Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement-1. 1-Butene o reaction with HBr in the presence of a peroxide produces 1-bromobutane. Statement-2. It involves formation of primary radical. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct explanation of statement-1. |
|
| 2. |
Statement-1 : |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1 |
|
| 3. |
STATEMENT - 1 : {:(""OH""OH""CH_(3)""O),(""|""|""|""||),(CH_(3)CH_(2)-C" "-C-CH_(2)CH_(3)underset(/_\)overset(H^o+)rarrCH_(3)CH_(2)" "-C" "-C-CH_(3)),(""|""|""|""|),(""CH_(3)""CH_(3)""CH_(3)CH_(2)""CH_(3)),(""("Major products")):} STATEMENT - 2 : CH_(3)CH_(2)group shifts during this reaction as it is a better electron donor so better migrator than -CH_(3)group. |
|
Answer» Statement - 1 is true, Statement - 2 is TURE, Statement-2 is a CORRECT explanation for Statement-1. |
|
| 4. |
Statement-1 : 0.1 M HCl solution has higher ozmotic pressure than "0.1 M NaCl" solution. Statement-2 : Cl^(-) ions being common, the small size H^(+) ions have greater ionic mobility than large size Na^(+) ions. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT explanation of Statement-1. |
|
| 5. |
Statemen I : In Pb_(3) O_(4) all Pb has + ( 8)/( 3) oxidation number. Statement II : Pb_(3) O_(4) is mixed oxide of PbO & PbO_(2) |
|
Answer» If both Statement-I and Statement-II are TRUE, and Statement-II is the CORRECT EXPLANATION of Statement–I. |
|
| 6. |
State which of these exhibit stereoisomerism and of what type? (a) CHCH_(2)-overset(Br)overset(|)(C)=overset(Br)overset(|)(C)-CH_(2)Cl (b) Cl-CH_(2)-underset(O)underset(||)(C)-CH_(2)-underset(CH_(3))underset(|)(CH)-CH_(3) (3) CH_(3)-overset(Cl)overset(|)(CH)-underset(Br)underset(|)(CH)-CH_(2)-overset(CH_(2)-CH_(2)-CH_(3))overset(|)(CH)-CH_(3) (d) CH_(3)-CH_(2)-CH(OH)COOH (e) CH_(3)CH=CHC_(2)H_(5) (f) CH_(3)CH(Br)COOH (g) H_(2)C=C(CH_(3))_(2) (h) C_(2)H_(5)CH=CHCl (i) 1-Bromo-3-chlorocyclobutane (j) 1,4-Dimethylcyclohexane |
| Answer» Solution :(a) GEOMETRICAL ISOMERSIM (c) OPTICAL isomerism (d) optical isomerism (e) geometrical isomersim (F) optical isomerism (h) geometrical isomerism (i) geometrical isomerism (J) geometrical isomerism. | |
| 7. |
State with balanced equation what happens when ? (a) Potassium ferrocyanide is heated with conc. H_2 SO_4. (b) A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. (c) Sodium chlorate reacts with sulphur dioxide. (d) Chlorine gas is passed into water saturated with hydorgen sulphide. (e) Hydrogen sulphide is passed through sodium bisulphite solution. |
|
Answer» Solution :(a) `CO` is evolved. `K_4 Fe(CN)_6 6H_2 SO_4 + 6H_2 O overset (DELTA)RARR` `2K_2SO_4 + FeSO_4 + 3(NH_4)_2 SO_4 + 6 CO uarr`. (b) Potassium chlorate is reduced while OXALIC ACID is oxidised. `{:(KClO_(3)+H_(2)SO_(4) rarr KHSO_(4)+HCl +3[O]),(3H_(2)C_(2)O_(4)+3[O] rarr 6CO_(2)+3H_(2)O),(ulbar(KClO_(3)+3H_(2)C_(2)O_(4)+H_(2)SO_(4) rarr KHSO_(4)+HCl+6CO_(2)+3H_(2)O)):}` (c) `NaClO_3` is reduced to `NaCl` `{:(SO_(2)+2H_(2)O rarr H_(2)SO_(4)+2 [H]xx 3),(NaClO_(3)+6 [H] rarr NaCl+3H_(2)O),(ulbar(NaClO_(3) +3SO_(2)+3H_(2)O rarr 3H_(2)SO_(4)+NaCl)):}` (d) `Cl_2 + H_2 Srarr 2HCl + S`. (e) `{:(2NaHSO_(3) rarr NaSO_(3) +H_(2)O +SO_(2)),(SO_(2)+2H_(2)S rarr 2H_(2)O+ 3S),(ulbar(2 NaHSO_(3) +2H_(2)S rarr NaSO_(3) +3S +3H_(2)O)):}`. |
|
| 8. |
