurs when the bond connectivity is same but the spatial arrangement of more than one type is possible. Stereoisomerism finds vast use in orgaic chemistry, mainly biochemistry as all the biomolecules, are generally stereospecific in their actions. Stereoisomerism in limited not only to organic compounds but also to anorganic compounds, mainly in co-ordination complexes. Stereoisomers include cis and trans isomers, chiral isomers, compound wilth different conformation of chelate rings and other isomers that differ only in the geometry of attachement of the metal ion. Select the correct option for tertraamminedichlorocobalt (III) ion, the trans isomer |
|
Answer» does not have any SYMMETRY 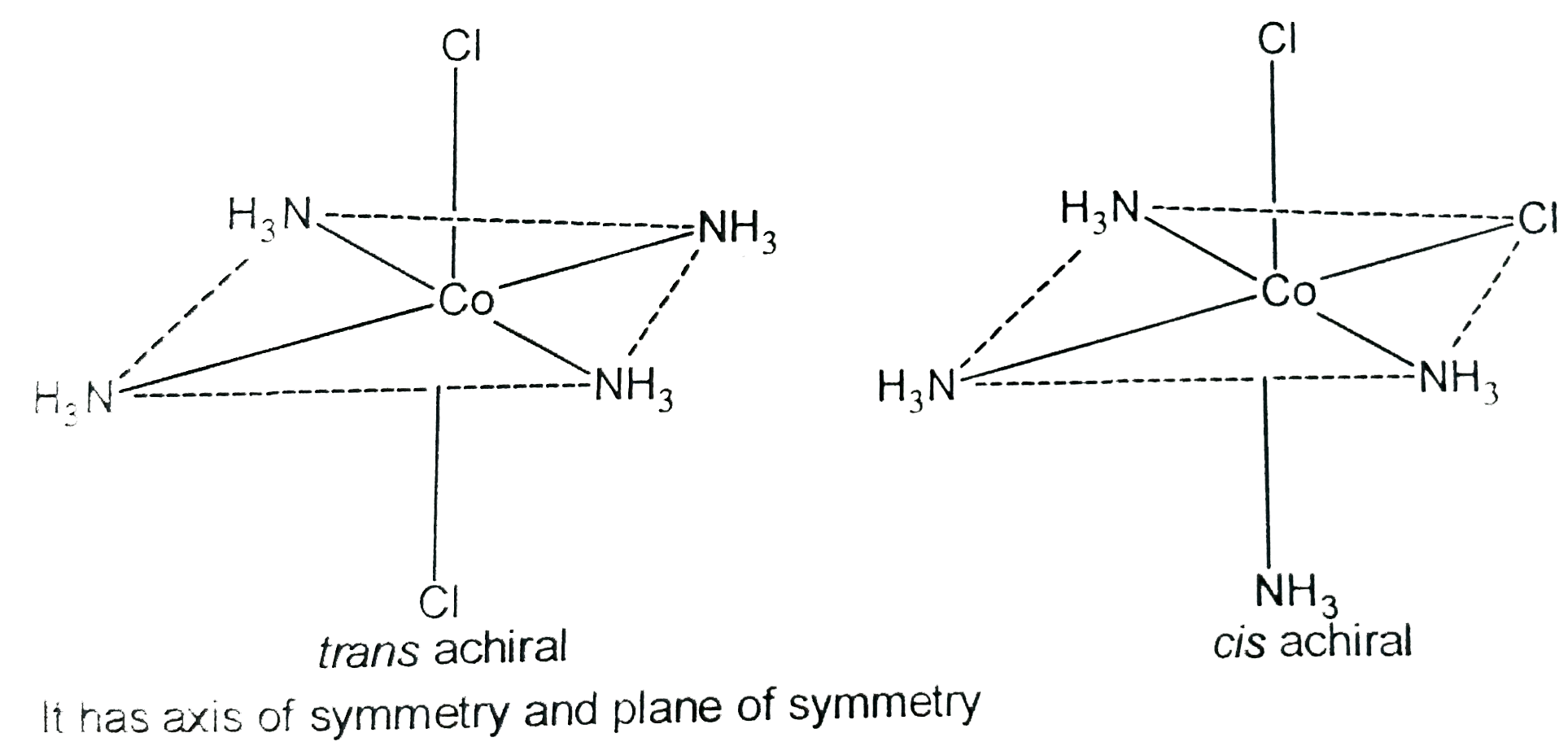 It is has AXIS of symmetyr and plane of symmetry. |
|
| 33. |
Stereoisomers (Geometrical or opticals ) which are neither superimposable nor mirror image to each other are called: |
|
Answer» Enantiomers |
|
| 34. |
Stereoisomerism arises on account of different positions and arrangement of lignads in space around the metal ion . These are classified into two types : (i) Geometrical isomerism is exhibited by octahedral and square planar complexes . (ii) Optical isomerism is shown when mirror images are non superimposable. Both geometrical and optical isomerism are shown by |
|
Answer» `[Co(en)_(2)Cl_(2)]^(+)` |
|
| 35. |
Stereoisomerism arises on account of different positions and arrangement of lignads in space around the metal ion . These are classified into two types : (i) Geometrical isomerism is exhibited by octahedral and square planar complexes . (ii) Optical isomerism is shown when mirror images are non superimposable. The total number of possible isomers for the complex compound [Cu(NH_(3))_(4)][PtCl_(4)] are : |
|
Answer» 3 |
|
| 36. |
Stereoisomerism arises on account of different positions and arrangement of lignads in space around the metal ion . These are classified into two types : (i) Geometrical isomerism is exhibited by octahedral and square planar complexes . (ii) Optical isomerism is shown when mirror images are non superimposable. Choose the correct answer : 11. Which of the following will not show geometrical isomerism ? |
|
Answer» `[Cr(NH_(3))_(4)Cl_(2)]Cl` |
|
| 37. |
Stereoisomerism arises on account of different positions and arrangement of lignads in space around the metal ion . These are classified into two types : (i) Geometrical isomerism is exhibited by octahedral and square planar complexes . (ii) Optical isomerism is shown when mirror images are non superimposable. The compound(s) that exhibi(s) geometrical isomerism is(are) : |
|
Answer» `[Pt(en)Cl_(2)]` |
|
| 38. |
Stephen's reduction convert |
|
Answer» ACYL CHLORIDE to ALDEHYDE |
|
| 39. |
Stephen's reaction is reduction of : |
|
Answer» ALKYL cyanide with` LiAIH_4` |
|
| 40. |
Stephen's reaction is used to prepare aldehydes from: |
|
Answer» Alcohol |
|
| 41. |
Step C (refining) involved in purification of Pb metal: |
|
Answer» |
|
| 42. |
Steels are generally prepared by----------- method |
|
Answer» COMPRESSED METHOD |
|
| 43. |
Steel is an alloy of _______ |
|
Answer» IRON and carbon |
|
| 44. |
Steel that is resistant to acids is : |
|
Answer» CARBON steel |
|
| 45. |
Steel contains carbon |
|
Answer» 0.12% to 0.25% |
|
| 46. |
Steel containing nickel is used in .......... |
|
Answer» AGRICULTURAL tools |
|
| 48. |
Steel becomes soft and pliable by |
|
Answer» Annealing |
|
| 49. |
Steam is passed over hot coke to give water gas. Which substance set as reductant? |
|
Answer» Solution :COKE removes OXYGEN from water to give hydrogen. `H_(2)O + C rarr H_(2) + CO` SUBSTANCE that removes oxygen is reductant. Here, coke is reductant. |
|