Saved Bookmarks
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
State the role of activated complex in a reaction and state its relation with activation energy. |
| Answer» Solution :Hint. ACTIVATION ENERGY = Energy of ACTIVATED complex-Average energy of REACTANTS | |
| 2. |
State the role of Depressant in froth floatation process. |
| Answer» SOLUTION :A depressant is USED when two sulphides are present in thr ORE. For example in the mixture of ZNS and PbS, NaCN is used as depressant. It selectively prevents ZnS from COMING to the froth but allows PbS to come to the froth. | |
| 3. |
State the relation between E_("cell")^(@) and equilibrium constant. |
|
Answer» `E_("CELL")^(@) = (RT)/(NF) LN K` |
|
| 4. |
State the reactions and reactions conditions for the following conversions : (i) Benzene diazonium chloride to nitrobenzene (ii) Aniline to benzene diazonium chloride (iii) Ethylamide to methylamine. |
Answer» Solution :(i)  (ii)  (III) `underset("Ethylamide")(CH_(3)-overset(overset(O)(||))(C)-NH_(2))+Br_(2)+4KOH RARR underset("METHYLAMINE")(CH_(3)NH_(2))+K_(2)CO_(3)+2KBr+2H_(2)O` |
|
| 5. |
State the reactions for obtaining benzoic acid from aniline. |
Answer» SOLUTION :Steps INVOLVED in the conversion of aniline to BENZOIC acid are GIVEN below : 
|
|
| 6. |
State the reactions and reaction conditions for the following conversions : (i) Benzene diazonium chloride to nitrobenzene. (ii) Aniline to benzene diazonium chloride. (iii) Ethyl amide to methylamine. |
Answer» Solution :(i)  (III) `UNDERSET("Ethylamide")(CH_(3)-overset(overset(O)(||))(C)-NH_(2))+Br_(2)+4KOH rarr underset("Methylamine")(CH_(3)NH_(2))+K_(2)CO_(3)+2KBr+2H_(2)O` |
|
| 7. |
State the reaction of conc. Nitric acid with sulphur. Give equation. |
| Answer» SOLUTION :When SULPHUR is TREATED with CONC. `HNO_3`, it OXIDISES sulphur to `H_2SO_4`. | |
| 8. |
State the reaction taking place when : Bromine water is added to the aqueous solution of aniline. |
Answer» SOLUTION :
|
|
| 9. |
State the reaction for obtaining benzoic acid from aniline. |
Answer» SOLUTION :
|
|
| 10. |
State the Raoult's law in its general form in reference to solutions. |
|
Answer» Solution :Raoult.s law in its general form in reference to solutions In a solution of VOLATILE LIQUIDS, the partial vapour pressure of each component in the solution is directly PROPORTIONAL to its MOLAR fraction. Thus, for component 1 `p_1 = p_1^0 xx x_1` where `p_1` = partial vapour pressure of component 1 in the solution `p_1^0` = vapour pressure of pure component 1 `x_1` = mole fraction of component 1 Similarly for component 2 ` p_2 = p_2^0 xx x_2` |
|
| 11. |
State the purpose of impregnating the paper with colloidion solution . |
| Answer» SOLUTION :To reduce pore SIZE of FILTER paper , so that COLLOIDAL particles cannot PASS through . | |
| 12. |
State the products of electrolysis obtained on the cathode and the anode in the following cases: (i) A dilute solutoin of H_(2)SO_(4) with platinum electrodes (ii) An aqueous solution of AgNO_(3) with silver electrodes. |
|
