Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-I: If decimolal solution of sodium chloride boils at 101.2^(@)C, then decimolal solution of calcium chloride will also boil at the same temperature. Because Statement-II: For same molal concentration of aqueous solutions of electrolytes, the elevation of boiling point may not be same. |
|
Answer» |
|
| 2. |
Statement I : Hard water does not give lather with soap . Statement II : Calcium and magnesium salts present in hard water form precipitate with soap . |
|
Answer» Both the statements individually true and Statement II is the correct explanation of Statement I. |
|
| 3. |
Statement-I: Glucose gives a reddish-brown precipitate with Fehiling's solution because Statement-2: Reaction of glucose with Fehling's solution gives CuO and gluconic acid |
|
Answer» Statement-I is TRUE, Statement-II is True : Statement-II is a CORRECT EXPLANATION for Statement-I |
|
| 4. |
Statement -I : Glass is not considered as a true compound Statement -II : Glass does not have a difinite melting point |
|
Answer» Both the statements are individually TRUE and statement II is the correct EXPLANATION of statement I |
|
| 5. |
Statement-I: Fuan is more soluble than DHF Dihydrofuran, in H_(2)O Statement-II: Greater e^(-) density on the O atom, stronger is the H-bonding and more soluble is the ether. |
|
Answer» If both statement-I & statement-II are TRUE & the statement-II is a CORRECT EXPLANATION of the statement-I |
|
| 6. |
Statement-I: Ethers o reaction with air and light form hydroperoxides. These peroxides decompose vilently at high temperature. Allyln-n-propyl ether with O_(2) in lgiht gives mainly l-hydroperoxide allyl-n-propyl ether Statement-II: The reaction proceeds via the formation of radical anion. |
|
Answer» If both statement-I & statement-II are TRUE & the statement-II is a CORRECT EXPLANATION of the statement-I |
|
| 7. |
Statement - I. For isotonic solutions C_1 = C_2 Statement - II. For isotonic solutions pi_1 = pi_2 |
|
Answer» Statement-1 is TRUE, statement-2 is true and statement-2 is CORRECT EXPLANATION for statement-1. |
|
| 8. |
Statement-I : Elements of oxygen family are not capable of forming p pi-p pi bonds with other elements of smaller size like C, N ets Statement-II : Only oxygen prossess this property of show multiple bonding |
|
Answer» Statement-I and Statement-II are CORRECT and Statement-II is correct EXPLANATION of Statement-I |
|
| 9. |
Statement -I : does not give bromamide reaction. Statement-II : secondary amides does not give bromamide reaction. |
|
Answer» If both Statement-I & Satement-II are TRUE&the Statement-II is not a correct EXPLANATION of the Statement-I. 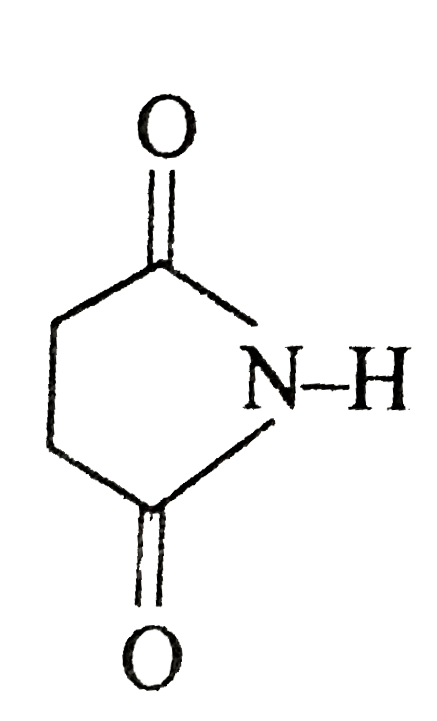 GIVE bromamide REACTION. GIVE bromamide REACTION.