Account for the following: (i) pK_(b) of aniline is more than that of methylamine. (ii) Ethylamine is soluble in water whereas aniline is not. (iii) Methylamine in water reacts with ferric chloride to precipitate hydrated feric oxide. (iv) Although amino group is o– and p– directing in aromatic electrophilicsubstitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline. (v) Aniline does not undergo Friedel-Crafts reaction. Diazonium salts of aromatic amines are more stable than those of aliphaticamines. (vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines. |
|
Answer» (i), (II) , (III) |
|
| 9. |
State what is observed when : the electrodes connected to a battery are dipped into a sol. |
| Answer» SOLUTION :The COLLOIDAL PARTICLES get ATTRACTED TOWARDS oppositely charged electrodes. This phenomenon is called electrophoresis. | |
| 10. |
State whether ethylene diammine is a monodentate or bidentate ligand. |
| Answer» SOLUTION :BIDENTATE LIGAND . | |
| 11. |
State what is observed when : an emulsion is subjected to high speed centrifugation. |
| Answer» SOLUTION :Emulsion GETS demulsified. We get SEPARATE LAYERS of the emulsion. | |
| 12. |
State what is observed when : an electrolyte solution is added to a sol. |
| Answer» Solution : The sol GETS coagulated. The CHARGE on the colloidal particles is NEUTRALISES by the ELECTROLYTE. | |
| 13. |
State uses of phosphine. |
|
Answer» Solution :PHOSPHINE is USED in making Holme.s signals. CONTAINERS containing calcium CARBIDE [`CaC_2`] and calcium phosphide [`Ca_3P_2`] are pierced and thrown in the sea when the gases evolved BURN and serve as a signal. It is used in preparation of smoke screen. |
|
| 14. |
State uses of dioxygen. |
|
Answer» Solution :In addition to its importance in normal respiration and COMBUSTION PROCESSES, oxygen is USED in oxyacetylene welding, in the manufacturing of steel. Oxygen cylinders are used in hospitals, high ALTITUDE flying and in mountaineering. The combustion of fuels, e.g., hydrazines in LIQUID oxygen provides tremendous thrust in rockets. |
|
| 15. |
State Van't Hoff-Boyle's law. |
| Answer» SOLUTION :At constant TEMERATURE, the osmotic PRESSURE of a solution isdirectly PROPORTIONAL to (molar) concentration of the solution. | |
| 16. |
State unpaired electrons in ""_(28)Ni |
|
Answer» 2 |
|
| 17. |
Write two uses of acetone. |
|
Answer» Solution :(i) Used ass an important SOLVENT. (ii) Used for the manufacture of Iodoform. Chloroform, ACETIC ANHYDRIDE, Synthetic RUBBER, ETC. |
|
| 18. |
State two uses of acetic acid. |
|
Answer» Solution :(i) It is USED in the preparation of rayon, plastics ETC. (ii) It is used in the MANUFACTURE of paints. |
|
| 19. |
Give two uses of formaldehyde. |
|
Answer» SOLUTION :(i) USED to PREPARE UROTROPINE, formalin. (ii) Used for the manufacture of bakelite. |
|
| 20. |
State two methods of preparation of formaldehyde. |
|
Answer» SOLUTION :(i) Used to prepare UROTROPINE, formalin. (II) Used for the MANUFACTURE of BAKELITE. |
|
| 21. |
State two main differences between globular and fibrous proteins. |
Answer» SOLUTION :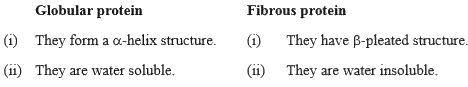
|
|
| 22. |
State two main differences betweenglobular proteins andfibrousproteins. |
Answer» SOLUTION :Differencces between globularproteins and FIBROUS PROTINE. 
