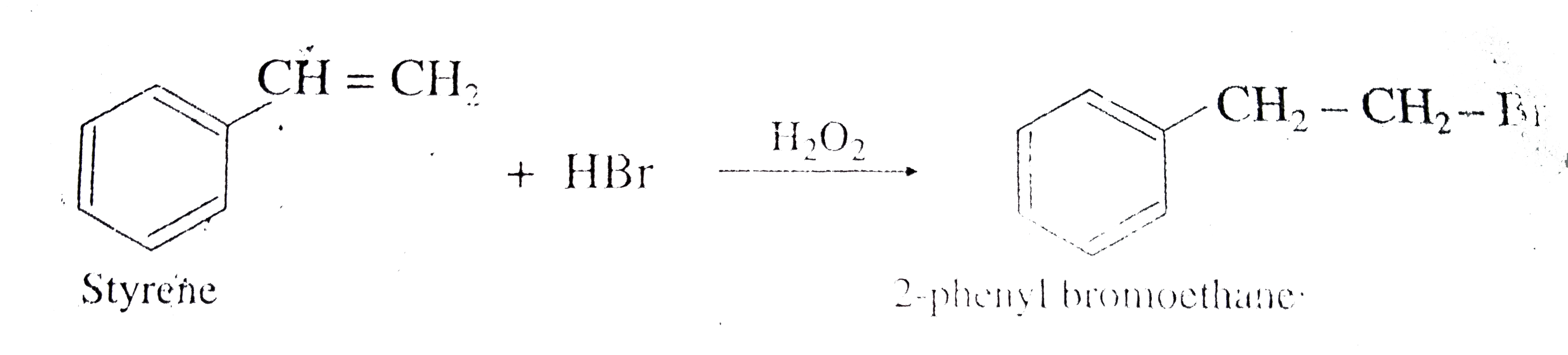Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Substances used in bringing down the body temperature in high fever are called |
|
Answer» Antiseptics |
|
| 3. |
Substances used in bringing down the body temperature in high fevers are called : |
|
Answer» analgesics |
|
| 4. |
Substances in which all the electron spins are paired are ul"termed as paramagnetic." |
| Answer» SOLUTION :TERMED as DIAMAGNETIC | |
| 5. |
{:("Substance","Oxidation state of N"),("A) "HNO_(3),"1) "-3","+5),("B) "NH_(4)NO_(3),"2) "-1//3),("C) "N_(3)H,"3) "+5),("D) "H_(3)PO_(3),"d) "+3),(,"5) "+1//3):} The correct match is |
|
Answer» `{:(A,B,C,D),(3,1,2,4):}` |
|
| 6. |
Substance which absorbs CO_2and violently reacts with H_2O with sound is: |
|
Answer» `CaCO_3` |
|
| 9. |
Substance used in Home's signal is : |
|
Answer» `NH_(3) & C_2H_(2)` `Ca_(3) P_2 +6H_(2)O rarr 3Ca(OH)_2+2PH_3` `CaC_2+H_2O rarr CAO +C_(2)H_2`. `PH_(3)+C_(2)H_2` - are used in Holme.s signal. |
|
| 10. |
Substance used in Holme's signal is: |
|
Answer» `NH_3` |
|
| 11. |
Substance used in glazing pottery is: |
|
Answer» `ZNO` |
|
| 12. |
Substance used for the preservation of coloured fruit juices is |
|
Answer» benzene |
|
| 13. |
Substance used for bringing down temperature in high fever are called |
|
Answer» Antibiotics |
|
| 14. |
Substance A_(2)B(g) can undergoes decomposition to form two set of products : If the molar ratio of A_(2)(g) to A(g) is 5 : 3 in a set of product gases, then the energy involved in the decomposition of 1 mole of A_(2)B(g) is : |
|
Answer» `48.75kJ//mol` |
|
| 15. |
Substance containing an asymmetric carbon atom is: |
| Answer» Answer :D | |
| 16. |
Sublimation method can be used for the purification of : |
|
Answer» Naphthalene |
|
| 17. |
Substance containing an asymmetric carbon atom |
| Answer» Answer :D | |
| 18. |
Substance (A) is a colourless pungent smelling gas of vapour density 8.5. It turned Nessler's reagent brown. (A) on oxidation at high temperature with platinum catalyst gave a colourless gas (B), which readily turned brown in air, forming a gas (C). (B) and (C) were condensed together to give substance (D), which reacted with water to form an acid (E). When (E) was heated with a solution of NH_(4)Cl, a stable gas (F) was evolved. (F) did not support combustion, but magnesium continued to burn in it. Itentify substances (A) to (F) and explain all the reactions involved. |
|
Answer» Solution :(i) `underset(("A"))underset("Ammonia")(NH_(3)) + underset("Nessler's reagent")(2K_(2)Hgl_(4)) + 3 KOH rarr underset(("Brown ppt."))underset("Iodide of Million's base")(H_(2)N.Hg.O.HGL) + 7 KI + 2 H_(2)O` (ii) `underset("Ammonia (A)")(4 NH_(3)) + 5 O_(2) overset("Pt. Heat")rarr underset("Nitric OXIDE (B)")(4 NO) + 6 H_(2)O` (iii) `2 NO + O_(2) rarr underset(("Brown"))underset("Nitrogen dioxide (C)")(2 NO_(2))` (iv) `NO + NO_(2) overset("Condensed")rarr underset("Dinitrogen trioxide (D)")(N_(2)O_(3))` (v) `N_(2)O_(3) + H_(2) O rarr underset("Nitrous acid (E)")(2 HNO_(2))` (VI) `HNO_(2) + NH_(4)Cl rarr underset("Nitrogen (F)")(N_(2)) + 2 H_(2)O + HCl` (vii) `underset("Magnesium")(Mg) + N_(2) overset("Heat")rarr underset("Magnesium nitride")(Mg_(3)N_(2))` |
