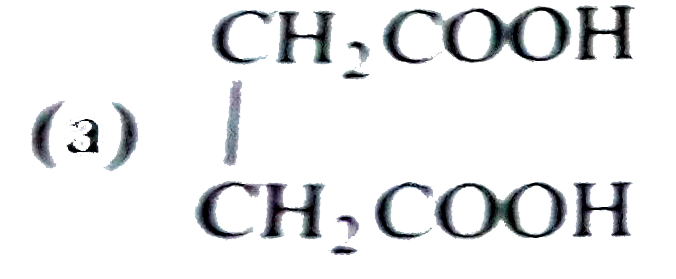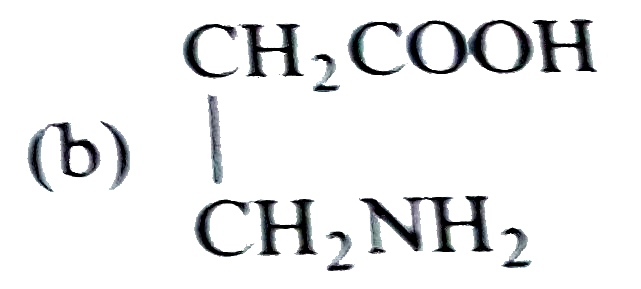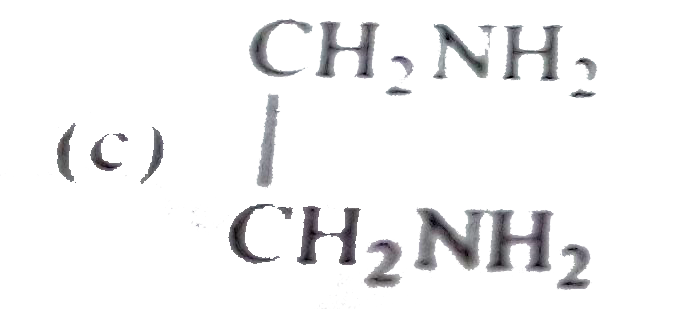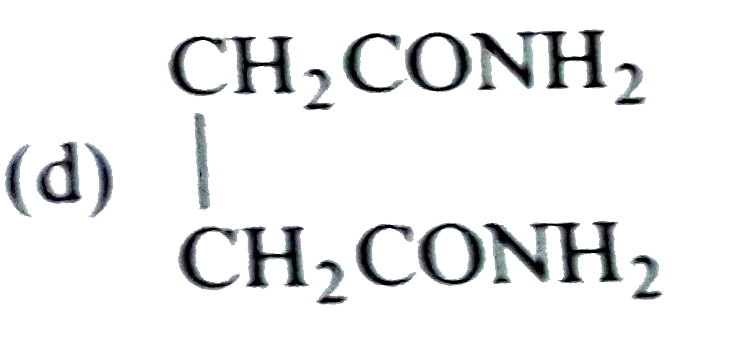Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sucrose is not a reducing sugar since |
|
Answer» It is chemically stable |
|
| 2. |
Sucrose is dextrorotatory but the mixture obtanied after hydrolysis is laevorotatory. Explain. |
| Answer» SOLUTION :Sucrose is dextrorotatory, with a specific rotation of `[alpha]_(D)=+66.5^@` but on hydrolysis , it GIVE EQUIMOLAR amounts of glucose which is dextrorotatory and FRUCTOSE which is laevorotatory. Since the laevorotation of fructose `(-92.4^@)` is much more than dextrorotation of glucose `(+52.5^@)` , therefore , the resulting SOLUTIONS becomes laevorotatory. | |
| 3. |
Sucrose is not |
|
Answer» a di-saccharide |
|
| 4. |
Sucrose is made up of : |
|
Answer» GLUCOPYRANOSE and fructopyranose |
|
| 5. |
Sucroseis dextroroatorybut the mixtureobtainedafter hydrolysis is lavorotatory . Explain. |
| Answer» Solution :On hydrolysis sucrosegives GLUCOSE WHICHIS dextrorotatory withspecific rotation of `+52.5^(@)` and fructosewhichis laevorotorywithspecific rotation`- 92.4^(@)` . As laevorationof fructoseis greaterthan the dextrorotationof glucose , the MIXTUREIS laveorotatory. | |
| 7. |
Sucrose is a non reducing sugar. Justify. |
| Answer» Solution :In sucrose, `C_(1)` of `ALPHA`- D-glucose is jioned to `C_(2)` of -D-fructose. The glycosidic bond THUS FORMED is called `alpha`-1,2 glycosidic bond , Since, the both the carbonyl carbons (reducin groups) are invoved in the glycosidic bonding . Sucrose is a nonreducing sugar. | |
| 8. |
Sucrose is commonly known as _____. |
| Answer» Solution :table sugar | |
| 9. |
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law, witht_(1//2)=3.00 hours. What fraction of the sample of sucrose remains after 8 hours? |
|
Answer» Solution :First order rate equation is : `k=(2.303)/(t)"log"([A]_(0))/([A])""…(i)` As `t_(1//2)=3.0` hours `therefore k=(0.693)/(t_(1//2))=(0.693)/(3H)=0.231h^(-1)` SUBSTITUTING the values in equation(i), we GET `0.231 H^(-1)=(2.303)/(8h)"log"([A]_(0))/([A]) or "log"([A]_(0))/([A])=0.8024` or `([A]_(0))/([A])="Antilog "(0.8024)=6.345 " or"([A])/([A]_(0))=(1)/(6.345)=0.158`. `([A])/([A]_(0))` represents the fraction of SUCROSE left after 8 hours. |
|
| 10. |
Sucrose hydrolyses in presence of enzyme invertase, to give glucose and fructose. However, this reaction does not occur in presence of maltase , because- |
|
Answer» in presence of maltase, the ENERGY of ACTIVATION of the reaction increases |
|
| 11. |
Sucrose decomposes in acid solution into glucose and fructose according to the first order rate law with t_(1//2)=3.00 hours. What fraction of the sample of sucrose remains after 8 hours ? |
|
