Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Assertion: 1-Butene on reaction with HBr in the presence of a peroxide produces 1-bromo-butane Reason: It involves the free radical mechanism. |
|
Answer» STATEMENT 1 is true , statement 2 is true , statement 2 is a correct explanation for statement 1 |
|
| 2. |
Statement -1: Zn^(+2) precipitated in the form of ZnS but not in the form Zn(NO_(3))_(2). And Statement -2: S^(-2) ion has high electron density w.r.t. NO_(3)^(-) ion. |
|
Answer» STATEMENT -1 is true, Statement -2 is true, Statement -2 is CORRECT explanation for statement -1. |
|
| 3. |
STATEMENT-1: Zinc does not show characteristic properties of transition metals. and. STATEMENT-2: In zinc outermost shell is completely filled. |
|
Answer» STATEMENT - 1 is TRUE, STATEMENT-2 is True, STATEMENT -2 is CORRECT explanation for STATEMENT-1 |
|
| 4. |
Statement -1: Zinc displaces copper from copper sulphate solution. and Statement 2: The E^(@) is Zn of - 0.76 volt and that of copper is +0.34 volt. |
|
Answer» Statement -1 is true, Statement -2 is true, Statement -2 is correct EXPLANATION for statement -1. |
|
| 5. |
Statement - 1 : Wolframite impurities are separated from cassiterite by electromagnetic separation. And Statement - 2 : Cassiterite being magnetic is attracted by the magnet and forms a separate heap. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is True, Statement - 2 is correct explanation for statement - 1. |
|
| 6. |
Statement-1: When the ligands are stronger, the distribution of six electrons of Co^(3+) ions is d_(xy)^(2),d_(yz)^(2)" and "d_(zx)^(2) Statement-2: t_(2g) orbitals corresponds to d_(xy),d_(yz)" and "d_(z^(2)). |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 7. |
Statement-1: When acidified zinc sulphate solution is electrolysed between zinc electrodes, it is zinc that is deposited at the cathode and no H_(2) gas is evolved. Statement-2: The electrode potential of zinc is more negative than that of hydrogen as the overvoltage for the H_(2) evolution on zinc is quite large. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANATION of statement-1 |
|
| 8. |
STATEMENT-1: Volume required for 0.1 M solution is in order V_(KMnO_(4)) lt V_(K_(2)Cr_(2)O_(7)) lt V_(H_(2)O_(2)) STATEMENT-2: The number of equivalents required will be in order H_(2)O_(2) gt KMnO_(4) gt K_(2)Cr_(2)O_(7) STATEMENT-3: The n_("factor") is in order n_(H_(2)O_(2)) lt n_(KMnO_(4)) lt n_(K_(2)Cr_(2)O_(7) |
|
Answer» T T F |
|
| 9. |
Statement-1: When 2 faraday of electricity is passed through 0.1 MH_(2)SO_(4)(aq),11.2 litre O_(2) evolved at STP. Statement-2: Molecular weight of oxygen is 32. |
|
Answer» Solution :MOLE of `O_(2)xx4=2(H_(2)OtoO_92)+4H^(+)+4e^(-))` Mole of `O_(2)=0.5` VOLUME of `O_(2)` at `S.T.P=11.2Lt`. |
|
| 10. |
STATEMENT-1: VO_(4)^(-3) is coloured due to charge transfer . STATEMENT-2: Colour due to charge transfer is highly intense colour STATEMENT-3: Fe may form Fe-Fe bond |
|
Answer» FTF |
|
| 11. |
Statement -1: Vinylidene chloride forms isotactic. Statement -2: Vinylidene chloride contains chiral carbon atoms |
|
Answer» Statement 1 is true, statement-2 is true , statmenent -2 is a CORRECT explanation for statement -1 Vinylidene chloride does not CONTAIN chiral carbon ATOMS. |
|
| 12. |
Statement-1: [V(CO)_(6)] cannot act as oxidising agent. Statement-2: It cannot be reduced by reducing agent. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 13. |
Statement-1. Vapour pressure of water is less than 1.013 bar at 373K. Statement-2. Water boils at 373 K as the vapour pressure at this temperature becomes equal to atmosphere pressure. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation of Statement-1. |
|
| 14. |
