Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-1:Potassium chromate solution in acetic acid precipitates only Ba^(2+) as BaCrO_4 in group V^(th) Statement-2:SrCrO_4 and CaCrO_4 are not precipitated with potassium chromate solution in acetic acid. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 2. |
Statement-1:Restricted rotation about a bond is the necessary condition for geometrical isomerism. Statement-2:Two different orientations are possible due to restricted rotation about a bond it the end groups are different . |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 3. |
Statement-1:Reddish brown gas, NO_2 when passed through alkaline gives nitrite only. Statement-2:NO_2 is acidic in nature and is a mixed anhydride of nitric acid |
|
Answer» Statement-1 and statement-2 are CORRECT andstatement-2 is the correct EXPLANATION for Statement-1 |
|
| 4. |
Statement 1:Polybutadiene is an example of step-growth polymer. Statement 2: Co[ |
|
Answer» STATEMENT `1` is true ,statement `2`is true,statement`2`is the correct explanation of statement`1` |
|
| 5. |
Statement-1:Polyacylation in benzene does not occur during Friedal Craft's acylation. Statement-2:As the product ketone is much more reactive than starting material. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 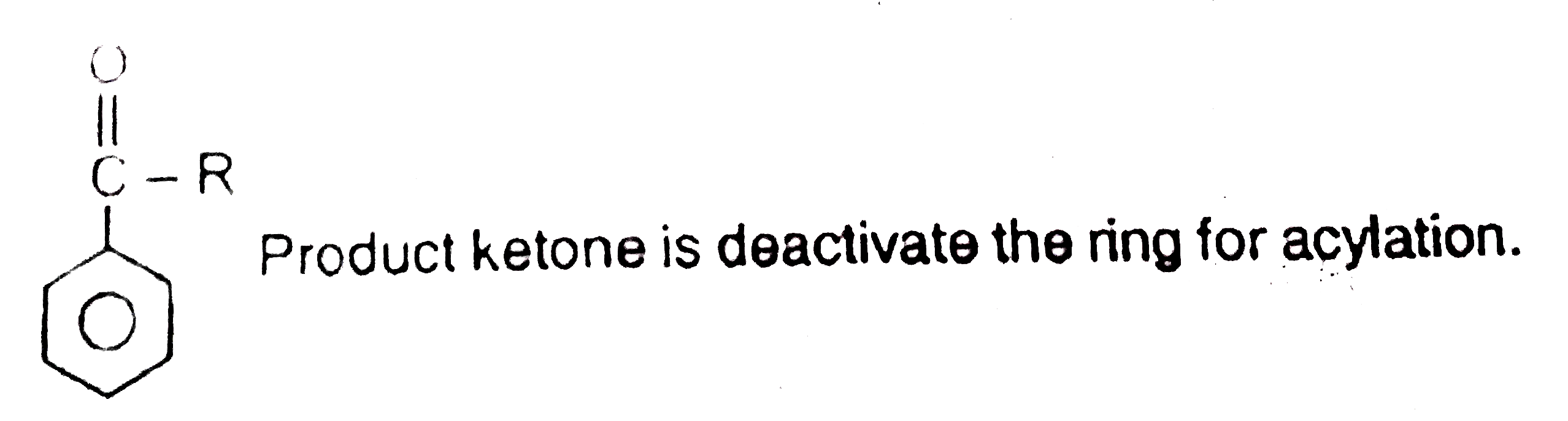
|
|
| 6. |
Statement-1:Pentacetate of glucose does not form oxime on treatment with H_2N-OH Statement-2:Glucose on reaction with acetic anhydride forms pentacetate under suitable conditions. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 |
|
| 7. |
Statement-1:Phenolphathalein can be used as indicator in the titration of weak acid with NaOH. Statement-2 :Near the end point in the titration of weak acid with NaOH, the pH of the solutions is alkaline due to hydrolysis of anion. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 8. |
Statement-1:p-type semiconductor are formed due to metal excess defect. Statement-2: F-centres are created due to metal excess defect. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANATION for statement-1 |
|
| 9. |
Statement-1:p-Hydroxybenzoic acid has a lower boiling point that o-Hydroxybenzoic acid. Statement-2:o-Hydroxybenzoic acid has intramolecular hydrogen bonding. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 |
|
| 10. |
Statement-1:NO^+ and CN^(-) both have same order and magnetism (i.e. magnetic property). Statement-2:NO^+ and CN^(-) are isoelectronic species. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 Number of electrons `CN^(-) =6+7+1=14` Bond ORDER `=(10-4)/2=3` Both are diamagnetic |
|
| 11. |
Statement-1:NO yellow precipitate is formed when an excess of a more concentrated (6M) solution of Kl. Statement-2:Yellow precipitate of Pbl_2 does not dissolve in excess of dilute solution of Kl. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 Reaction is reversible and SHIFTS in backward direction on dilution with water. |
|
| 12. |