Answer» Solution :As sucrose decomposes ACCORDING to first order RATE law, `k=(2.303)/(t)log""([A]_(0))/([A])` The aim is to find `[A]//[A]_(0)` As `t_(1//2)=3.0" hour,":.k=(0.693)/(t_(1//2))=(0.693)/(3" hr")=0.231" hr"^(-1)` HENCE, `0.231" hr"^(-1)=(2.303)/(8" hr")log""([A]_(0))/([A])" or "log""([A]_(0))/([A])=0.8024" or "([A]_(0))/([A])=" Antilog "(0.8024)=6.345` or `([A])/([A]_(0))=(1)/(6.345)=0.158.` |
|
| 13. |
Sucrose decompose in acid solution into glucose andfructose according to the first order rate law, with t_(1/2)=3.00 hours. What fraction of sample of sucrose remains after 8 hours ? |
|
Answer» 1.158M |
|
| 14. |
Sucrose contains which of the following groups |
| Answer» Answer :D | |
| 15. |
Sucrosecontain |
|
Answer» 1-2 `alpha-beta`-ACETAL BOND |
|
| 16. |
Sucrose (cane sugar) is a disaccharide. One molecule of sucrose on hydrolysis gives .......... |
|
Answer» 2 MOLECULES of glucose |
|
| 17. |
Sucrose (canc sugar) is a disaccharide. One molecule of sucrose on hydrolysis gives………………… |
|
Answer» 2 MOLECULES of glucose |
|
| 18. |
Sucrose contains glucose and fructose linked by |
|
Answer» `C_(1)-C_(1)` |
|
| 19. |
Sucrose (can sugar) is a disaccharide. One molecule of sucrose on hydrolysis gives. |
|
Answer» 2 molecules of GLUCOSE |
|
| 20. |
Sucrose and lactose are diferentiatedby : |
|
Answer» Tollen's reagent |
|
| 21. |
Sucroseandmaltoseare disaccharidesbutsucrosein a nonreducingsugar while maltoseis a reducingsugar . Givereason . |
|
Answer» Solution :(i)Sucrrose isa disaccharide that composed of `alpha ` -D glucose and `beta` - Dfrucotose. In sucrose C1 of`alpha ` - Dglucoseis joinedto C2 of D - FRUCTOSE . The glycosidicc bondthus formedis CALLED `alpha` -1,2- glycosidicbond . Since boththe carbonlycarbons(reducingcarbons) are involvedin thhe glycosidic bonding , sucroseis a non - reduccingsugar. (ii) Butmaltosecontianstwomolecules ofx - Dglucoseunitsthat are linked by an `alpha -1, 4`glycosidic bond . Anomericcarbon of one unitand C- 4 of otherunit are CONNECTED together . Sinceone of theglucose has the carbonlygroupintact it actsas reducingsugar. |
|
| 22. |
Sucorse hydrolyses readily in acids to give |
|
Answer» TWO MOLECULES of GLUCOSE |
|
| 24. |
Succinimide when subjected to Hofmann bromamide reaction gives a compound having one of the given structures. Select the correct structure: |
|
Answer»
|
|
| 25. |
Successive ionization enthalpies (in eV/atom) of on element are 5,8,9,90,100. The number of valence electrons are: |
| Answer» Solution :NUMBER of valence ELECTRON =3 as DIFFERENCE between `IE_(3) & IE_(4)` is very high. | |
| 26. |
Succinaldehyde on reaction with P_(2)O_(5)0 produces organic compound x, x is having number of pi electrons. |
|
Answer» |
|
| 27. |
Successive ionisation energies (I.E.) for an element A are as A overset(I.E_(1))toA^(+) overset(I.E_(2))toA^(2+) overset(I.E_(3))toA^(3+) If I.E_(1) and I.E_(3) are 27 kJ/mol and 51 kJ/mol respectively, then the value of I.E_(2) is |
|
Answer» 63 |
|
| 28. |
Successive emission of an alpha - particle and two beta- particles by an atom of an element results in the formation of its : |
|