Statement-1: Vapour density of CH_(4) is half of O_(2). Because Statement-2: 1.6 g of CH_(4) contains same number of electrons as 3.2 g of O_(2). Statement-2: Specific gravity is dimensionless quantity. Because Statement-2: Specific gravity. is relative density of a substance, measured with respect to density of water at 4^(@)C? |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANANTION for statement-1 |
|
| 15. |
Statement-1: Unsymmetrical ketone reacts with HCN to give (dil) mixture of carbohydrine. Statement-2: Cyanide ion is amident nucleophile. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-3 |
|
| 16. |
STATEMENT -1 :unlikethe gtC = Ogroupof aldehyesandketones,the gt C = OofR - overset(O)overset(||)(C) - OH does not undergonucleophillicadditionreactions. andSTATEMENT- 2:Carboxylicacidsexistas dimers due tointermolecular hydrongen bonding . |
|
Answer» STATEMENT - 1IS True, STATEMENT- 2 is True, , STATEMENT-2 , isa correct explanationfor STATEMENT- 1 . |
|
| 17. |
Statement-1 : undergoes inversion in DMSO with hydroxide ion Statement-2 : The reaction proceeds by SN_(2) mechanism. |
|
Answer» STATEMENT -I is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-6 |
|
| 18. |
Statement-1. Under identical conditions, 1 L of O_(2) gas an 1 L of O_(3) gas contain the same number of oxygen atoms. Statement-2. 1 L of O_(2) and 1 L of O_(3) contain the same number of moles under identical conditions. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is the correct EXPLANATION for Statement-1. |
|
| 19. |
Statement-1 : Two reversible adiabats can never intersect. Statement-II : Efficiency of carnot engine cannot be one. |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 20. |
Statement-1: Toxic metal ions are removed by the chelating ligands. Statement-2: Chelate complexes tend to be more stable. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 21. |
Statement-1: Triethylenetrtraamine is bidentate monoanion. Statement-2: Complex containing propylenediamine ligand shows ligand isoemrism. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 22. |
Statement-1 : Total number of moles in a closed system at new equilibrium is less than the old equilibrium if some amount of a substance is removed from a system. A(g)hArrB(g) equilibrium. Statement-2 : The number of moles of the substance which is removed, is partially compensated as the system reached to new equilibrium. |
|
Answer» Statement-1 is TRUE, statement-2 is true and statement-2 is CORRECT EXPLANATION for statement-4 |
|
| 23. |
Statement-1. To a solution of potassiumchromate, if a strong acid is added, it changes its colour from yellow to orange. Statement-2. The colour change is due to the change in the oxidation state of potassium chromate. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation of Statement -1 |
|
| 24. |
Statement 1 : To a solution of potassium chromate if a strong acid is added it changes its colour from yellow to orange. Statement 2 : The colour change is due to the oxidation of potassium chromate. |
|
Answer» Statememt 1 is true, statement 2 is true , statement 2 is a correct EXPLANATION for statement 6 There is no CHANGE in OXIDATION state. So the explanation is wrong. |
|
| 25. |
Statement -1: Time taken for the completion of 75% of a 1st order reaction is double that t_(1//2). Statement-2: Time taken for completion of any fraction of 1st order reaction is a constant value. |
|
Answer» Statement-1 is TRUE, statement-2 is True, Statement-2 is a correct explantion for Statement-1. |
|
| 26. |
Statement-1: [Ti(H_(2)O)_(6)]^(3+) is violet in colour while Ti^(3+) is colourless. Statement-2: Light correspondign to dnergy of blue-green region is absorbed by the complex to excite the electron from t_(2g) level to e_(g) level. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 27. |
Statement-1 : There is no change in enthalpy of an ideal gas during compression at constant pressure. Statement-II : Enthalpy of an ideal gas is a function of temperature . |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 28. |