Statement-1:Methyl alpha-D-fructofuranoside (I) , undergoes acid catalysed hydrolysis at faster rate than that of methyl alpha-D-glucofuranoside (II). Statement-2:The intermediate carbocation in case of I is more stable than in case of II. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 
|
|
| 13. |
Statement-1L The bond angles in molecules depneds upon hybridization electronagativity of central atom, no. of lone pair, odd electron and multiplicity of bond. Statement-2 NO_(2) and NO_(2)^(-) have angles 134^(@) and 115^(@) respectively. |
|
Answer» If both the STATEMENT are TRUE and Statement -2 is the correct explanation of Statement-1: 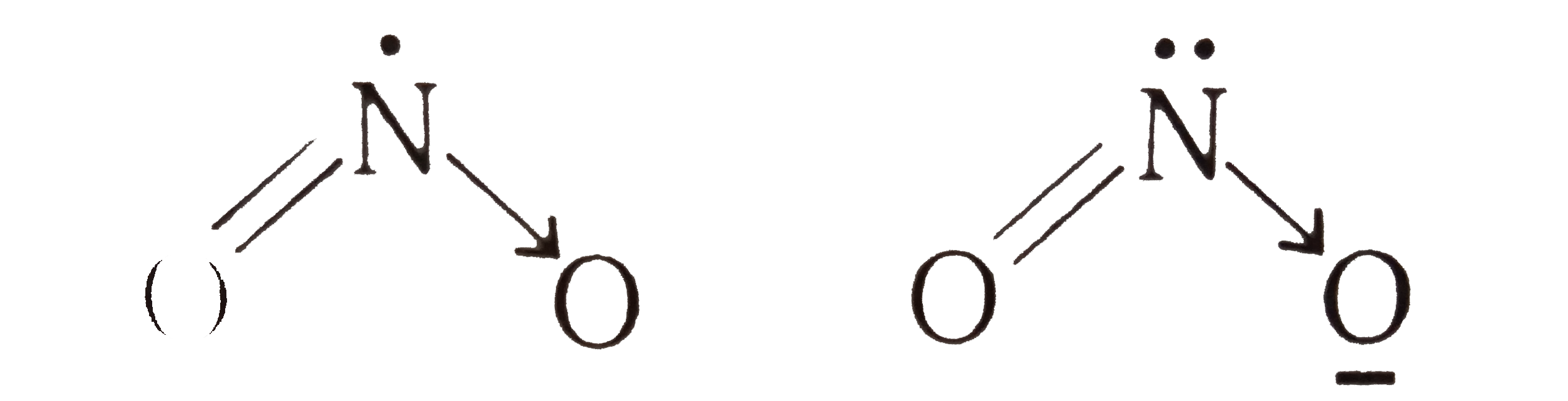
|
|
| 14. |
STATEMENT-1:K_(2)CO_(3) cannot be prepared by Solvay's process. and STATEMENT-2: K_(2)CO_(3) does not decompose on heating |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is a CORRECT explanation for STATEMENT-1 |
|
| 15. |
Statement-1:In natural tris chelating complex, [Co(acac)_3], in each ring the two M-O bonds are equal in length, as are two C-O and the two C-C bonds because. Statement-2:In each ring resonance take place. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 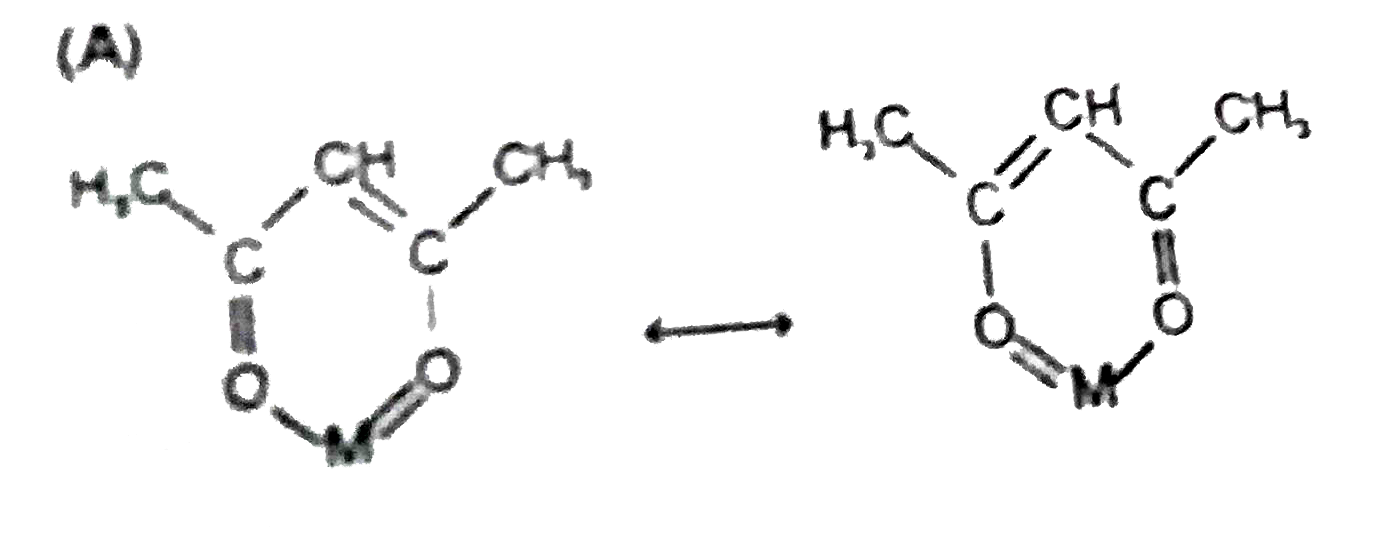
|
|
| 16. |
Statement-1:In the electrolytic reduction of Al_(2)O_(3), cryolite lowes the melting point of the mixture and bring conductivity: Statement-2: Cryolite is an ore of aluminium. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explantion for Statement-1. Statement-2: CRYOLITE is `Na_(2)AlF_(4)` and is ore of aluminium. Therefore, (b) is correct. |
|
| 17. |
Statement-1:In cyanide process for the extraction of gold and silver from their native ores, the cyanide solution acts as a reducing agent to reduce the gold and silver compounds present in the ores, the cyanide solution acts as a reducing agent to reduce the gold and silver compounds present in the ores into the metallic states. Statement-2:In the extraction of gold and silver the cyanide solution acts as complexing reagent in the presence of air and form their respective soluble complexes. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 Statement-2:`4M(s)+8CN^(-)(aq.)+2H_2O(aq)+O_2(g)to overset(+I)(4[M(CN)_2]^(-)(aq)+4OH^(-)(aq).` M=Au or Ag. |
|
| 18. |
Statement-1.If a liquid more volatile than the solvent is added to the solvent, the vapour pressure of the solution may increase, i.e., p_(s)gtp^(@). Statement-2. In the presence of more volatile liquid solute, Raoult's law does not hold good. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a CORRECT explanation of Statement-1. |
|
| 19. |
Statement-1:Hydrolysis of sucrose brings a change in sign of rotation towards plane polarised light. Statement-2:Fructose has specific rotation -92.4^@ and glucose has +52.5^@ |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 |
|
| 20. |
Statement-1:Graphite is used as anode by not diamond. Statement-2: Mobile electrons are present in graphite layer which help in the electrical conductivity. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANTION for Statement-1. |
|
| 21. |
