Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement -1: alpha-Keratin is a structural protein Statement -2: It is globular protein. |
|
Answer» Statement 1 is TRUE, statement-2 is true , statmenent -2 is a correct explanation for statement -1 |
|
| 2. |
Statement 1 : Al(OH)_(3) is amphoteric in nature statement 2: Al-O and O-H bonds can be broken with equal ease in Al(OH)_(3) |
|
Answer» STATEMENT 1 is true, statement 2 is true, statement 2 si a correct explanation for statement 6 |
|
| 3. |
Statement 1: All the halogens are coloured. Statement 2: Halogen molecules absorb some wavelengths of visible light and the electrons are promoted to higher energy molecular orbitals. |
|
Answer» STATEMENT-1 is true, statement-2 is true and statement -2 is correct EXPLANATION for statement-1 |
|
| 4. |
Statement-1: All tetrahedral complexes are mainly high spin and the low spin configurations are rarely observed. Statement-2: Delta_(t) is always much smaller even with stronger field ligands and it is never energetically favourable to pair up the electrons. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 5. |
Statement - 1: All minerals areores. Statement - 2: Oresare mineralsfrom whichmetal canbeextractedconvenientlyand economically. |
|
Answer» Statement - 1 is TRUE, Statement -2 isTrue, Statement-2iscorrectexplanationofStatement -1. |
|
| 6. |
Statement 1 : All metals can not be obtained by carbon reduction. Statement 2 : Carbon is a strong reducing agent. |
|
Answer» Statement 1 is TRUE, statement 2 is true : statement 2 is a correct EXPLANATION for statement 1 |
|
| 7. |
Statement-1: All known monouclear complexes of chromium with +3 oxidation state, irrespective of the strength of the ligand must have three unpaired electrons. Statement-2: In complex [Cr(NH_(3))_(4)BrCl]Cl,the 'spin only' magnetic is close to 3.87 BM. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 8. |
Statement-1: Aldehyde and ketone give nucleophilic addition reactions with carbon oxygen, nitrogen and sulphur nucleophiles. Statement-2: Sulphite ion is ambident nucleophile. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-4 |
|
| 9. |
Statement-1: Alcohols gives only substitution products with HX and not elimination product. Statement-2: overline(X) is a very weak which can not abstract proton from alcohol. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-10 |
|
| 10. |
Statement-1: Alcohols can not be dried by CaCl_(2). Statement-2: CaCl_(2) forms solvated molecules with alcohols. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-8 |
|
| 11. |
STATEMENT-1: Alcino steel is used for making permanent magnets. and STATEMENT-2: Alcino steel is highly magnetic. (A) (B)(C) S (D) |
|
Answer» STATEMENT - 1 is True, STATEMENT-2 is True, STATEMENT -2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 12. |
Statement - 1 : Al is used as a reducing agent in aluminothermy. And Statement - 2 : Al has a lower melting point than Fe, Cr and Mn. |
|
Answer» STATEMENT - 1 is true, Statement - 2 is True, Statement - 2 is correct explanation for statement - 1. |
|
| 13. |
Statement 1 : Al is used as a reducing agnet in aluminothermy. Statement 2 : Al has a lower melting point than Fe. Cr and Mn. |
|
Answer» Statement 1 is true, statement 2 is true : statement 2 is a correct explanation for statement 1 |
|
| 14. |
STATEMENT-1: AICI_(3) forms dimer Al_(2)CI_(5) but it dissolves in water forming [Al(H_(2)O_(6))^(3+) and CI ions. and STATEMENT-2: Aqueous solution of AICI, is acidic due to hydrolysis. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is a CORRECT EXPLANATION for STATEMENT-1. |
