Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
State the common characteristic of actinoids and lanthanoids which places them in the f-block of elements. |
| Answer» SOLUTION :The LAST ELECTRON ENTERS the f-orbital. | |
| 2. |
State the common characteristic of lanthanoids and actanoids which places them in the f-block of elements. |
| Answer» SOLUTION :The LAST ELECTRON in them ENTERS the f-orbitals. | |
| 3. |
State the chemical formula of pyrolusite |
|
Answer» `Mn_(2)O_(3)` |
|
| 4. |
State the characteristics of order of reactions. (OR) Give the characteristics of orderr of a reactions. |
|
Answer» Solution :(i) The magnitude of order of a reaction MAY be zero or fractional or integral values. Order is never fractional for ELEMENTARY reaction. (ii)It should be determined only by EXPERIMENTS. (iii) Simple reactions possess low values of order like n = 0, 1, 2 reactions with order greater than or equal to 3.0 are called complex reactions. (iv) Some reactions show fractional order depending on rate. (v) Higher order reactions may be EXPERIMENTALLY coverted into simpler order (Pseudo) reactions by using EXCESS concentrations of one or more reactants. |
|
| 5. |
State the applications of the d and f-block elements or compounds: (i) In Steel or Iron (ii) In Pigmentation (iii) In Batteries (iv) In Currency Coins (v) As a Catalyst |
|
Answer» Solution :(i) In Steel or Iron: Iron and steel are the most important construction materials Their production is based on the reduction of iron oxides, removal of impurities and the addition of carbon and alloying metals such as Cr, Mn and Ni (ii) In Pigmentation : TiO is used in the pigment industry (iii) In Batteries: The Manganese dioxide `(MnO_(2))` and ZINC are used in the preparation of dry battery cells whereas nickel and cadmium are used in the preparation of batteries In Currency Coins: The elements of Group -11 are called ..Coinage Elements.. because they are precious The Ag and Au are restricted to collection items and the contemporary UK .copper. coins are copper-coated stell The .silver. UK coins are a Cu/Ni alloy (v) As a Catalyst: The transition elements and their compounds are essential catalysts in the chemical industry `V_(2)O_(5)` catalysis the oxidation of `SO_(2) " to " SO_(3)` in the manufacture of sulphuric acid `TiCl_(4)` and `Al(CH_(3))_(3)` forms the basis of Zieglar-Natta catalysts used to manufacture POLYTHENE In Haber.s PROCESS for the production of ammonia from `N_(2)//H_(2)` mixtures, iron catalyst is used Nickel catalysts enable the hydrogenation of fats to proceed In Wacker.s process, the oxidation of ethyne to ethanal is catalyzed by `PdCl_(2)`. NIKEL complexes are useful in the polymerization of alkynes and other organic compounds such as benzene The PHOTOGRAPHIC industry relies on the special light-sensitive properties of AgBr. |
|
| 6. |
State the Arrhenius equation for the rate constant of a reaction. |
|
Answer» Solution :a) A catalyst LOWERS the VALUE of activation energy by LOWERING the height of the energy BARRIER for the reaction. b) Since a catalyst has no effect on the energy of the reactants or products. It has, therefore no effect on the MAGNITUDE of `DeltaG` of a reaction. |
|
| 7. |
State the applications of d and f-block elements and their compounds |
|