|
|
| 23. |
State two important uses of noble gases. |
|
Answer» SOLUTION :(i) AR is used in electrical bulb. (II) Xe is used in head LIGHT of cars these days. |
|
| 24. |
State two advantages of H_2 - O_2 fuel cell over ordinary cells. |
|
Answer» Solution :(i) It is HIGHLY EFFCIENT. (II) It is POLLUTION FREE. |
|
| 25. |
State two advantages of H_(2)-O_(2) fuel cell over ordinary cell. |
|
Answer» Solution :(i) They do not CAUSE any POLLUTION. (ii) They have high EFFICIENCY of 60-70%. |
|
| 26. |
State the various uses of : Tri - iodomethane (Iodoform) |
|
Answer» SOLUTION :Tri - iodomethane : It was used EARLIER as an ANTISEPTIC but the antiseptic properties are due to the liberation of iodine and not due to iodoform itself. Due to its objectionable smell, it has been replaced by other formulations CONTAINING iodine. |
|
| 27. |
State the various uses of : Trichloromethone (Chloroform) |
|
Answer» Solution :Trichloromethane `(CHCl_(3))` : Chloroform is employed as a solvent for fats, oils alkaloids, iodine and other substances. The major use of chloroform is in the PRODUCTION of REFRIGERANT Freon R - 22. It was once USED as a eneral an anaesthesia in surgery but now has been replaced by LESS toxic safer anaesthetic such as ETHER. |
|
| 28. |
State the various uses of : Tetrachloromethane (Carbon tetrachloride) |
|
Answer» Solution :Tetrachloromethane : It is USED as feedstock in the synthesis of chlorofluorocarbons and other chemicals, pharmaceutical MANUFACTURING and general solvent use. It is produced in a large QUANTITIES for use in the manufacture of refrigerants and PROPELLANTS for aerosol cans. Until the MID 1960s, it was widely used as a cleaning fluid, both in industry, as a degreasing agent, and in the home, as a spot remover and as a fire extinguisher. |
|
| 29. |
State the value of C-O-C bond angle in ether. |
|
Answer» `108^(@)` |
|
| 30. |
State the various uses of : Freons |
|
Answer» Solution :FREONS : These are used as aerosol propellants, refrigeration and AIRCONDITIONING purposes. The chlorofluorocarbon compounds of methane and ethane are COLLECTIVELY known as freons. They are extremely stable, unreactive. non - toxic, non - corrosive and easily liquefiable gases. Freon 12 `(C Cl_(2)F)` is one of the most common freons in inductrial use. It is MANUFACTURED from tetrachloromethane by Swarts reaction. These are usually produced for aerosol propellants, refrigeration and air CONDITIONING purposes. By 1974, total freon production in the world was about 2 billion pounds annually. |
|
| 31. |
State the various uses of : Dichloromethane (Methylene chloride) |
| Answer» SOLUTION :Dichloromethane `(CH_(2)Cl_(2))` : It is used as a propellants in aerosols, and as a process solvent in the manufacture of DRUGS. It is also used as a metal cleaning and FINISHING solvent. | |
| 32. |
State the uses of noble gases. |
|
Answer» Solution :Helium : Helium is a non-inflammable and light gas and hence, it is used in filling balloons for meteorological observations. It is used in gas-cooled nuclear reactors. Liquid helium (b.p. 4.2 K) finds use as cryogenic AGENT for carrying out various experiments at low temperatures. It is used to produce and sustain powerful superconducting magnets which form essential part of modern NMR spectrometers and Magnetic Resonance Imaging (MRI) systems for clinical diagnosis.It is used as a diluent for oxygen in modern diving apparatus because of its very low solubility in blood. Neon : It is used in DISCHARGE tubes and fluorescent bulbs for advertisement DISPLAY purposes. Neon bulbs are used in botanical gardens and in green houses. Argon : Argon is used mainly to provide an inert ATMOSPHERE in high temperature metallurgical processes (arc welding of metals or alloys) and for filling electric bulbs. It is also used in the laboratory for handling substances that are air-sensitive. Xenon and Krypton : They are used in light bulbs DESIGNED for special purposes. |
|
| 33. |
State the uses of sulphuric acid. |