Answer» Solution :(i) `H_(2)SO_(4)(aq)to2H^(+)(aq)+SO_(4)^(2-)(aq),""H_(2)OhArrH^(+)+OH^(-)` At cathode: `H^(+)+etoH,H+HtoH_(2)(g)` At ANODE: `OH^(-)toOH+E^(-),4OHto2H_(2)O(L)+O_(2)(g)` THUS, `H_(2)` is liberated at the cathode and `O_(2)` at anode. (ii) `AgNO_(4)(s)+aqtoAg^(+)(aq)+NO_(3)^(-)(aq),""H_(2)OhArrH^(+)+OH^(-)`. At cathode. `Ag^(+)` ions ahve lower discharge potential than `H^(+)` ions. hence, `Ag^(+)` ions will be deposited on the cathode `(Ag^(+)+e^(-)toAg)` At anode. Ag anode will be ATTACHED by `NO_(3)^(-)` ions. hence, Ag anode will dissolve to form `Ag^(+)` ions in the solution `(AgtoAg^(+)+e^(-))` |
|
| 13. |
State the products of the following reactions: (i) CH_(3)CH_(2)CH_(2)OCH_(3)+HBrto (iii) (CH_(3))_(3)C-OC_(2)H_(5)overset(HI)(to) |
Answer» SOLUTION :(i) `CH_(3)CH(_2)CH_(2)OCH_(3)+HBrtoCH_(3)CH_(2)CH_(2)OH+CH_(3)BR` 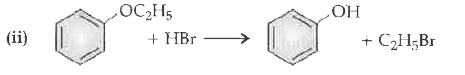 (iii) `(CH_(3))_(3)C-O-C_(2)H_(5)+HI to ` `CH_(3)-underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C)-I+C_(2)H_(5)OH` |
|
| 14. |
State the product formed during reaction betweeen sodium phenoxide and ethyl iodide on heating |
| Answer» Solution :NA | |
| 15. |
State the product available by the following reaction CH - underset(CH_(3))underset(|)overset(CH_(3))overset(|)(C) - NH_(2)underset([O])overset(KMnO_(4))to |
|
Answer»
|
|
| 16. |
State the principle of refining of metal by the following methods : (a) Zone refining (b) Electrolytic refining (c) Vapour phase refining |
|
Answer» Solution :(a) The impurities are more soluble in the melt than in the SOLID STATE ofthe METAL. (b) Impure metal is made to act as ANODE, while the strip of same metal in pureform as CATHODE. |
|
| 18. |
State the oxoacids of phosphorus with their formula, methods of preparation and presence of characteristics bonds in their structure . |
Answer» Solution : EXISTS in POLYMERIC FORMS only. Characteristic bonds of `(HPO_3)_3` have been given in the table. The compositions of axoacids are INTERRELATED in TERMS of loss or gain of `H_2 O` or O-atom. |
|
| 20. |
State the monomer present in natural rubber. |
|
Answer» ISOPRENE |
|
| 21. |
State the main difference between bacteriostatic and bactericidal antibiotics. Give one example of each. |
|
Answer» Solution :BACTERIOSTATIC ANTIBIOTICS have inhibitory effect while bactericidal antibiotics have killing effect on MICROBES. EXAMPLE : Bacteriostatic antibiotic : Tetracycline Bactericidal antibiotic : OFLOXACIN |
|
| 22. |
State the main advantage of molality over molarity as the unit of concentration. |
| Answer» Solution :MOLALITY is independent of TEMPERATURE, whereas molarity is a FUNCTION of temperature. | |
| 23. |
State the limitations of open chain structure of glucose. |
|