Answer» Solution :(i) In Steel or Iron: Iron and steel are the most IMPORTANT construction materials Their production is based on the reduction of iron oxides, removal of impurities and the addition of carbon and alloying metals such as Cr, Mn and Ni (ii) In Pigmentation : TiO is used in the PIGMENT industry (iii) In Batteries: The Manganese dioxide `(MnO_(2))` and Zinc are used in the preparation of dry battery cells whereas nickel and cadmium are used in the preparation of batteries In Currency Coins: The elements of Group -11 are called ..Coinage Elements.. because they are precious The Ag and Au are restricted to collection items and the contemporary UK .copper. coins are copper-coated stell The .silver. UK coins are a Cu/Ni alloy (v) As a CATALYST: The transition elements and their compounds are essential catalysts in the chemical industry `V_(2)O_(5)` catalysis the oxidation of `SO_(2) " to " SO_(3)` in the manufacture of sulphuric acid `TiCl_(4)` and `Al(CH_(3))_(3)` forms the basis of Zieglar-Natta catalysts used to manufacture polythene In Haber.s process for the production of ammonia from `N_(2)//H_(2)` mixtures, iron catalyst is used Nickel catalysts enable the hydrogenation of fats to proceed In Wacker.s process, the oxidation of ethyne to ethanal is catalyzed by `PdCl_(2)`. Nikel complexes are useful in the polymerization of alkynes and other organic compounds such as benzene The photographic industry relies on the special light-sensitive PROPERTIES of AgBr. |
|
| 8. |
State the adverse effects of polyhalogenated compounds. How the adverse effects of chlorofluorocarbons can be prevented ? |
|
Answer» Solution :(i) Methylene dichloride : Exposure to lower levels of methylene chloride in air cause dizziness, nausea, tingling and numbness in the fingers and toes. In humans, direct skin contact with methylene chloride causes intense burning and mild redness of the skin. Direct contact with the eyes can burn the cornea. (ii) Trichloromethane (Chloroform) : Inhaling chloroform depresses the central nervous system. Breathing about 900 parts of chloroform for short time can cause dizziness, fatigue and headache. Chronic chloroform exposure may cause DAMAGE to the liver where chloroform is metabolised to phosgene and to the kidneys, and some people develop sores when the skin is immersed in chloroform. `2CHCl_(3)+O_(2)overset(" Light ")underset(" Oxidation ")rarr 2COCl_(2)+2HCl` Chloroform is slowly oxidised by air in the presence an extermely poisonous gas and therefore it is stored in closed dark coloured bottles completely filled so that air is kept out. (iii) Tetrachloromethane `(C Cl_(4))` : Exposure to carbon tetrachloride causes liver cancer in humans. The most common effects are dizziness, light headedness, nausea and vomiting, which can cause permanent damage to nerve cells. In severe cases, the carbon tetrachloride leads rapidly to stupor, coma, unconsciousness or death. Exposure to `C Cl_(4)` can make the HEART beat irregularly or stop. It causes irritation to eyes upon contact. When `C Cl_(4)` is released into the air, it rises to the atmosphere and DEPLETES the ozone layer. Depletion is ozone layer is believed to increase human exposure to ultraviolet rays, LEADING to increased sking cancer, eye diseases anbd disorders, and possible disruption of the IMMUNE system. (iv) Freons : In stratosphere, the freon is able to initiate the radical chain reactions that can upset the natural ozone balance. Prevention of adverse effects of chlorofluorocarbons (CFCs) :CFCs are highly stable and non - toxic gases. These asre also non - reactive. With the ozone, they initiate the chemical chain reaction and disturbs the natural balance of the ozone layer. If the ozone layer is thinned out, the UV rays reach to the earth which is harmful for flora and fauna. It is recommended that CFCs of refrigerator is replaced by some other compounds. |
|
| 9. |
Give any two uses of formic acid. |
|
Answer» SOLUTION :(i) It is used for REMOVING LINES from the hides in the textile industry. (ii) It is used in DYEING textile and medicine. (iii) It is used as a COAGULATING agent for rubber latex. |