Statement-1:Finely divided iron does not form ferric chloride with hydrochloric acid. Statement-2:Hydrochloric acid produces hydrogen gas with iron. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 Liberation of hydrogen PREVENTS the formation of ferric CHLORIDE. |
|
| 22. |
Statement-1:Extraction of gold from its native ore involves leaching themetal ore with dilute solution of NaCN in presence of air. Statement-2:This is an oxidation reaction and leads to the formation of a soluble complex. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 `Au^+ + 2CN^(-) (aq.) to [Au(CN)_2]^(-)` soluble complex. |
|
| 23. |
Statement-1:Ethers are prepared from alcohols in acid medium through S_(N^1) or S_(N^2) mechanisms depending upon nature of alcohol. Statement-2:ROH+H^(+)hArr R-overset(+)OH_2 |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 |
|
| 24. |
Statement-1:Dipositive zinc exhibits diamagnetism due to loss of two electrons from 3d-orbital of neutral atom. Statement-2:The electronic configuration of zinc (Z=30) is [Ar]^(18)3d^10 4s^2 |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 25. |
Statement-1:Cyclopropane has the highest heat of combustion per methylene group Statement-2:Its potential energy is raised by angle strain. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 26. |
Statement-1:Compounds having -NR_(3)^(+),-SR_(3)^(+) etc. as leaving groups give Hofmann product in E_(2) elimination. Statement-2: E_(2) elimination is a single step reaction. |
|
Answer» If both the STATEMENT are TRUE and Statement -2 is the correct explanation of Statement-1: |
|
| 27. |
Statement-1:Coordination number of cobalt in the complex [Co(en)_(3)]^(3+) is six. Statement-2: Ethylenediamine acts as a bidentate ligand. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 28. |
Statement-1:Cuprous ion (Cu^+) is colourless whereas cupric ion (Cu^(2+))produce blue colour in solution. Statement-2:Cupros ion Cu^+ has unpaired electrons while cupric ion Cu^(2+) does not . |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 29. |
Statement-1:[Co(en)(NO_(2))_(2)(pn)]^(+) ion exists in four geometrical forms : two are cis and two are trans. Statement-2:en (symmetrical ligand) and pn (unsymmetrical ligand) are etylene diamine and 1, 2-diamino propane repectively forming complex of the type M(A A)(AB)a_(2). |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 30. |
Statement-1:Cast iron is different from pig iron. Statement-2: Cast iron is made by melting pig iron with scrap iron and coke using hot air blast and has about 3% carbon content. |
|
Answer» STATEMENT -1 is true, Statement-2 are true and Statement-2 is the CORRECT explanation of Statement -1 |
|
| 31. |
Statement 1:Bromobenzene upon reaction with Br_2//Fe gives 1,4-dibromobenzene as the major product. Statement 2:In bromobenzene , the inductive effect of the bromo group is more dominant than the mesomeric effect in directing the incoming electrophile |
|
Answer» Statement 1 is true , statement 2 is true , statement 2 is a correct EXPLANATION for statement 1 |
|
| 32. |
Statement-1:Bis(dimethylglyoximato)nickel(II) can show geometrical isomerism. Statement-2:Tetrahedral complexes with chiral structures exhibit optical isomerism. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 