Answer» Solution :The open chain structure of glucose could not explain the following observations: (i) Despite having the aldehydic group, glucose does not give Schiff.s test and it does not FORM the hydrogensulphite addition product with `NaHSO_(3)`. (ii) The pentaacetate of glucose does not react with hydroxylamine indicating the ABSENCE of free -CHO group. (iii) Glucose is found to exist in two different CRYSTALLINE forms which are named as `alpha`- and `beta` - glucose. The glucose shows the property of Mutarotation i.e., the change in the specific ROTATION. However, the aldehyde does not show the property of the Mutarotation. (iv) On reaction with 1 mole of methanol, it yields two monomethyl derivatives which are known as `alpha`-D-glucoside and `beta`-D-glucoside. |
|
| 24. |
State the important physical properties of phenols. |
|
Answer» SOLUTION :Physical properties of phenols. The important physical properties of phenols are as follows: 1. The pure, phenols are generally colourless liquids or solids. But they readily turn brownish due to the atmospheric oxidation. 2. Phenols undergo the formation of intermolecular hydrogen bonding.  Therefore, their boiling points are much higher than those of corresponding aromatic hydrocarbons and aryl halides. For example, the boiling points of TOLUENE, CHLOROBENZENE and phenol are as follows : Toluene (384 K) , Chlorobenzene (403 K) , Phenol (455 K). 3. As in the case of alcohols, phenols can also formhydrogen bonds with water and therefore they should be soluble in water. But with the exception of phenol itself the solubility of phenols in water is very small. This is possible due to the fact that in phenols, the hydrocarbon part of the molecule is RELATIVELY quite large. |
|
| 25. |
State the hybridisation of carbon atom in carbonyl group. |
| Answer» SOLUTION :The hybridisation of carbon ATOM in carbonyl GROUP is `sp^(2)` - hybridised. | |
| 26. |
State the hybridisation and magnetic behaviour of [Cr(CO)_(6)]. |
|
Answer» Solution :Hybridisation of Cr in `[Cr(CO)_(6)]` is `d^(2)SP^(3)` which gives rise to octahedral SHAPE. As all the unpaired ELECTRONS are used in bond formation, no unpaired electron is LEFT. The compound is THEREFORE diamagnetic. |
|
| 27. |
State the Henry's law about partial pressure of a gas in a mixture. |
|
Answer» <P> Solution : Henry.s law about PARTIAL pressure of a gas in a mixtureThe partial pressure of the gas (p) in vapour phase is proportional to the mole FRACTION of the gas (x) in the solution. It MAY be EXPRESSED as `p = K_H.x` where p = partial pressure of the gas `K_H` =Henry.s constant x = mole fraction of the gas |
|
| 28. |
State the genral electronic configuration of elements of actinoid series. Why there are irregularities in the electronic configurations? |
Answer» Solution :The general electronic configuration of elements of actinoid series is `[Rn] 5f^(0-14) 6d^(0-2) 7s^(2)`. In tripositive state, the configuration is `5f^(N)` (n=1 to 14). Except actinium and thorium in tetrapositive state, the configuration is `5f^(n)` (n=1 to 13)  Reason for irregularities: The irregularities in the electronic configuration of actinoid series is due to very less difference in the energies of 5f and 6d orbitals. All actinoids believe to have the electronic configuration of `7s^(2)` and variable occupancy of 5f and 6d subshells. From protactinium (PA) the 14 electrons are added successively in 5f-orbitals till lawrencium. The irregularities in the configuration of actinoids are related to the STABILITIES of `f^(0), f^(7) and f^(14)` occupancies of 5f-orbitals. Thus the configurations of Am and CM are `[Rn]4f^(7) 7s^(2) and [Rn] 5f^(7) 6d^(1) 7s^(2)` respectively. Due to presence of one d-electron in actinium and lawrencium, these elements are some time considered as group 3 elements |