|
| 10. |
State required quantity of electricity to produce 5.12 kg of Al. (atomic weight of Al=27 gm mol^(-1)) |
|
Answer» `1.83xx10^(6)` COULOMB by 3F electricity 1 mol Al=27 GM Al is produced. If 27 gram Al is produced then `3xx96500` coulomb electricity is REQUIRED. So, `5.12xx10^(3)` gm Al is obtained by how much coulomb electricity. * So, coulomb `=(5.12xx10^(3)xx3xx96500)/(27)` `=5.489xx10^(7)` Coulomb |
|
| 11. |
State Rioult's Ltw. How is It formul.ited for solutions of non-volatile solutes ? Derive expression for Rioult's law when the solute is non-volatile. |
|
Answer» Solution :Rioult., law tor solution of non-voliilile solution: The relative lowering of vapour pressure for a solution Is equal to the mole FRACTION of solute when solvent ALONE is VOLATILE. `(P_(A)^(0) - P_(A))/(P_(A)^(0)) = X_(B)` where , `P_(A)^(0) RARR ` vapour pressure of pure component . A. . `P_(A) rarr` Vapour pressure of component .A. `X_(B) rarr ` mole fraction of solute `P_(A)^(0) - P_(A) rarr ` Lowering of vapour pressure `(P_(A)^(0) - P_(A))/(P_(A))rarr` Relative lowering of vapour pressure. |
|
| 12. |
State Saytzeff's rule. |
| Answer» Solution :During intramolecular DEHYDRATION, If there is a possibility to form a carbon - carbon DOUBLE a possibility to form a carbon - carbon double bond at DIFFERENT locations, the PREFERRED location is the one that gives the more (highly) substituted alkene, i.e., the stable alkene. | |
| 13. |
State reasons for the following:The N - O bond in NO_2^(-) is shorter than the N - O bond in NO_3^(-). |
| Answer» SOLUTION :Oxidation state of N in `NO_2^(-)` is +3 while the oxidation state of N in `NO_3^(-)` is +5 . Due to this difference, N-O BOND in `NO_2^(-)` is shorter than the N-O bond in `NO_3^(-)`. | |
| 14. |
State reasons for the following : (i) pK_(b) value for aniline is more than that for methylamine. (ii) Ethylamine is soluble in water whereas aniline is not soluble in water. (iii) Primary amines have higher boiling points than tertiary amines. |
Answer» SOLUTION :(i)  Electron density on N in ANILINE decreases due to RESONANCE with benzene ring whereas electron density on N in methylamine increases due to inductive effect of `-CH_(3)` group. Thus, methylamine is more basic than aniline. In other words `K_(b)` for methylamine is higher. Since, `pK_(b)` is equal to `-log K_(b)`, it is more for aniline than for methylamine. (ii) Ethylamine forms a hydrogen bond with water. But hydrogen bond formation tendency decreases as the alkyl/aryl part increases in size because it is hydrophobic in nature. Hence, due to big phenyl group, aniline is not soluble in water. (iii) Primary amines are able to form intermolecular hydrogen BONDS among their molecules, leading to association of molecules. This raises the b.p. of primary amines. There being no hydrogen attached to N in tertiary amines, there is no POSSIBILITY of hydrogen bonding. Hence, tertiary amines have lower boiling points or primary amines have higher boiling points. |
|
| 15. |
State reasons for the following situations: (i) Monochloroethanoic acid is a weaker acid than dichloro ethanoic acid. (ii) Benzoic acid is a stronger acid than ethanoic acid. |
Answer» Solution :(i)  Chlorine atom exerts -I effect. This helps in the release of protons. There is a ONE chlorine atom inmonochloroethanoic acid and two chlorine atoms in dichloroethanoic acid. Due to smaller -I-effect in monochloroethanoic acid than dichloroethanoci acid, the former is weaker acid than the latter.  Benzene ring withdraws electrons towards itself due to resonance. One the other hand, `-CH_(3)` group is electron replling group. Therefore, the release of protons in benzoic acid will tbe INCREASED while it will be decreased in the case of ethanoic acid. Thus, benzoic acid is STRONGER acid than ethanoic acid. |