|
Answer» SOLUTION :Sulphuric acid is a very important industrial chemical. A nation.s industrial strength can be judged by the quantity of sulphuric acid it produces and consumes. It is used in manufacturing fertilizers such as ammonium sulphate and ammonium superphosphate. In petroleum refining. In manufacture of pigments, paints and dye stuff intermediates. In detergent industry and metullargical applications e.g. CLEANSING metals before ENAMELING, electroplating and galvanising. In lead storage batteries and as a LABORATORY reagents. In the manufacture of NITROCELLULOSE products. It is known "King of chemicals". |
|
| 34. |
State the uses of nitric acid. |
|
Answer» Solution :The major use of nitric acid is in the manufacture of ammonium nitrate for fertilisers and other NITRATES for use in EXPLOSIVES and pyrotechnics. Nitro compounds such as NITROGLYCERIN, trinitrotoluene etc. are prepared from `HNO_(3)` . It is also used in the picking of stainless steel, ETCHING of metals and as oxidizer in ROCKET fuels. |
|
| 35. |
State the uses of halogen containing organic compounds. |
|
Answer» Solution :These compounds are used as a solvents for RELATIVELY non - polar compounds and as a starting materials for the synthesis of WIDE range of organic compounds. Chloramphenicol, a Chlorine containing antibiotic, produced by microorganisms is very effective for the tretment of typhoid fever. Our body produces iodine containing hormone, thyroxine, thedeficiency of which causes a disease called goiter. Synthetic halogen compounds, viz. chloroquine is used for the treatment of MALARIA, halothane is used as an ANAESTHETIC during surgery. Certain FULLY fluorinated compounds are being considered as potential blood substitutes in surgery. |
|
| 36. |
State the use of Potassiumdichromat [K_(2)Cr_(2)O_(7)] |
|
Answer» As an OXIDIZING AGENT to convert FERRIC ion from FERROUS ion in acidic MEDIUM |
|
| 37. |
State the uses of antiseptics. |
|
Answer» Solution :(1) Antiseptics are used for dressing of WOUNDS. (2) They are used to DESTROY fungal growth. (3) They are used to sterilize surgical instrument. (4) They are used as anti-infectives in deodorants, SHAMPOOS and mark for each POINT) surgical soaps. |
|
| 38. |
State the type of catalysis and the principle of catalysis related to them in the following reactions : (i) 2SO_(2)(g)+O_(2)(g)overset(NO(g))to2SO_(3)(g) (ii) 2SO_(2)(g)+O_(2)(g)overset(Pt(s))to2SO_(3)(g) |
| Answer» | |
| 39. |
State the third law of thermodynamics . |
|
Answer» Solution :Third law of THERMODYNAMICS STATES that the ENTROPY of a pure, perfectlyordered crystalline substancesis zeroat absolutezero temperature(0 K). HENCE at 0K , for pure crystallinesubstance, `S = 0`. |
|
| 40. |
State the significance of numbers 6 and 6,6 in the polymer names nylon -6 and nylon 6,6. or in nylon 6,6 what does the designation 6,6 mean ? |
| Answer» Solution :NYLON 6 means that it is a condensation POLYMER of only one type of MONOMER molecules containing six CARBON atoms , i.e., caprolactam. Nylon 6,6, on the other hand , implies that it is a condensation polyer of two types of monomer molecules each containing six carbon atoms, i.e., adiple acid `(HOOCH_2CH_2CH_2CH_2CH_2COOH)` and hexamethylendiamine `(H_(2)NCH_(2)CH_(2)CH_(2)CH_(2)CH_(2)CH_(2)NH_(2))`. | |
| 41. |
State the sequence of steps to obtain: a. Acetophenone from benzene b. Acetone from acetyl chloride. |
Answer» SOLUTION :
|
|
| 42. |
State the sigh of entropy change involved when the molecules of a substance get adsorbed on a solid surface . |
| Answer» SOLUTION :`DELTA =-ve` | |
| 43. |
State the significance of number in polymer names Nylon-6 and Nylon-66 .Write the monomers used for making Nylon-66. |
|
Answer» Solution :(a) Nylon-6 . The number 6 INDICATES that the polymer is made of MONOMER having siz carbon atoms. The monomer is caprolactum `(C_6 H_(11) NO)` . (b) Nylon-66 . The number 66 indicates that the polymer is made up of two monomers, each having SIX carbon atoms. The monomers are HEXAMETHYLENE diamine `(H_2 N (CN_2)_6 NH_2) ` and adipic acid `(HOOC (CH_2)_4 COOH)` . |