|
| 15. |
Statement - 1 : Ag and Au are extracted by leaching their ores with a dilute solution of NaCN. And Statement - 2 : Impurities associated with these ores dissolve in NaCN. |
|
Answer» STATEMENT - 1 is TRUE, Statement - 2 is True, Statement - 2 is CORRECT EXPLANATION for statement - 1. |
|
| 16. |
Statement-1 : Adiabatic process must be isentropic process. Statement-II : dS=(dq_("rev"))/T |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 17. |
Statement 1: Addition of bromine to trans-2-butene yields meso-2,3-dibromobutane. Statement 2: Bromine addition to an alkene is an electrophilic addition. |
|
Answer» Statement 1 is TRUE, statement 2 is true, statement 2 is a correct explanation for statement 1 |
|
| 18. |
Statement-1: Addition of bromine on trans -2 butene yeilds meso-2,3-dibromo butane. Statement-2: The addition of Br_(2) on double bond is anti-addition. |
|
Answer» If both the statement are TRUE and Statement -2 is the correct EXPLANATION of Statement-1: |
|
| 19. |
Statement-1: active mass of pure solid and pure liquid is taken unity. and Statement -2 : The active mass of pure solids and liquids depends on density and molecular mass . The density and molecular mass of pure liquidsand solids are constant. |
|
Answer» Statement -1 is TRUE, Statement -2 is true , Statement-2 is correct EXPLANATION for Statement -4 |
|
| 20. |
Statement-1. Addition of Br_(2) to 1-butene gives two optical isomers. Statement-2: The product contains one asymmetric carbon. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION of statement-1. |
|
| 21. |
Statement 1: Acrylic acid is a weaker acid than benzoic acid. Statement 2: Ethylenic double bond is less electron donating than benzene ring. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a CORRECT EXPLANATION for statement 1 |
|
| 22. |
Statement 1: Acryaldehyde (CH_(2)=CH-CHO) is oxidised to acrylic acid (CH_(2)==CH-COOH) by Benedict's solution. Statement 2: Benedict's solution is ammoniacal CuSO_(4) solution containing sodium potassium tartarate. |
|
Answer» a.Statement 1 and Statement 2 are true and Statement 2 is the correct explanation of Statement 1. |
|
| 23. |
Statement 1: Acrylic acid (CH_(2)=CHCOOH) is a weaker acid than benzoic acid (C_(6)H_(5)COOH). Statement 2: Ethylenic double bond is less electron-donating than benzene ring. |
|
Answer» STATEMENT-1 is True, statement-2 is true,statement-2 is a correct explanation for statement-1 |
|
| 24. |
Statement-1: AcidcatalyzeddehydrationfollowsE_(1)mechanismStatement - 2 :Tertiaryalcohols are morereactivethanprimaryalcoholstowards HBr. Statement-3 :Dehydrohalogenation of alkylhalidefollows E_(2) mechanism |
| Answer» Answer :A | |
| 25. |
Statement-1: Acid catalysed dehydration of t-butanol is faster than n-butanol. Statement-2: The order of stability of carbocation is 3^(@)gt2^(@)gt1^(@). |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for statement-9 |
|
| 26. |
Statement-1: Acetylaceotne complexes of the metal ions are more stable than the corresponding ethylenediamine complexes. Statement-2: Acetylacetone complexes of the metal ions contain a six membered conjugate chelate rings and the resonance in its chelate ion is an additional factor contributing to the stability. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 27. |
Statement 1: Aceto acetic ester (CH_(3)COCH_(2)COOC_(2)H_(5)) contains CH_(3)CO gp. But does not give Iodoform test. Statement 2: The H-atoms of the CH_(3) gp. Are more acidic than those of CH_(2)gp. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a correct EXPLANATION for statement 1 |