Answer» Isodiapher |
|
| 29. |
Substrate is bound with ........ of enzyme. |
| Answer» Solution :active site | |
| 30. |
Successive emission of an alpha-particle and two beta -particle by an atom of an element results in the formation of its |
|
Answer» Isobar |
|
| 31. |
Subunits present in haemoglobin are : |
|
Answer» 2 |
|
| 32. |
Substrate E2 elimination SN2 — substitution {:((A)CH_(3)-CH_(2)-Br,(P)1,(W)~~0),((B)(CH_(3))_(2)CH-Br,(Q)80,(X)20),((C)(CH_(3))^(3)CBr,(R)100,(Y)90):} |
|
Answer» <P> |
|
| 33. |
Substitution reactions of alkyl halide are initiated by, |
|
Answer» ELECTROPHILE |
|
| 34. |
Substitution of one or more hydrogen atom(s) from a hydrocarbon by another atom or a group of atoms result in the formation of an entirely new compound having altogether different properties and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by -OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spirit used for polishing wooden furniture is chiefly a compound containing hydroxyl group, ethanol. The sugar we eat, the cotton used for fabrics, the paper we use for writing, are all made up of compounds containing -OH groups. Just think of life without paper, no note-books, books, newspapers, currency notes, cheques, certificates, etc. The magazines carrying beautiful photographs and interesting stories would disappear from our life. It would have been really a different world. An alcohol contains one or more hydroxyl (OH) group(s) directly attached to carbon atom(s), of an aliphatic system (CH_(3)OH) while a phenol contains -OH group(s) directly attached to carbon atom(s) of an aromatic system (C_(6)H_(5)OH). Give the name of next two homologoues of CH_(3)OH. |
| Answer» SOLUTION :`C_(2)H_(5)OH" (Ethanol) and "C_(3)H_(7)OH" (PROPANOL)"`. | |
| 35. |
Substitution of one or more hydrogen atom(s) from a hydrocarbon by another atom or a group of atoms result in the formation of an entirely new compound having altogether different properties and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by -OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spirit used for polishing wooden furniture is chiefly a compound containing hydroxyl group, ethanol. The sugar we eat, the cotton used for fabrics, the paper we use for writing, are all made up of compounds containing -OH groups. Just think of life without paper, no note-books, books, newspapers, currency notes, cheques, certificates, etc. The magazines carrying beautiful photographs and interesting stories would disappear from our life. It would have been really a different world. An alcohol contains one or more hydroxyl (OH) group(s) directly attached to carbon atom(s), of an aliphatic system (CH_(3)OH) while a phenol contains -OH group(s) directly attached to carbon atom(s) of an aromatic system (C_(6)H_(5)OH). Write the structure of a ring compound containing the hydroxyl group but not directly linked to the ring. |
Answer» SOLUTION : BENZYL ALCOHOL. BENZYL ALCOHOL.