|
| 29. |
State the general electronic configuration of lanthanoids. Why there are irregularities in the electronic configuration of lanthanoids? |
Answer» Solution :The general electronic configuration of lanthanoid elements is `[Xe] 4f^(1-14) 5d^(0-1) 5s^(2)` however, in a tripositive state the general configuration is `[Xe] 4f^(n) (n =1 " to " 14)`  REASON for the irregularities: The irregularities in electronic configurations are because of very less ENERGY difference between 4f adn 5d orbitals. Hence, the last ELECTRON may enter EITHER in 4f and 5d orbitals In lanthanoids, f-orbitals must be filled successively with the increase in atomic number. However, in lanthanum (La), cerium (Ce), gadolinium (Gd) and lutetium, one electron is present in 5d ORBITAL. These irregularities are because of relative stabilities of `f^(0), f^(7) and f^(14)` configuration in lanthanum, gadolinium and lutetium respectively. However, cerium is accepted presently as exception. As lanthanum and lutetium has one 5d electrons, these elements are sometime referred as group 3 elements |
|
| 30. |
State the general electronic configuration of p-block elements. Which factors largely governs the properties of p-block elements ? |
|
Answer» Solution :The general electronic configuration of p-block element is `ns_2 np^(1-6)`. Except HELIUM (He) which has `1s^(2)` configuration. The properties of p-block elements are influenced by : (i) Atomic SIZE (ii) Ionisation enthalpy (iii) ELECTRON gain enthalpy (iv) Electronegativity (v) Absence of d-orbitals in second period elements and PRESENCE of d or d and f-orbitals in HEAVIER elements. The presence of all the three types of elements, metals, metalloids and non-metals bring diversification in chemistry of these elements. |
|
| 31. |
State the function along with one example each of : (i) Antihistamines(ii) Antioxidants |
|
Answer» Solution :(i) Autihistamines are drugs USED to TREAT allergy such as skin RASHES, conjuctivities , rhinitis, etc. (ii) ANTIOXIDANTS are the food additives which retard the action of oxygen on the food and thereby help in its preservation. E.g, butylated hydroxytoluene (BHT). |
|
| 32. |
State the formula relating pressure of a gas with its mole fraction in a liquid solution incontact with it. |
|
Answer» <P> SOLUTION :`P_(gas) = K_H XX ` mole fraction of the gaswhere `K_H`= Henry.s law CONSTANT. |
|
| 33. |
State the formula relating pressure of a gas with its mole fraction in a liquid solution in contact with it. |
|
Answer» <P> SOLUTION :ACCORDING to Henry's law,Partial pressure of a gas above the solution `= K_(H)xx" MOLE fraction of the gas in the solution"` or `p_(A)=K_(H)xx x_(A),` where `K_(H)`= Henry's constant. |