Statement-1 : There is no change in enthalpy of an ideal gas during compression at constant temperautre. Statement-II : Enthalpy of an ideal gas is a function of temperature and pressure. |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 29. |
Statement 1 : There is a natural asymmetry between converting work to heat and converting heat to work. Statement 2 : No porcess is possible in which the sole result is the absorption of heat from a reservoir and its complete conversion into work. |
|
Answer» Statement 1 is true, statement-2 is true, statement 2 is a correct explanation for statement 4 |
|
| 30. |
Statement-1: The value of Delta_(0) for M^(3+) complexes are always much higher than value for M^(2+) complexes (for the same set of ligands) Statement-2: The crystal field stabilization energy of [Co(NH_(3))_(6)]^(3+)lt[Rh(NH_(3))_(6)]^(3+). |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 31. |
STATEMENT-1: The value of Boyle's temperature for real gas is (T_(B) =(a)/(R_(b))) and STATEMENT-2: At Boyle's temperature, T_(B) real gases. behave ideally over long range of pressure. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 32. |
Statement-1: The time of completion of reactions of type A rarr product (order lt 1) may be determined. Statement-2 : Reactions with order ge 1 are either too slow or too fast and hence the time of completion can not be determined. |
| Answer» ANSWER :C | |
| 33. |
Statement 1 : The tin obtained after smelting of black tin is known as block tin. Statement 2 : The tin can be cast into ingots or black. |
|
Answer» STATEMENT 1 is TRUE, statement 2 is true : statement 2 is a correct EXPLANATION for statement 1 |
|
| 34. |
Statement -1 : The two strands of DNA are complementary. Statement- 2: Cytosine always pairs with guanine and thymine pairs with adenine. |
|
Answer» Statement -1 is True , Statement -2 is True, Statement -2 is a CORRECT explanation of Statement -5 |
|
| 35. |
Statement-1 :Thethermal stability of hydrides of carbon family is in order: CH_(4)gtSiH_(4)gtGeH_(4)gtSnH_(4)gtPbH_(4) Statement-2: E-H bond dissociation enthalpies of the hydrides of carbon family decrease down the group with increasing atomic size. |
|
Answer» STATEMENT-1 is TURE, Statement-2 is TRUE,Statement-2 is a correct explanation for Statement-1. |
|
| 36. |
Statement-1 : The SRP of three metallic ions A^+,B^(2+),C^3 are -0.3,-0.5,0.8 volt respectively,so oxidising power of ions is C^(3+)ltA^+ltB^(2+) Statement-2: Higher the SRP, higher the oxidising power. |
|
Answer» STATEMENT-1 is TRUE, statement-2 is true and statement2 is correct explanation for statement-2 |
|
| 37. |
Statement-1: The species [CuCl_(4)]^(2-) exists but [CUI_(4)]^(2-) does not. Statement-2: [NiCl_(2)(PPh_(3))_(2)] have tetrahedral geometry. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 38. |
Statement-1. The standard unit for expressing the mass of atoms in amu. Statement-2. amu is also called as avogram. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is the CORRECT EXPLANATION for Statement-1. |
|
| 39. |
Statement 1: The standard emf (E_(cell)^(@)) of following concentration cell is zero. Zn(s)|ZnSO_(4)(C_(1))||ZnSO_(4)(C_(2))|Zn(s) Statement 2: The electrolyte concentration cell will be is greater in cathodic half-cell. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 =0 for all concentration cells `E=(0.059)/(N)log_(10)((C_(2))/(C_(1)))` (for the given cell) LTBR the cell will be spontaneous (E=+ve) when `C_(2)` is greater than `C_(1)`, i.e., concentrtion in cathodic half-cell is greater than that of ANODIC half-cell. |
|
| 40. |
Statement-1: The solution of Na in dry ether not only contains Na_("soln")^(+) and e_("soln")^(-) but also contains Na_("soln")^(+) formed by the following disproportionation reaction. 2Na(s) |
|