|
| 16. |
State reasons for the following:SF_6 is kinetically an inert substance. |
| Answer» SOLUTION :`SF_6` has an octahedral structure. It is a symmetrical and stable structure. Hence, it does not decompose or REACT EASILY and, therefore, acts as KINETICALLY inert. | |
| 17. |
State reasons for the following: (i) Cu (1) ion is not stable in an aqueous solution. (ii) Unlike Cr^(3+), Mn^(2+), Fe^(3+) and the subsequent other M^(2+) ions of the 3d series of elements, the 4d and 5d series metals generally do not form stable cationic species. |
|
Answer» Solution :(i) Cu (I) ion does not EXHIBIT sufficiently NEGATIVE value of enthalpy of hydration and it proportionates into 0 and +2 oxidation STATES of Cu. `2Cu^(+)toCu^(2+)+Cu` (II) 4d and 5d series transition metals have greater enthalpies of atomisation. Thus, there are strong metal-metal interactions between them. This is the REASON why they do not form stable cationic species. |
|
| 18. |
State reasons for the following: (i) Cu(I) ion is not stable in an aqueous solution. (ii) Unlike Cr^(3+)Mn^(2+), Fe^(3+) and the subsequent other M2+ ions of the 3d series of elements, the 4d and the 5d series metals generally do not form stable cationic species. |
| Answer» SOLUTION :(ii) DUE to lanthanoid contraction the expected increase in SIZE does not OCCUR. | |
| 19. |
State reasons for each of the following: Sulphur has greater tendency for catenation than oxygen. |
| Answer» Solution :Sulphur has greater tendency for catenation than oxygen. Oxygen has smaller atomic radius. Thus, there are STRONG `p pi- p pi ` bonds in `O_2` molecule. Sulphur being larger in size than oxygen, strong `ppi - p pi `bonds are not formed between two S ATOMS. Hence, S SATISFIES its valencies by catenation with other S atoms FORMING `S_8` molecule. Thus, while O forms `O_2` molecule, S forms `S_8`. | |
| 20. |
State reason for the following The molecular shape of Ni(CO)_4 is not the same as that of [Ni(CN)_4]^(2-) . |
|
Answer» Solution :Electronic configuration of: `NI : [AR] 4s^2, 3d^8` Ni (O) :`[Ar] 4s^0 , 3d^(10)`  `[Ni(CO)_4` has `sp^3` HYBRIDISATION and has TETRAHEDRAL shape. `[Ni(CN)_4)_2^(-)` `Ni^(2+) ,4s^0 3d^8` 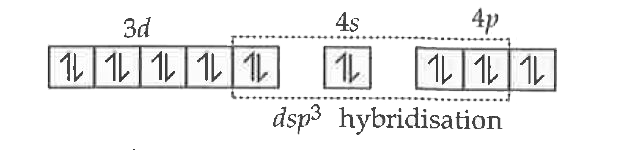
|
|
| 21. |
State reasons for each of the following: All the P-Cl bonds in PCl_5 molecule are not equivalent. |
|
Answer» Solution :All the P-Cl bonds in `PCl_5` molecule are not equivalent. The structure of `PCl_5` is GIVEN below :  It has trigonal bipyramidal structure as SHOWN. The three equatorial P-Cl bonds are equivalent, while the two axial bonds are longer than the equatorial bonds. This is DUE to the reason that axial BOND pairs suffer more repulsion as compared to equatorial bond pairs. |
|
| 22. |
State reason for the following CO is stronger complexing reagent than NH_3. |
| Answer» Solution :Since CO can form `sigma` as well as `pi` BOND, WHEREAS `NH_3` has LONE pair of ELECTRONS and can form `sigma` bond only. Therefore , COis better complexing reagent than `NH_3`. | |
| 23. |
State Raoult's law. Why is the vapour pressure of a solvent lowered by the addition of nonvolatile solute to it? A solution containing 18 g of non-volatile solute in 200 g of water freezes at 272.07 K. Calculate the molecular mass of the solute. (Given K_f = 1.86 K/m). |
|