|
|
| 33. |
Statement-1:Benzene and ethene both give reactions with electrophillic reagents. Statement-2:Benzene and ethene both have loosely bound pi electrons, which can be donated to vacant orbital of the electrophilie. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 34. |
Statement-1:Beryllium resembles aluminium. Statement-2: Charge/radius ratio of Be^(2+) ion is nearly the same as that of the Al^(3+) ion. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 35. |
Statement-1:A(s)hArrB(g)+C(g),Kp_(1) X(s)hArrB(g)+Y(g) , Kp_2=3Kp_1 Total pressure of B over the mixture of solid A and X is greater than pressure of B either over excess solid A or over excess solid X But less then their directly sumup value when excess of solid A and excess of solid X kept in different container. Statement-2 :In presence of each other, degree of dissociation of both solids decreases. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 36. |
Statement-1:Ar C(CH_3)_3+Br_2overset(AlBr_3)toArBr+(CH_3)_2C=CH_2 but ArCH_2CH_3+Br_2overset(AlBr_3)to o,p-BrC_6H_4CH_2CH_3 Statement-2:In the first case ispo substitution takes place in which Br^(o+)displaces (CH_3)_3C^+, a stable cation but removal of CH_3CH_2 required large amount of energy. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 37. |
Statement-1:Aqueous solution of hydrogen peroxide is kept in glass or metel container containing some urea or phosphoric acid. Statement-2:Urea or phosphoric acid acts asa negative catalyst for the decomposition of hydrogen peroxide. |
|
Answer» Statement-1 and statement-2 are CORRECT andstatement-2 is the correct explanation for Statement-1 |
|
| 38. |
Statement -1:Aluminothermy is used for extraction of chromium from chromium oxide. Statement-2: Aluminium is reducing agent. |
|
Answer» STATEMENT -1 is true, Statement-2 are true and Statement-2 is the correct EXPLANATION of Statement -1 |
|
| 39. |
Statement-1:An element has variable equivalent mass. Because Statement-2: The valency of element is variable. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a correct explanantion for statement-1 |
|
| 40. |
Statement-1:All halogens except F_2 are more reactive than interhalogens. Statement-2: Bond in the interhalogens (X-Y) is weaker than X-X bond in the halogens. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 41. |
Statement 1:Addition of Br_2 to 1-butene gives two optical isomers. Statement 2:The product contains one asymmetric carbon |
|
Answer» Statement 1 is true , statement 2 is true , statement 2 is a correct explanation for statement 1 `underset"1-Butene"(H-undersetundersetH|oversetoversetH|C-undersetundersetH|oversetoversetH|C-oversetoversetH|C=undersetundersetH|oversetoversetH|C)-HBroverset"Peroxide"tounderset"1-Bromo BUTANE"(H-undersetundersetH|oversetoversetH|C-undersetundersetH|oversetoversetH|C-undersetundersetH|oversetoversetH|C-undersetunderset"Br"|oversetoversetH|C-H)` In this reaction anti-Markownikoff's addition is explained on the basis of the fact that in the presence of peroxide the addition TAKES place via a free radical mechanism rather than the ionic mechanism |