|
|
| 36. |
Substitution of one or more hydrogen atom(s) from a hydrocarbon by another atom or a group of atoms result in the formation of an entirely new compound having altogether different properties and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by -OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spirit used for polishing wooden furniture is chiefly a compound containing hydroxyl group, ethanol. The sugar we eat, the cotton used for fabrics, the paper we use for writing, are all made up of compounds containing -OH groups. Just think of life without paper, no note-books, books, newspapers, currency notes, cheques, certificates, etc. The magazines carrying beautiful photographs and interesting stories would disappear from our life. It would have been really a different world. An alcohol contains one or more hydroxyl (OH) group(s) directly attached to carbon atom(s), of an aliphatic system (CH_(3)OH) while a phenol contains -OH group(s) directly attached to carbon atom(s) of an aromatic system (C_(6)H_(5)OH). What is the name and formula of the compound used to polish wooden furniture ? |
| Answer» SOLUTION :`C_(2)H_(5)OH,` Ethanol. | |
| 37. |
Substitution reactions may be: |
|
Answer» FREE RADICAL substitution |
|
| 38. |
Substitution of one or more hydrogen atom(s) from a hydrocarbon by another atom or a group of atoms result in the formation of an entirely new compound having altogether different properties and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by -OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spirit used for polishing wooden furniture is chiefly a compound containing hydroxyl group, ethanol. The sugar we eat, the cotton used for fabrics, the paper we use for writing, are all made up of compounds containing -OH groups. Just think of life without paper, no note-books, books, newspapers, currency notes, cheques, certificates, etc. The magazines carrying beautiful photographs and interesting stories would disappear from our life. It would have been really a different world. An alcohol contains one or more hydroxyl (OH) group(s) directly attached to carbon atom(s), of an aliphatic system (CH_(3)OH) while a phenol contains -OH group(s) directly attached to carbon atom(s) of an aromatic system (C_(6)H_(5)OH). Give some examples of compounds of daily use which contain hydroxyl group. |
| Answer» SOLUTION :SUGAR, COTTON and PAPER. | |
| 39. |
Substitution of one or more hydrogen atom(s) from a hydrocarbon by another atom or a group of atoms result in the formation of an entirely new compound having altogether different properties and applications. Alcohols and phenols are formed when a hydrogen atom in a hydrocarbon, aliphatic and aromatic respectively, is replaced by -OH group. These classes of compounds find wide applications in industry as well as in day-to-day life. For instance, have you ever noticed that ordinary spirit used for polishing wooden furniture is chiefly a compound containing hydroxyl group, ethanol. The sugar we eat, the cotton used for fabrics, the paper we use for writing, are all made up of compounds containing -OH groups. Just think of life without paper, no note-books, books, newspapers, currency notes, cheques, certificates, etc. The magazines carrying beautiful photographs and interesting stories would disappear from our life. It would have been really a different world. An alcohol contains one or more hydroxyl (OH) group(s) directly attached to carbon atom(s), of an aliphatic system (CH_(3)OH) while a phenol contains -OH group(s) directly attached to carbon atom(s) of an aromatic system (C_(6)H_(5)OH). How are alcohols and phenols formed ? |
| Answer» Solution :ALCOHOLS and phenols are formed by removing a hydrogen from the ALIPHATIC and AROMATIC hydrocarbon respectively and SUBSTITUTING with a hydroxyl group. | |
| 40. |
Substitution of one alkyl group by replacing hydrogen of primary amines : |
|
Answer» INCREASES the BASE STRENGHT |
|
| 41. |
Substitution can take place with alkyl halides by two different mechanisms, S_(N)1 " and " S_(N)2. |
|
Answer» |
|
| 42. |
Substitution of chlorine takes place readily at higher temperature in |
|
Answer» `CH_(3)-CH=CH_(2)` |
|
| 43. |
Substituent on phenyl ring. {:((A)-CH_(2)-CH_(3),(P) o//p-"directors"),((B)-O-overset(O)overset("||")(S)-CH_(3),(Q) "meta directors"),((C)-NH-overset(O)overset("||")(C)-CH_(3),(R)" Activating group"),((D)-underset(O)underset("||")(S)-CH_(3),(S)" Deactivating group"):} |
|
Answer» <P> |
|
| 44. |
Substances which rotate the plane polarised light are called : |
|
Answer» OPTICALLY active |
|
| 45. |
Substances which relieve body pains are termed as |
|
Answer» ANTIPYRETICS |
|
| 46. |
Substances which have identical chemical properties but differ in atomic weights are called |
|
Answer» Isothermals |
|
| 47. |
Substances which are repelled by the external magnetic field are called |
|
Answer» DIAMAGNETIC |
|
| 48. |
Substances which do not react with cold water but react with steam are : |
|
Answer» `C` ,` CA` , `SO_2` |
|
| 49. |
Substances which bring down the body temperature during fever are known as |
|
Answer» ANTIBIOTICS |
|
| 50. |
Substances used in bringing down the body temperature in high fevers are called: |
| Answer» SOLUTION :ANTIPYRETIC | |
 group
group