Answer» Solution :Raoult.s law may be stated in two different .ways: (i) Solution containing a non-volatile solute. According to the law, the vapour pressure of the solution at a given temperature is equal to the product of the vapour pressure of the pure SOLVENT and its mole fraction. `p = p^@X_A` where p = vapour pressure of solution `p^@ =` vapour pressure of pure solvent `X_A =` mole fraction of pure solvent. (ii) Solution containing both yolatile solute and solvent. According to Raoult.s law, the vapour pressure of each component in the solution is equal to the product of the vapour pressure of the pure solvent and its mole fraction. `P_A= P_A^@ X_A, P_(B)^(@) X_B` When a non-volatile solute is added to a solvent (GENERALLY water), the vapour pressure of the solution gets lowered. Actually, evaporation is a surface phenomenon i.e. it takes PLACE from the surface of the container. The non-volatile solute particles occupy CERTAIN surface area and as a result, the tendency of the solvent molecules to change to vapours decreases. In other words, the vapour pressure of the solution falls. Numerical Mass of the solute (WB) = 18 g Mass of water (WA) = 200 g `Delta T_f =273 -272 .07 =0.93 K` `K_f =1.86 K//m` `M_b=?` We know that, `M_B =(K_f xx W_Bxx 1000 )/(Delta T_f xx W_A) ` `=(1.86 xx 18 xx 1000 )/( 0.93 xx 200 )=180.` |
|
| 24. |
State Raoult's l.iw for the solution containing voLttile components. Wh.it is the simi.Ltrity between Raoult's law and Henry's law? |
|
Answer» Solution :For the solution containing VOLATILE components, the PARTIAL vapour pressure of each component is directly proportional to its mole fraction. LN both case, P `PROP` x. Henry.s Law is a special case of Raoults Law. |
|
| 25. |
State Raoult's law for solutions containing non-volatile solutes in volatile solvents. Derive a mathematical expression for the law. |
|
Answer» Solution :Raoult.s law. It states that at any TEMPERATURE, PARTIAL vapour PRESSURE of a volatile component of a solution is equal to the vapour pressure of that component and its mole fraction in solutions. Let two volatile liquid components A and B of a solution has mole fractions `X_A and X_B` respectively. Let `P_(A)^(@) =` V.P. of pure A in solution. `P_(B)^@ = V.P.` of pure B in solution. `P_(A)` = Partial V.P. of A in solution. `P_B` = Partial V.P. of B in solution. According to Raoult.s law, `P_A = P_(A)^(@)X_A` `P_B = P_(B )^(@) X_B` Total V.P. of solution, `P = P_A + P_B ` `P = P_(A)^(@) X_A +P_(B)^(@) X_B` If the component B is a non-volatile solute. `P_B = 0` ` therefore P = P_A = P_(A)^(@) X_ A` `or (P_A)/(P_(A)^(@))=X_A` `(P_A)/(P_(A)^(@))=1-X_B` or ` 1-(P_A)/(P_(B) ^(@) )=X_B` `(P_A^(@)-P_A)/(P_(A)^(@))=X_B` |
|
| 26. |
State Raoult's law of a binary solution for two volatile liquid components. |
| Answer» Solution :For any solution the PARTIAL vapour pressure of each VOLATILE component in the solution is DIRECTLY proportional to its mole fraction. | |
| 27. |
State Raoult's law for solution containing non-volatile solute. |
|
Answer» <P> SOLUTION :The vapour PRESSURE of the solution `(P_A)` containing a non-volatile solute is equal to the product of the vapour pressure of pure SOLVENT `(P_(A)^@)` and its MOLE fraction XA. Mathematically,`P_A = P_A^(@)X_A` |
|
| 28. |
State Raoult's law for a solution containing volatile liquids. Explain with suitable example the concept of maximum boiling azeotropes. |
|
Answer» Solution :Raoult.s law : For a solution of volatile liquids, the partial vapour pressure of each componentin the solution is directly proportional to its MOLE fraction. Let us say that there are two components 1 and 2 in a solution. For component 1, `p_1 prop x_1`, where `p_1` and `x_1` are partial pressure and mole fraction of component 1. and `p_1 p_1^0 x_1` where `p_1^0`is the vapour pressure of pure component 1 at the same temperature. Similarly for component 2, `p_2 = p_1^0x_2` Maximum boiling azeotropes : SOLUTIONS that show large NEGATIVE deviations from Raoult.s law form maximum boiling azeotropes at a SPECIFIC composition. Nitric acid and water is an EXAMPLE of this type of azeotropes. This azeotrope has the approximate composition, 68% nitric acid and32% water by mass, with a boiling point of 393.5 K. |