|
| 28. |
Statement 1: Acetoacetic ester (CH_(3)COCH_(2)COOC_(2)H_(5)) contains CH_(3)CO group but does not give iodoform test. Statement-2: The H-atoms of the CH_(3) group are more acidic than those of CH_(2) group. |
|
Answer» STATEMENT-1 is True, statement-2 is true,statement-2 is a correct explanation for statement-1 |
|
| 29. |
STATEMENT-1 : According to the lock - and - key model. When the key (substrate) fits the lock (active site) the chemical change bedins. STATEMENT-2 : Modern X-ray crystallographic and spectroscopic methods show that in many cases, the enzyme changes shape when the substrate lands at the active site. STATEMENT- 3 : Enzyme catalysed reactions processed through a fast, reversible formation of an enzyme substrate complex, followed by a slow conversion to product and free enzyme. |
|
Answer» T F T |
|
| 30. |
STATEMENT - 1 : Aceticaciddoes not undergo haloform reaction . and STATEMENT - 2 : Acetic acidhas noalpha hydrogen . |
|
Answer» STATEMENT -1 is True ,STATEMENT -2IS True, STATEMENT-2is a correct explanationfor STATEMENT -1 . |
|
| 31. |
Assertion (A): A is very dilute acidic solution of Cd^(2+) and Ni^(2+) gives yellow precipitate of CdS on passing hydrogen sulphide. Reason (R) : Solubility product of CdS is more than that of NiS. |
|
Answer» STATEMENT 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 |
|
| 32. |
Statement-1.A sol Sa_(2)S_(3) prepared by the action of H_(2)S on As_(2)O_(3) is negativity charged. Statement-2. It is due to the presence of S^(2-) ions in the diffused layer. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct explanation of Statement-1. |
|
| 33. |
Statement-1. A single C^(12) atom has a mass exactly 12 amu and a mole of these atoms has a mass of exactly 12 grams. Statement-2. A mole of atoms of any element has a mass in grams equal to its atomic mass. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is the CORRECT EXPLANATION for Statement-1. |
|
| 34. |
Statement 1 : A mixture of the solutions of a weak acid and its conjugate base is acts as a good buffer. Statement 2 : The ratio of the conjugates base of acid in the mixture does not change substantially when small amount of acids or alkalies are added to the buffer. |
|
Answer» STATEMENT 1 is true, statement 2 is true , statement 2 is correct explanation for statement 2 |
|
| 35. |
STATEMENT - 1 : A mixture of ZnO and CuO can be separated by boiling the mixture with NaOH solution. and STATEMENT - 2 : ZnO dissolves in NaOH solution while CuO remains undissolved. |
|
Answer» Statement-1 is TRUE, Statement-2, is True, Statement-2 is a CORRECT EXPLANATION for Statement-4 |
|
| 36. |
Statement-1. A low level of noradrenaline in the body causes depression. Statement-2. Antidepressant drugs catalyse the degradation of noradrenaline. |
|
Answer» If both ASSERTION and reason are TRUE, and reason is the true explanation of the assertion. |
|
| 37. |
Statement-1: A fractional order reaction must be a complex reaction. Statement-2 : Fractional order of RDS equals to |
| Answer» ANSWER :C | |
| 38. |
Statement-1 : A decrease in pressure leads to an increase in freezing point of water. Statement-2 : For ice on melting volume decreases. |
|
Answer» Statement-1 is TRUE, statement-2 is true and statement-2 is CORRECT EXPLANATION for statement-2 |
|
| 39. |
STATEMENT - 1 : A dark blue colour is obtained on adding excess of dilute NH_(4)OH solution in aqueous soluiton of copper sulphate. and STATEMENT - 2 : Dark blue colour is due to the formation of [Cu(NH_(3))_(4)]^(2+) complex ion. |