|
|
| 10. |
Statement-I: Diphynyl ether (I) on dinitration gives the |
|
Answer» If both statement-I & statement-II are TRUE & the statement-II is a CORRECT EXPLANATION of the statement-I |
|
| 11. |
Statement I : Colour of nitrogen dioxide changes to colourless at low temperature Statement II : At low temperature Nitrogen tetroxide (N_(2)O_(4)) is formed which is colourless. |
|
Answer» Both the statements individually true and Statement II is the CORRECT explanation of Statement I. `(N_(2)O_(4))` . At higher temperature of `NO_(2)` is favoured while at low temperature `N_(2)O_(4)` predominates. |
|
| 12. |
Statement-I : Chlorination of ethyl benzene with Cl_(2) in prescence of heat and light nearly yield 1-yield 1-chloro-1-phenyl ethane as major product. Because Statement-II : The reaction oC Curs through intermediate formation of the radical C_(6)H_(5)CH-CH_(3). |
|
Answer»
|
|
| 13. |
Statement -I :Cryolite is used in electrolytic extraction of Al from alumuna.Statement -2: It dissilves alumina |
|
Answer» |
|
| 14. |
Statement-I : C_(6)H_(5) is a meta-directing group. Because Statement-II : -C_(2)H_(5) is a + l group. |
|
Answer» Statement-I is TRUE, Statement-II is True : Statement-II is a CORRECT EXPLANATION for Statement-I |
|
| 15. |
Statement-I : Buta-1,3-diene is less stable than Penta-1,4-do,eme, ltbr. Because Statement-II : Buta-1,3-diene has greater number of reasonating structures and delocallsed electron cloud. |
|
Answer» |
|
| 16. |
Statement - I. Benzene-toluene mixture forms ideal solution. Statement - II. Components with structural similarity forms ideal solution in general. |
|
Answer» Statement-1 is TRUE, statement-2 is true and statement-2 is CORRECT EXPLANATION for statement-1. |
|
| 17. |
Statement I : At high temperature , hydrogen can reduce PbO to elemental lead. Statement II : Hydrogen has great affinity to oxygen. |
|
Answer» Both the statements individually true and STATEMENT II is the correct explanation of Statement I. |
|
| 18. |
Statement-I: At any level of temperature (-6^(@)C "to" 300^(@)C) the elevation or depression can be measured by adjusting the Hg level in the bulb of Beckman's thermometer unlike in convention thermometer . Statement-II: In Cottrell's method, pumping device which resembles an inverted funnel is used to reduce superheating during boiling . (III): Rast's method is generally used for Solid solution (solute-solid, solvent-solid) Statement-IV: In Berckeley-Hartley method, copper ferrocynaide is precipitated in the pores Correct statement is /are: |
|
Answer» only I, II |
|
| 19. |
Statement-I : Antiseptic are not injected into the body. Statement-II : Antiseptic are intravenous drugs |
|
Answer» Both I and II are TRUE |
|
| 20. |
Statement-I : Alkyness are more reactive than alkenes towards electrophilic reagents like H^(+). Because Statement-II : The alkyl carbocation formed from alkene is more stable than the uinyl carbocation formed from alkyne. |
|
Answer» |
|
| 21. |
Statement-I : Addition of Br_(2^(-)) water containing dissolved NaCl to ethylene gives a mixture of 1, 2- dibromoethane, 1-bromo-2-chloroethane and 2-bromoethanol. Because Statement-II : Addition oC Curs through a carbocation intermediate. |
|
Answer» |
|
| 22. |
Statement I :After cutting an apple or a banana the colour of the cut surface becomes brown Statement II : Polyphenolic compunds present in fruits get oxidized in air and show colour |
|
Answer» Both the statements are individually TRUE and statement II is the CORRECT EXPLANATION of statement I `Ca(s)+H_(2)SO_(4)(AQ)rarrCaSO_(4)+H_(2)` |
|
| 23. |
Statement - I : Aldehydes restore the megenta colour of schiff's base Statement - II : Schiff's reagent is a colourless P - rosaniline hydrochloride |
|
Answer» I is TRUE II is WRONG |
|
| 24. |
Statement-I: Acyl halide are more reactive than acid substance amide towards nucleophilic substitution. Because Statement-II: X^(-) are better leaving group than NH_(2). |
|