|
| 44. |
Second law of thermodynamics |
|
Answer» Solution :The second law or thermodynamics STATES the total entropy of the system and its surroundings (universe) INCREASES in a spontaneous PROCESS. OR Since all the natural PROCESSES are spontaneous, the entropy of the universe increases. It is expressed mathematically as `underset("(for spontaneous process)")(DeltaS_("Total")=DeltaS_("system")+DeltaS_("surr") GT 0 )` `DeltaS_("Univers")=DeltaS_("system")+DeltaS_("surr")gt0` |
|
| 45. |
State second law of thermodynamics. |
|
Answer» Solution :SINCE all the natural PROCESSES are spontaneous,they takeplacewith increases in the entropy. Hence secondlaw of thermodynamicscan be representedmathematically as `DeltaS_("Total") = DeltaS_("system") + DeltaS_("SURR") GT 0 ` (for spontaneousprocess) `DeltaS_("Universe") = DeltaS_("System") + DeltaS_("surr") gt 0` |
|
| 46. |
State the rules for writing the IUPAC names of co-ordination compounds. |
|
Answer» Solution :(i) In ioniccomplexes cation is NAMED first and then the anion. (ii) In co-ordination entity ligands are named first then the central metal ion atom. (iii) (a) Anionic ligands ends in 'O'  (b) Cation ligands end in 'ium' `NO^(+) "Nitrosonium" "" NO_(2)^(+) "Nitronium"` ( c ) Neutral ligands are named as such with few exceptions.  (iv) When more than one type of ligands are present, they are named in alphabetical order. (V) Prefixes di, tri, etc.... are used to indicate the number of the individual ligands in the co-ordination entity. (vi) When the names of the ligands include a numerical prefix, then the terms BIS, tris, tetrakis are used. (vii) When the COMPLEX is CATIONIC or neutral, the names of the metal is written without any characteristic ending. For anionic complex, the name of the central metal atom ends in 'ate'.  (viii) Oxidation state of the metal atom/ion is indicated by Roman numeral in parenthesis. |
|
| 47. |
State the role of silica in the metallurgy of copper and crydit in the metallurgy of aluminium. Differentiate between roasting and calcination. |
|
Answer» Solution :Silica act as flux in the metallurgy of `CuSiO_(2)+FeOrarrFeSiO_(3)` and Gyolite is going to decrease the MELTING point of melt or MIXTURE. CALCINATION is heating are in limited supply of oxygen/air, while roasting is heating are in a regular supply of air in a FURNACE at a temperature below the melting point of the metal. |
|
| 48. |
State the role of Silica in the metallurgy of copper. |
|
Answer» Solution :Copper ore is heated in a reverberatory furnace after MIXING with silica. IRON oxide impurity is converted to a SLAG of iron silicate which is removed. Copper is produced in the FORM of copper matte. `{:(FeO+ SiO_(2) to FeSiO_(3)),("Slag "):}`. |
|
| 49. |
State the role of silica in the metallurgy of copper. |
|
Answer» Solution :During roasting, copper PYRITES are CONVERTED into a mixture of FeO and `Cu_(2) O`. `{:(2CuFeS_(2) + O_(2) overset(Delta)rarr Cu_(2)S + 2FES + SO_(2)),(2Cu_(2)S + 3O_(2)overset(Delta)rarr 2Cu_(2)O + 2SO),(2FeS + 3O_(2) rarr 2FeO + 2SO_(2)):}` To remove FeO (basic), an acidic flux silica is added during smelting. FeO then combines with `SiO_(2)` to form ferrous silicate `(FeSiO_(3))` slag which floats over molten matte (and hence can be EASILY removed.) Thus, the role of silica in the metallurgy. of copper is to remove ferrous oxide as slag. |
|
| 50. |
State the role of Graphite rod in the electrolytic reduction of alumina. |
|
Answer» Solution :The electrolysis of the molten `Al_(2)O_(3)` is carried out in an electrolytic cell using graphite rods. The oxygen liberated at the ANODE REACTS with the carbon of the anode producing CO and `CO_2`. For each kilogram of aluminium produced, about `0.5kg` of the anode is burnt away. The electrode reactions are : `Al^(3+)+3e^(-) to Al "CATHODE "` `{:(C+O^(2-) to CO +2e^(-)),(C+2O^(2-) to CO_(2)+4e^(-)):}}" Anode "`. |
|