|
Answer» Statement-1 is TRUE, Statement-2, is True, Statement-2 is a CORRECT EXPLANATION for Statement-2 |
|
| 40. |
Statement-1 : A catalyst provides an alternative path to the reaction in which conversion ofreactants into products takes place quickly Statement-2 : The catalyst forms an activated complex of lower potential energy, with the reactants by which more number of molecules are able to cross the barrier per unit of time. |
| Answer» ANSWER :A | |
| 41. |
STATEMENT-1: +4 oxidation state of Pb is less stable than +2 state. and STATEMENT -2: Pb_(4) is not a stable compound. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is a CORRECT EXPLANATION for STATEMENT- |
|
| 42. |
Statement-1. 50% of a reaction is completed in 50 sec, therefore, 75% of the reaction will be completed in 75 sec. Statement-2. The rate constant of a zero order reaction depends upon time as k=(1)/(t){[A]_(0)-[A]}. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-1 is a correct explanation of Statement-1. `t=(1)/(k){[A]_(0)-[A]}` For `50%,t_(50%)=(1)/(k){[A]_(0)-([A]_(0))/(2)}=([A]_(0))/(2k)=50" sec"` For `75%,t_(75%)=(1)/(k){[A]_(0)-([A]_(0))/(4)}` `=([A]_(0))/(k)xx(3)/(4)=([A]_(0))/(2k)xx(3)/(2)=75" sec"` |
|
| 43. |
STATEMENT-1 : A catalyst enhances the rate of a reaction and STATEMENT-2 : The energy of activation of the reaction is lowered in presence of a catalyst. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 44. |
Statement-1 : 5f orbitals resemble the 4f orbitals in their angular part of the wave-function. Statement-2 : 5f orbitals are not as buried as 4f orbitals and hence 5f electrons can participate in bonding to a far greater extent. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 45. |
Statement-1: 2A+3B to C 4/3 moles of 'C' are always produced when 3 moles of 'A' & 4 moles of 'B' are added. Statement-2: 'B'is the limiting reactant for the given data. |
|
Answer» Statement-1 is true, statement-2 is true and statement-2 is CORRECT EXPLANATION for statement-1. |
|
| 46. |
Statement-1: 3M solution of alkali metal in liquid ammonia has copper bronze colour. Statement-2: copper bronze colour is due to the formation of metal cluster. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANATION for statement-1 |
|
| 47. |
Statement-1: 2-Pentanol and 3-pentanol cannot be distinguished by iodoform test. Stetement-2: 2-Pentanol and 3-pentanol both are secondary alcohols. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a correct explanation for statement-1. |
|
| 48. |
STATEMENT - 1 : 1,1,1-trideutero-2-priopanol reacts with conc. H_(2)SO_(4)at high temperature to give only one alkene, 3,3,3-trideutero propene. STATEMENT - 2 :C-D bond is stronger than C-H bond. |
|
Answer» Statement - 1 is true, Statement - 2 is Ture, Statement-2 is a CORRECT explanation for Statement-1. 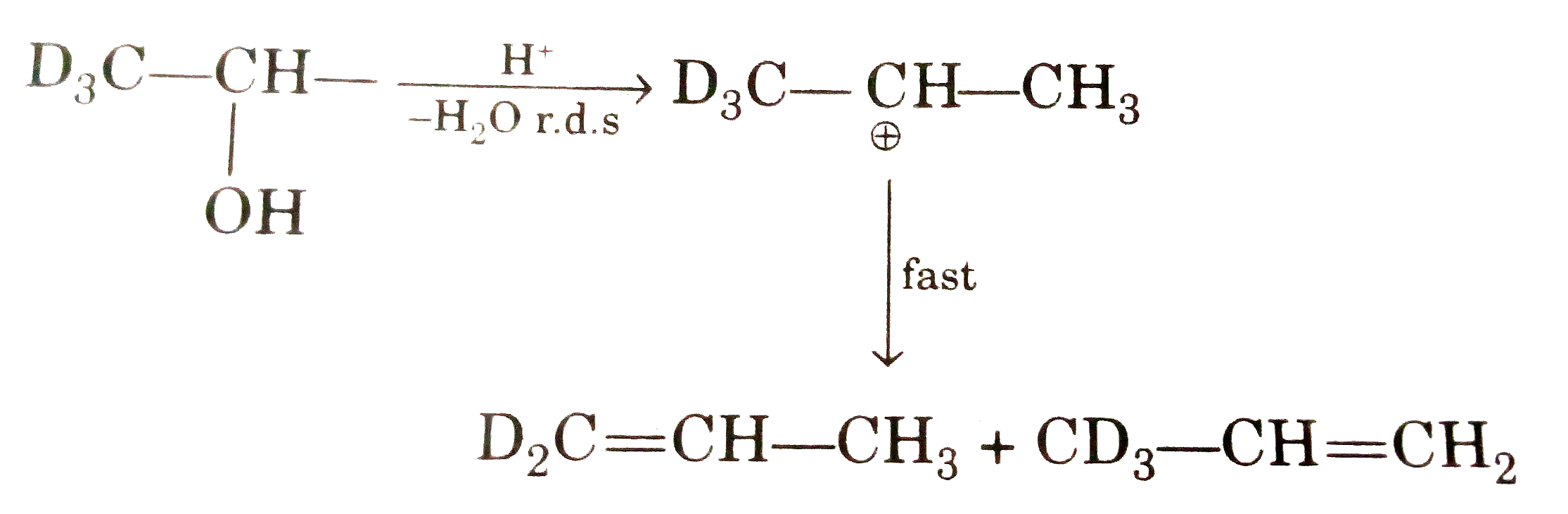 Breakng of C-H or C-D bond is not RDS so both ALKENE are formed. |
|
| 49. |
STATEMENT-1: 1-Butene on reaction with HBr in the presence of peroxide produce 1-bromobutane and STATEMENT-2: It involves the ingormation of primary radical. |
|
Answer» STATEMENT-1 I true, STATENENT-2 is true, STATEMENT-2 is a correct explanation for STATEMENT-1. |
|
| 50. |
Statement-1 : 0.5 M aq. NaOH solution is identical to 2% (w/v) aq. NaOH solution Statement-2 : Concentration in % (w/v) is 4 times the molar concentration for all aqueous solution |
|
Answer» Statement-1 is TRUE, statement -2 is true and statement-2 is correct EXPLANATION for statement-5 |
|