Answer» Statement-I is TRUE, Statement-II is True : Statement-II is a CORRECT EXPLANATION for Statement-I |
|
| 25. |
Statement I: Acetic acid does not undergo haloform reaction. Statement II: Acetic acid has no alpha hydrogen. |
|
Answer» a.STATEMENT I and Statement II are true and Statement II is the correct explanation of Statement I. In acetic acid `(underset(alpha)(C )H_(3)-COOH)`, `alpha`-hydrogen atoms are present. Hence, statement `I` is correct but statement `II` is incorrect. So the answer is `(c ). |
|
| 26. |
Statement - I: A very dilute acidic solution of Cd^(2+) and Ni^(2+) givesyellowprecipitate of CuS on passing H_(2)S Statement - II : The solubility productof CdS is morethan that of NiS |
|
Answer» Statement - I is true ,Statement - II is also true , Statement - II is the correct explatationfor Statement - I Therefore on passing `H_(2)S` in acidic solution a yellow precipirtate of CuS is formed |
|
| 27. |
Statement-I: 1M solution of Glauber's salt is isotonic with 1M solution of KNO_(3). Because Statement-II: Solutions having same molar concentration fo solute may or may not have same osmotic pressure. |
|
Answer» |
|
| 28. |
Statement: Fluresence is the emission of light by sucstances after absorbing light. Explanation: It may continue for appreciable time after the exciting light is switched off. |
|
Answer» (a) `S` is correct but `E` is WRONG |
|
| 29. |
Statement (A) : Zinc will free Cu metal from copper sulphate solution. Reason (R) : At 288 K temperature, E_(Zn^(2+)|Zn)^(@)=-0.76 volt and E_(Cu^(2+)|Cu)^(@)=0.34 Volt |
|
Answer» Both STATEMENT and reasons are true. And REASON is correct explanation of statement. `E_(Cu^(2+)|Cu)^(@)=0.34V,E_(Zn^(2+)|Zn)^(@)=-0.76V` From the above VALUE it can be shown that zinc liberated ELECTRON and reduction of copper occur. |
|
| 30. |
Statement: According transition state theory for the formation of an activated complex, one of the vibrational degree of freedom is converted into a transitional degree of freedom. Explanation: Energy of the the activated complex is higher than the energy of reactant molecules. |
|
Answer» (a) `S` is CORRECT but `E` is wrong |
|
| 31. |
Statement (A): Determination of cathode and anode is done with the help of thermometer. Reason (R) : High and less value of reduction potential is strong reducing agent. |
|
Answer» Both statement and REASONS are true. And REASON is CORRECT explanation of statement. |
|
| 32. |
Statement (A) : Corrosion of galvanized iron is not possible. Reason (R) : Electrode potential of zinc is more negative than of iron. |
|
Answer» Both statement and reasons are true. And REASON is correct explanation of statement. |
|
| 33. |
Statement “Ozone in atmosphere is decreased by emission of chloro-fluoro-carbons like "CCl"_(2)F_(2) ” |
|
Answer» Is true |
|
| 34. |
Statement-2: The 'spin only' magnetic moment of green complex, potassium amminetetracyanidonitrosonium chromate(I) is 1.73 BM. Statement-2: To have two d-orbital empty for d^(2)sp^(3) hybridization, the pairing of electrons take place having behind one unpaired electron as CN^(-) is a stronger field ligand. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT explanation for Statement-1. |
|
| 35. |
Statement-2: Hund's rule violates in [Co(CN)_(6)]^(3-) complex ion. Statement-2: Degeneracy of d orbitals is lost under any field ligand. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 36. |
Statement-2: All pseudohalide ions are not stronger coordinating ligands than the halide ion and hence the complex formed by pseudohalide ions are of high spin. Statement-2: The halide ions have higher values of electronegativity in their corresponding periods. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a CORRECT explanation for Statement-1. |
|
| 37. |
Statement-1:When PhCHO is made to react in OD^(Theta)//D_2O no D is incorporated into the CH_2 ol PhCH_2OH, PhCOO^(-) mixture during Cannizaro's reaction. Statement-2:Intermolecular H^(Theta) transfer takes place in the rate determining step, with out getting free in solution. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 38. |