|
| 19. |
SubstanceA overset(Cu//573K)to isobutlyene, which is structural formula of substance A in this reaction ? |
|
Answer» `CH_(3)CH_(2)CH_(2)CH_(2)-OH` |
|
| 20. |
Subance used in hyper acidity are known as |
|
Answer» Antixidant |
|
| 21. |
Styrene polymerises to polystyrene when heated in the presence of peroxide initiator. What is the mechanism involved? |
| Answer» SOLUTION :FREE RADICAL POLYMERISATION. | |
| 22. |
Styrene to benzoic acid |
Answer» SOLUTION :
|
|
| 23. |
Styrene polymerises to polystyrene when heated in the presence of peroxide initiator. How does the chain propogate in the above mechanism? |
Answer» SOLUTION : Propagation step  The stabilized radical attacks another monomer molecule to GIVE an ELONGATED radical.  CHAIN growth will CONTINUE with the successive addition of several thousands of monomer units. |
|
| 24. |
Styrene polymerises to polystyrene when heated in the presence of peroxide initiator. How is the above chain reaction terminated? |
Answer» Solution :The above CHAIN reaction can be stopped by STOPPING the SUPPLY of monomer or by coupling of two chains or reaction with an impurity such as OXYGEN. 
|
|
| 25. |
Styrene polymerises to polystyrene when heated in the presence of peroxide initiator. Give the initiation step for the above process. |
Answer» Solution :styrenepolymerises to polystyrene when it is heated to ionic with a peroxide initiator. The MECHANISM INVOLVES the FOLLOWING STEPS. 
|
|
| 27. |
Styrene on hydroboration oxidation gives |
|
Answer» 2-phenyl ethan-1-ol |
|
| 28. |
Styrene can be purified by: |
|
Answer» SIMPLE DISTILLATION |
|
| 29. |
Study the two photochemical reactions and answer the questions given below: For the overall reaction between A and B to yield C and D two mechanisms are proposed: (I) A+B rarr AB* rarr C+D,k_(1)=1xx10^(-5)M^(-1)s^(-1) (II) A rarr A* rarr E,k_(1)=1xx10^(-4)M^(-1)s^(-1) E+Brarr C+D,k_(2)=1xx10^(10)M^(-1) s^(-1) (species with * are short-lived) Rate according to mechanism II when concentration of each reactant is 1 M will be: |
|
Answer» `1xx10^(-4) M s^(-1)` |
|
| 30. |
Study the two photochemical reactions and answer the questions given below: For the overall reaction between A and B to yield C and D two mechanisms are proposed: (I) A+B rarr AB* rarr C+D,k_(1)=1xx10^(-5)M^(-1)s^(-1) (II) A rarr A* rarr E,k_(1)=1xx10^(-4)M^(-1)s^(-1) E+Brarr C+D,k_(2)=1xx10^(10)M^(-1) s^(-1) (species with * are short-lived) At what concentration of B, rates of two mechanism are equal? |
|
Answer» 1 M |
|
| 31. |