Answer» Statement -1 is true, Statement -2 is true, Statement -2 is CORRECT EXPLANATION for statement -1. |
|
| 41. |
STATEMENT-1: The reference electrode of silver-silver chlorlde is used as secordary reference electrode. and STATEMENT-2: The electrode is reversible with respect to CI^(-) ions. |
|
Answer» STATEMENT-1 is True, STATEMENT-2 is True, STATEMENT -2 is correct EXPLANATION for STATEMENT-1 |
|
| 42. |
STATEMENT -1: Thesolubilityof maleicacidin wateris higherthan thatof fumaricacid . and STATEMENT- 2 :Maleicacidand fumaricacid arecis- trans isomers . |
|
Answer» STATEMENT -1 is TRUE , STATEMENT-2 si True ,STATEMENT-2 is a correctexplanationfor STATEMENT -1 |
|
| 43. |
Statement-1 : The ratio of Zn^2+ and F^(-) per unit cell of ZnS and CaF_2 is 1 :2 Statement-2 : In ZnS,Zn^2+ ions occupy alternate tetrahedral voids and in CaF_2, F^(-) ions occupy all the tetrahedral voids of FCC unit cell. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 `CaF_2` [Flourite structure] [`AB_2` type] (8:4 coordination number compound): `S^(2-)` ion from the FCC lattice. `toZn^(2+)` ion occupy alternate (non adjacent) four tetrahedral void. `Ca^(2+)` ion from the FCC lattice. `to F^(-)` ion occupy all tetrahedral voids. 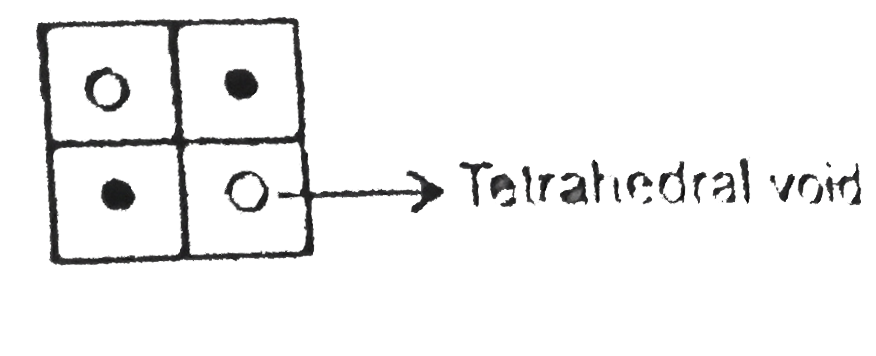
|
|
| 44. |
Statement-1. The rate constant of a pseudounimolecular reaction has the units of the rate constant of a second order reaction. Statement-2. A pseudounimolecular reaction is a reaction of second order in which one of the reactant is present in large excess. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-1 is a correct EXPLANATION of Statement-1. Statement-1 `(k'=k[H_(2)O]" so that "k=k'//[H_(2)O]=" time"^(-1)//"mol L"^(-1)="L mol"^(-1)" time"^(-1))` |
|
| 45. |
Assertion (A): The rate of reaction increases generally by 2 to 3 times for every 10^(@)C rise in temperature. Reason (R ): An increase intemperature increases the colliison frequency. |
|
Answer» |
|
| 46. |
Statement 1: The product formed is (B). Statement 2: The reaction proceeds via the formation of the following species in the order: Radical anion rarr Vinylic anion rarr Vinylic redical rarrProduct |
|
Answer» a.Statement 1 and Statement 2 are true and Statement 2 is the correct explanation of Statement 1. Radical ANION `rarr` Vinylic redical `rarr` Vinylic anion rarr product. Statement `1` is true and statement `2` is false. So the answer is `(c )`. |
|
| 47. |
Statement 1: The product (B) formed will be a racemic mixture. Statement 2: The above reaction is oxymercuration and demercuration, and it proceeds via the addition of D_(2)O, according to Markovnikov's rule, and with antiregiospecificity. |
|
Answer» a.Statement 1 and Statement 2 are TRUE and Statement 2 is the CORRECT explanation of Statement 1. |
|
| 48. |
Statement 1: The plot of atomic number (y-axis) versus number of neutrons (x-axis) for stable nuclei shows a curvature towards x-axis from the line of 45^(@) slope as the atomic number is increased, and Statement 2: Proton-Proton electrostatic repulsions begin to overcome attractive forces involving protons and neutrons in heavier nuclides. |
|
Answer» STATEMENT 1 is TRUE, statement 2 is true, statement 2 is CORRECT EXPLANATION for statement 1. |
|
| 49. |
Statement-1: The oxidation state of Mo in the oxido complex species[Mo_(2)O_(2)(C_(2)H_(4))_(2)(H_(2)O)_(2)]^(2+) is +3. Statement-2: TheC_(2)H_(4)" and "H_(2)O are neutral ligands whereas O in as anionic ligand. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-1. `O^(2-)` DONOR LIGAND |
|
| 50. |
Statement-1 : The overall rate of a reversible reaction may decrease with the increase in temperature. Statement-2 : When the activation energy of forward reaction is less than that of backward reaction, then the increase in the rate of backward reaction is more than that of forward reaction on increasing the temperature. |
| Answer» ANSWER :A | |