Statement-1:White crystalline precipitate of silver sulphite dissolves if sulphite ions are added in excess. Statement-2:Sulphite ions decolourise the pink colour of acidified KMnO_4 |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 Statement-2:`5SO_3^(2-)+2MnO_4^(-)`(pink)+`6H^(+)to 2Mn^(2+)`(colourless)+`5SO_4^(2-)+3H_2O` |
|
| 39. |
Statement-1:White precipitate of zinc phosphate is soluble in water. Statement-2:Zinc phosphate forms a soluble complex with ammonia. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 |
|
| 40. |
Statement-1:Wrought iron is prpared by pudding process Statement-2:Pig iron is the purest form of iron |
|
Answer» Statement -1 is true, Statement-2 are true and Statement-2 is the CORRECT EXPLANATION of Statement -1 |
|
| 41. |
Statement-1:When 3-Chlorobut-1-ene react H_2O then give four products. Statement-2:Intermediate is carbocation and it show resonance and recemisation. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 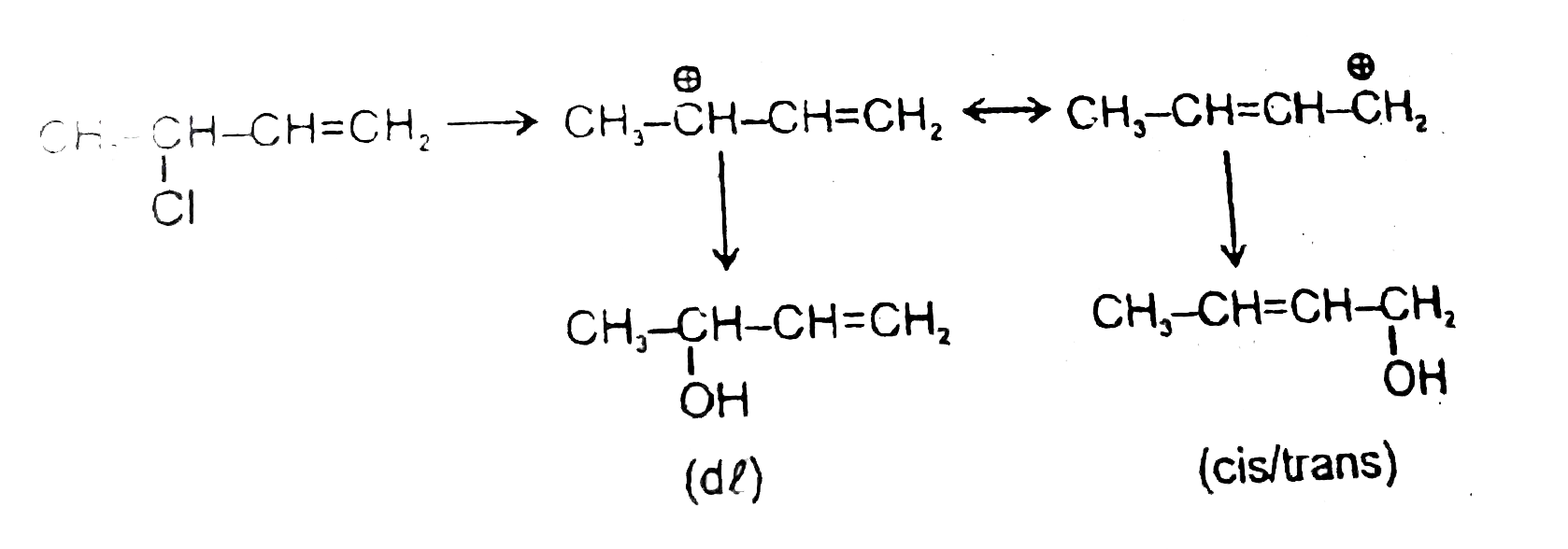
|
|
| 42. |
Statement-1:The order of base catalysed hydrolysis of ester is : (i)CH_3-COOCH_3gtCH_3g-COOC_2H_5gtCH_3-COOCH(CH_3)_2 Statement-2:S_N2 Th is sterically as well as electronically controlled reaction. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 43. |
Statement-1:The preparation of G.R. occurs in solution phase. Statement-2: The reaction will be explosive in solid phase. G.R. is stable only in solution phase. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 
|
|
| 44. |
Statement-1:When 1 mole of benzaldehyde and 1 mole of cyclohexanone is treated with 1 mol of semicarbazide cyclohexanone semicarbazone precipitates first and finally precipitate of semicarbazone of benzaldehyde is formed Statement-2:First one is Kinetically controlled product and later one is thermodynamically controlled product. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 45. |
Statement 1:Teflon has high thermal stability and chemical inertness. Statement 2:It has strong (C-F)bonds. |
|
Answer» Statement `1` is TRUE ,statement `2`is true,statement`2`is the CORRECT EXPLANATION of statement`1` |
|
| 46. |
Statement-1:Sulphur dioxide is a more powerful reducing agent in alkaline medium than in acidic medium. Statement-2:Sulphur dioxides reacts with hydrogen sulphide in presence of moisture and sulphur is precipitated. |
|
Answer» Statement-1 and statement-2 are correct andstatement-2 is the correct explanation for Statement-1 `SO_2 +4OH^(-)hArr SO_4^(2-)+2H_2O+2e^(-)`(increase in `[OH^-]` will shift the equilibrium in forward direction ) `S_2:2H_2S+SO_2 overset("moisture")to2H_2O +3S darr` |
|
| 47. |
Statement-1:Specific conductance decreases with dilution whereas equivalent conductance increases. Statement-2:On dilution number of ions per millilitre decreases but total number of ions increases consideably. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 |
|
| 48. |
Statement-1:Sodium chloride is added during electrolysis of fused anhydrous magnesium chloride. Statement-2:Anhydrous magnesium chloride is obtained by heating hydrated magnesium chloride, MgCl_2. 6H_2O |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation for Statement-1 S-2: `MgCl_2. 6H_2O` is calcined in a stream of dry HCL gas to have anhydrous `MgCl_2` . In absence of dry HCl. It forms `Mg_2OCl_2 (MgO. MgCl_2)` because of HYDROLYSIS . |
|
| 49. |
Statement-1:Silica is added as a flux in reverberatory furnace, in the extraction of copper from copper pyrites. Statement-2:Silica acts as basic flux. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 `2FeS+3O_2 to 2FeO +2SO_2` `FeO+SiO_2 to FeSiO_3`, thus remove the basic impurity of IRON oxide. `S_2`:Silica acts as an ACIDIC FLUX. |
|
| 50. |
Statement-1:Reduction of But-2-yne of Na/liq. NH_5 gives trans-But-2-ene. Statement-2: It is an example of 'anti' addition. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|