Study the two photochemical reactions and answer the questions given below: For the overall reaction between A and B to yield C and D two mechanisms are proposed: (I) A+B rarr AB* rarr C+D,k_(1)=1xx10^(-5)M^(-1)s^(-1) (II) A rarr A* rarr E,k_(1)=1xx10^(-4)M^(-1)s^(-1) E+Brarr C+D,k_(2)=1xx10^(10)M^(-1) s^(-1) (species with * are short-lived) Rate according to mechanism I when concentration of each reactant is 0.1 M will be: |
|
Answer» `1XX10^(-7)M s^(-1)` |
|
| 32. |
Study the reactionscarefully. (i) HOCl +H_(2)O_(2) to H_(3)O^(+) +Cl^(-)+O^(2) (ii) PbS +4H_(2)O_(2) to PbSO_(4) + 4H_(2)O Mark the correct option. |
|
Answer» In (i), HOCL is REDUCED and in (II) PBS is oxidised. 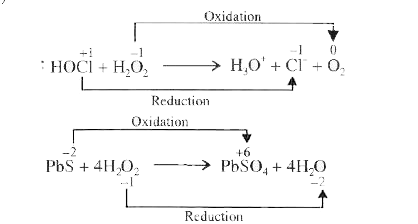
|
|
| 33. |
Study the parallel first order reaction: Aoverset(k_(1))to2B 2Aoverset(k_(2))to3C At any time (tne0) the products contain 40% B and 60% C, by moles .If the overall rate constant for the reaction of A is 0.09min^(-1) ,then value of K_(1) is : |
|
Answer» `0.036 MIN^(-1)` |
|
| 34. |
Study the given graphs and select the correct statement regarding them. |
|
Answer» GRAPH (A) represents physisorption isobar. |
|
| 35. |
Study the following table: {:("Compound","Mass of the compound"),(CO_(2)(44),4.4),(NO_(2)(46),2.3),(H_(2)O_(2)(34),6.8),(SO_(2)(64),1.6):} Which two compounds have least mass of oxygen? (Molercular masses of compounds are given in brackets. |
|
Answer» II nd II II. Mass of oxygen PRESENT `=(2.3)/(46)xx32=1.6g` III. Mass of oxygen present`=(6.8)/(34)xx32=6.4g` IV. Mass of oxygen present `=(1.6)/(64)xx32=0.8g` `therefore` II and IV have least mass of oxygen. |
|
| 36. |
Study the following sequence of reaction and identify the product (Y). CH_(3)CHO +HCHO overset("dil."NaOH)underset("heat") to X overset(HCN) underset(H_(3)O^(+)) to Y |
|
Answer» `CH_(2)=CH-underset(OH)underset(|)(CH)-COOH` |
|
| 37. |
Study the following reactions and mark the appropriate choice. (A) + C_(2)H_(5)OH to (B)+(C ) (C ) +HOH overset(H^(+)) to (B)+(D) (D) overset([O]) to (B) (B) +Ca(OH)_(2) to "Calcium salt"+H_(2)O overset("dry distillation")to underset(("Acetone"))(CH_(3)COCH_(3)) |
|
Answer» `{:("(A)","(B)","(C)","(D)"),((CH_(3)CO)_(2)O,CH_(3)COOH,CH_(3)COOC_(2)H_(5),C_(2)H_(5)OH):}` `underset((C))(CH_(3)COOC_(2)H_(5))+HOH overset(H^(+))to underset((B))(CH_(3)COOH)+underset((C))(C_(2)H_(5)OH)` `underset((D))(C_(2)H_(5)OH)overset([O])to underset((B))(CH_(3)COOH)` `underset((B))(2CH_(3)COOH)+Ca(OH)_(2) to underset(underset(("ACETONE"))underset(CH_(3)COCH_(3))(""darr overset(" ")underset(" ")"dry distillation"))((CH_(3)COO)_(2))Ca+H_(2)O` |
|
| 38. |
The major product of the following reaction is: CH_(3)-underset(H)underset(|)overset(CH_(3))overset(|)C-underset(Br)underset(|)(CHCH_(3)) overset(CH_(3)OH) to |
|
Answer» 1. `{:("(A)","(B)","(C)","(D)"),((CH_(3)CO)_(2)O,CH_(3)COOH,CH_(3)COOC_(2)H_(5),C_(2)H_(5)OH):}` `underset((C))(CH_(3)COOC_(2)H_(5))+HOH overset(H^(+))to underset((B))(CH_(3)COOH)+underset((C))(C_(2)H_(5)OH)` `underset((D))(C_(2)H_(5)OH)overset([O])to underset((B))(CH_(3)COOH)` `underset((B))(2CH_(3)COOH)+CA(OH)_(2) to underset(underset(("Acetone"))underset(CH_(3)COCH_(3))(""darr overset(" ")underset(" ")"dry distillation"))((CH_(3)COO)_(2))Ca+H_(2)O` |