|
| 34. |
State the fromula relating the pressure of the gas with its mole fraction in a liquid solution in contact with ot. |
| Answer» SOLUTION :`P=K_(H)xx"MOLE FRACTION of GAS in solution".` | |
| 35. |
State the following: Raoult's law in its general term with reference to solution? Henry's law about partial pressure of a gas in mixture. |
|
Answer» SOLUTION :For answer, sonsult SECTION 5 (VAPOUR PRESSURE of liquid SOLUTIONS) For answer, sonsult section 3 (Solubility) |
|
| 36. |
State the first law of thermodynamicsin two ways . |
|
Answer» Solution :(1) Energy can neitherbe creatednor DESTROYED,however,it maybe convertedfrom ONE forminto another. (2) Whenever, a QUANTITY of one KIND of energyis consumed or disappears, an EQUIVALENT amount of another kind of energy appears. |
|
| 37. |
State the first and second law of electrolysis. |
|
Answer» Solution :First law of electrolysis : The mass of any substance deposited or liberated at any electrode is directly proportional to the quantity of electricity passed. Thus if `w_(g)` of the substance is deposited on PASSING Q coulombs of electricity, then `WpropQ` or `W=ZQ` where Z is a constant of proportionality called electrochemical equivalent of the substance deposited. If a current of C amperes is passed for t SECONDS then `Q=Cxxt` so at `W=ZxxCxxt` or, `W=("Equivalent wt. of the substance")/(96500)xxCxxt` Second law of electrolysis : When the same quantity of electricity is passed through solutions of weight of the SUBSTANCES produced at the electrodes is directly proportional to their equivalent weights. For example, for `CuSO_(4)` solution and `AgNO_(3)` connected in the series if the same quantity of electricity is passed then, `("Weight of Cu deposited")/("Weight of Ag deposited")=("eq. wt. of Cu")/("eq. wt. of Ag")` |
|
| 38. |
State the features of chemical adsorption which are not nound in physical adsorption. |
| Answer» | |
| 39. |
State the Faraday's first law of electrolysis. How many Faraday of electricity is required for the reduction of 1 mole of Mg^(2+) ions? |
| Answer» SOLUTION :2 Faraday'sof ELECTRICITY is REQUIRED. | |
| 40. |
State the equations for the preparation of the following compounds . (Equations need not to be balance). (i) Chlorobenzene from aniline (in two steps ) (ii) N-Propyl amine from ethyl chloride (in two steps ) |
|
Answer» Solution :` (##KSV_CHM_ORG_P2_C15_E01_164_S01.png" WIDTH="80%"> II. `UNDERSET ("Ethyl chloride" ) (CH_3CH_2Cl) + Alc.KCN rarr CH_3 CH_2 CN` ` underset ((Reduction)) overset (H_2//Ni)rarr underset ("n-Propylamine") (CH_3CH_2CH_2NH_2)`. |
|
| 41. |
State the element X by the following electronic configuration. X= [Ar] 3d^(10) 4s^(1) |
|
Answer» Ni |
|
| 42. |
State the electronic configurations of Group-17 elements. |
Answer» SOLUTION :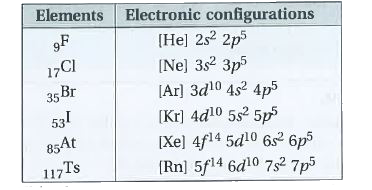 This elements have SEVEN electrons in valance shell. Hence, the general electronic CONFIGURATION is `ns_2np_5`. Halogens are highly reactive as they have tendency to gain one electron to acquire a NOBLE gas configuration. |