|
| 42. |
Statement-1:Alkanes float on the surface of water Statement-2: Density of alkanes is in the range of 0.6-0.9 g/ml , which is lower than water. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 
|
|
| 43. |
Statement-1:Acid catalysed hydrolysis of ester is reversible while base catalysed hydrolysis is irriversible . Statement-2: In acid catalysed ester hydrolysis carboxylic acid is formed on which nucleophilic attack of alcohol is possible but in base catalysed ester hydrolysis carboxylate anion is formed on which nucleophilic attack is not possible. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 44. |
Statement-1:Addition of ammonium chloride to a solution containing ferric and magnesium ions is essential for selective precipitation of ferric hydroxide by aqueous ammonia. Statement-2:The function of NH_4Cl is to supress the ionization of NH_4OH by common ion effect and thus prevents the precipitation of Mg(OH)_2 becuause K_(sp) of Mg(OH)_2 is high. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 45. |
Statement-1:A recemic mixture of 2-chloropropanoic acid is treated with excess of (+)-2-Butanol.The Statement 2: The solution of reaction mixture at time (t=0) , will be dexterorotatory. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 46. |
Statement-1:A solution containing S^(2-) ions gives purple / violet colour with sodium nitroprusside solution in alkaline solution. Statement-2:Sodium sulphide gives black precipitate with silver nitrate solution. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 Statement-2:`Na_2S+2AgNO_3toAg_2S darr` (BLACK) +`2NaNO_3` |
|
| 47. |
Statement-1:Aceteldehyde responds positively with all the test of carbonyl compounds like Tollen's Test. Fehling test, 2,4-DNP test, as well as Iodoform test. Statement-2:All aldehydes respond all the four test given in assertion. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 Statement-2:False, all ALDEHYDES do not GIVE iodoform test. |
|
| 48. |
STATEMENT-1:2,3- dimethylbut -2-ene decolorize Br_2 water. and Statement-2 : 2,3 - dimethylbut -2-ene is an unsaturated compound. |
|
Answer» STATEMENT-1 I true, STATENENT-2 is true, STATEMENT-2 is a correct explanation for STATEMENT-1.  Does not DECOLORIZE `Br_2` water as it does not contain olefinnic H ATOM i.e. all the olefinic H atoms are substitued by `-CH_3` group. No doubt it is an UNSATURATED COMPOUND. |
|
| 49. |
Statement-1:1,3,5-Trihydroxybenzene reacts with NH_2OH to give oxime derivation where a 1,3-Dihydroxybenzene do not. Statement-2:Former exist in predominantly where as later exists in predominantly no substantial keto form is present which can give oxime derivation. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 50. |
Statement-1: Zn displaces Ag from thecomplex Na[Ag(CN)_(2)] to give Na_(2)[Zn(CN)_(4)] Statement-2:Zn is more electro positive than silver |
|
Answer» STATEMENT -1 is TRUE, Statement-2 are true and Statement-2 is the CORRECT explanation of Statement -1 |
|