|
| 39. |
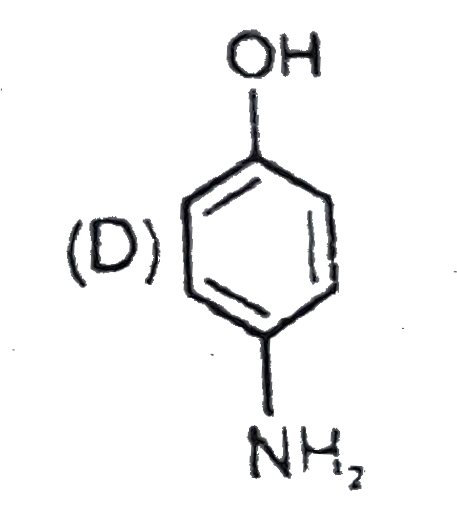
Study the following reactions and identify the reactant 'R'. It can be |
|
Answer»
|
|
| 40. |
Study the following experiment and answer the question at the end of it The following reactions were studied at 25^(@) C in benzene solution containing 0.10 M pyridine CH_(3)OH+(C_(6)H_(5))_(3)"CCl" rarr (C_(6)H_(5))_(3)C.OCH_(3)+HCI ABC The following sets of data were observed: {:("Set","Initial concentration",,"Time different",,"Final concentration[C]"),(,[A],[B]_(0),,,),("I",0.10M,0.05M,25 min,,0.0033 M),("II",0.10M,0.10 M,15 min,,0.0039 M),("III",0.20M,0.10M,7.5 min,,0.0077M):} Rate constant of the above experiment is: |
|
Answer» `1.3xx10^(-1)` |
|
| 41. |
Study the following figure and choose the correct options from the given below (assuming both dissociate completely). |
|
Answer» There will be no movement from any solution across the membrane |
|
| 42. |
Study the following experiment and answer the question at the end of it The following reactions were studied at 25^(@) C in benzene solution containing 0.10 M pyridine CH_(3)OH+(C_(6)H_(5))_(3)"CCl" rarr (C_(6)H_(5))_(3)C.OCH_(3)+HCI ABC The following sets of data were observed: {:("Set","Initial concentration",,"Time different",,"Final concentration[C]"),(,[A],[B]_(0),,,),("I",0.10M,0.05M,25 min,,0.0033 M),("II",0.10M,0.10 M,15 min,,0.0039 M),("III",0.20M,0.10M,7.5 min,,0.0077M):} Rate law of the above experiment is: |
|
Answer» `r=k[A][B]` |
|
| 43. |
Study the following experiment and answer the question at the end of it The following reactions were studied at 25^(@) C in benzene solution containing 0.10 M pyridine CH_(3)OH+(C_(6)H_(5))_(3)"CCl" rarr (C_(6)H_(5))_(3)C.OCH_(3)+HCI ABC The following sets of data were observed: {:("Set","Initial concentration",,"Time different",,"Final concentration[C]"),(,[A],[B]_(0),,,),("I",0.10M,0.05M,25 min,,0.0033 M),("II",0.10M,0.10 M,15 min,,0.0039 M),("III",0.20M,0.10M,7.5 min,,0.0077M):} Rates (d[C])/(dt) in sets I, II and III are respectively (in M min^(-1)) |
|
Answer» `{:("I",,"II",,"III"),(1.30xx10^(-4),,2.6xx10^(-4),,1.02xx10^(-3)):}` |
|
| 44. |
Study sequence of thereactions above and answer the following questions . The reagent 'A' is |
|
Answer» PCC |
|
| 45. |
Study of actinide elements is difficult. Give two reasons. |
|
Answer» SOLUTION :1. They are RADIOACTIVE elements. 2. They have LONG half - life. |
|
| 46. |
Study sequence of thereactions above and answer the following questions . The function group present in the product 'X' is |
|
Answer» ANHYDRIDE 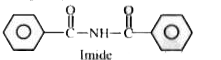
|
|
| 48. |
Study of solar spectrum revealed the presence of |
|
Answer» `H_2` and He |
|
| 49. |
Study following mecahnism of haloform reaction Which step products most acidic compound |
|
Answer» a |
|
| 50. |
Strutures of products , Starting materials or reagents |
Answer» SOLUTION :
|
|