|
| 43. |
State the electronic configurations of group-16 elements. |
Answer» Solution :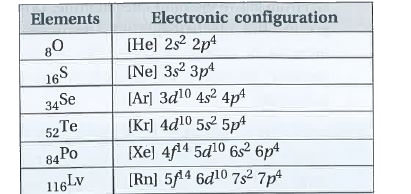 The elements of group-16 have SIX electrons in VALANCE SHELL. Hence, the GENERAL configuration is `ns^2np_4`. |
|
| 44. |
State the electronic configurations of group-18 elements. |
Answer» Solution :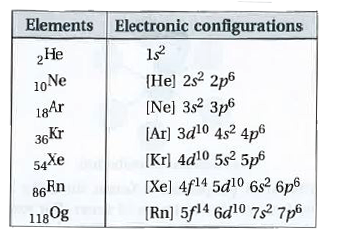 All these elements have eight electrons in valance shell, HENCE the general electronic configuration in `ns^2 np^6` except `He(1S^(2))` . DUE to presence of completely filled shells, these elements are chemically unreactive and forms very few COMPOUNDS. Hence they are called NOBLE gases. |
|
| 45. |
state the electronic configuration of group-15 elements. |
Answer» Solution :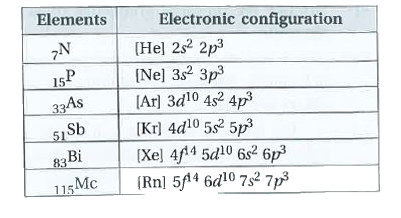 All the ELEMENTS of group-15 have 5 electrons in their valance SHELL. Hence, the GENERAL electronic configuration is `ns^(2)np^(3)` |
|
| 46. |
State the difference between Schottky and Frenkel defects. Which of these two changes the density of the solid ? |
| Answer» SOLUTION :SCHOTTKY DEFECT. | |
| 47. |
State the conditions under which the followingpreaprations are carried out. Giver the necessaryeqactions which need not be balanced. i. Ethabnol form accetllene. ii. Lead tetracthyl from sodium lead alloy. iii. Methyl chloride from aluminium carbide. |
Answer» Solution :i. Etanol from aceetylene: 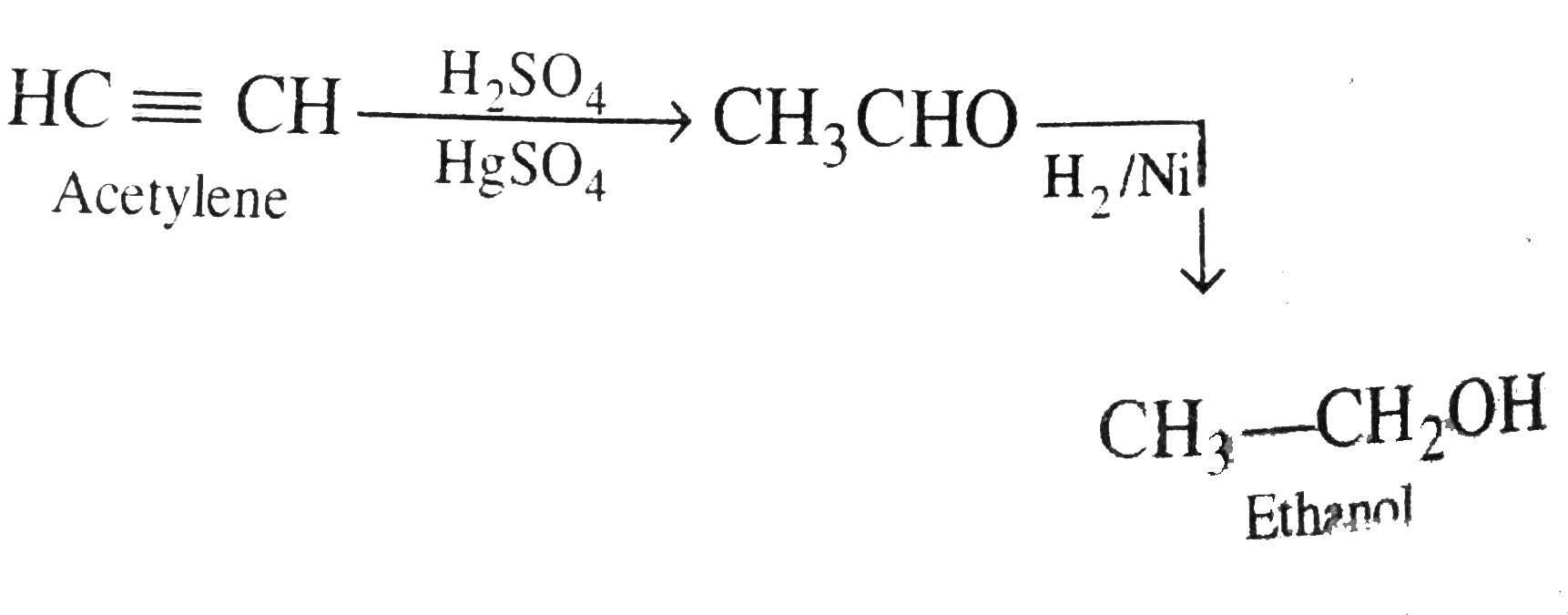 ii. Lead tetrathyl from sodium - lead alloy: `4C_(2)H_(5)Br + underset("alloy")underset("Sodium-lead")(4(Na//Pb)) rarr underset("lead")underset("Tetrachtyl")(Pb (C_(2) H_(5))_(4)) + 4 NaBr + 3Pb` iii. Methyl CHLORIDE form aluminium carbide: `underset("carbide")underset("Aluminium")(Al_(4)C_(3)) overset(H_(2)O)(rarr) CH_(4) underset(hv)overset(Cl_(2))(rarr) underset("chloride")underset("Methyl")(CH_(3)Cl)` |
|
| 48. |
State the constituents of natalite. |
| Answer» SOLUTION :The CONSTITUENTS of NATALITE are DIETHYL ether and ETHANOL. | |
| 49. |
State the consequences of lanthanoid contraction |
|
Answer» Solution :The atomic RADII of elements of second and third transition series are similar because of lanthanoid CONTRACTION. Ex `Zr- Hf, Nb- Ta` ETC. have nearly same size. Because of similar atomic radii, these elements show similar chemical properties and hence are very difficult to separate. The ionization enthalpies of elements of third transition series are higher than second transition series elements because of lanthanoid contraction through these elements belong to same groups The basic strength of hydroxides of lanthanoids decrease from lanthanum to leutetrium as a result of lanthanoid contraction the ioni character of hydroxide decreases with the decreases in size of lanthanoids. Hence, `Ce(OH)_(3)` is most basic while `Lu(OH)_(3)` is LEAST basic. These hydroxides are separated on the basis of basic strength. Elements of third transition series show abnormally high DENSITIES because of lanthanoid contraction. |
|