|
| 29. |
State Raoult's law for a solutioncontaining volatile liquids. Explain with suitable example the concept of maximum boiling azeotropes. |
|
Answer» Solution :For Raoult's law, conslut SECTION 5 For maximum BOILING AZEOTROPE, consult section 7. |
|
| 30. |
State Raoult's law of liquid mixtures. |
| Answer» Solution :The vapour pressure of each LIQUID in the solution is EQUAL to the product of MOLE FRACTION of solute and VAPOURPRESSURE of the pure component. | |
| 31. |
State Raoult's law for a solution containing volatile components. What is the similarity between Raoult's law and Henry's law? |
|
Answer» Solution :For a solution of volatile liquids, the partial vapour pressure of each component of the solution is directly proportional to its MOLE FRACTION present in the solution. According to Raoult.s law , vapour pressure of a volatile component is given by `p_1-x_1p_1^u` According to Henry.s law `p=K_Hx` If we COMPARE the TWO equations, we find that partial pressure of the volatile component or gas is directly proportional to its mole fraction in solution. Only the proportionality constant `K_H` differs from `p_1^0`. Thus there is a SIMILARITY between the two laws. |
|
| 32. |
State Raoult's law for a solution containing volatile components. How does Raoult's law become a special case of Henry's law ? |
|
Answer» Solution :Raoult.s law : It states that for a solution of volatile liquids, the partial vapour pressure of eachcomponent in the solution is directly proportional to its mole fraction. Raoult.s law as a special case of Henry.s law : According to Raoult.s law vapour pressure of a volatile component in a given solution is given by `p_i = x_ip_i^0`In the solution of a gas in a liquid, ONE of the components is so volatile that it exists as a gas and its solubility is given by Henry.s Law as `p= K_H x ` where x is the mole fraction of the gas in the solution. On comparing the equations for Raoult.s law and Henry.s law, it can be SEEN that partial pressure of the volatile component or gas is directly proportional to mole fraction of that component. Only the proportionality constant `K_H`DIFFERS from `p_0^0` THUS Raoult.s law becomes a special case of Henry.s law in which `K_H`becomes equal to `p_i^0`. |
|
| 33. |
State Raoult' s law for a soluti.on of 2 volatile liquids. Give an example for liquid mixture that show negative deviation from Raoult's law. |
|
Answer» Solution :Partial Vapour PRESSURE of each volatile component in the solution is DIRECTLY proportional to its mole FRACTION. EXAMPLE : - Acetone + CHLOROFORM OR Nitric acid + Water. |
|
| 34. |
State Raoult's law for the solution containing volatile components. What is the similarity .between Raoult's law and Henry's law ? |
| Answer» Solution :STATEMENT of Raoult's LAW : The law STATES that, at CONSTANT temperature , the partial vapour pressureof any volatile component of a solution is equal to the product of vapour pressure of the PURE component and the moe fraction of that component in the solution . | |
| 35. |
State properties and uses of dichlorine. |
|
Answer» Solution :(i) Physical properties : It is a greenish-yellow gas with pungent and suffocating odour. It is about 2-5 times heavier than air. It is soluble in water and can be liquefied easily into yellow liquid which boils at 239K TEMPERATURE. (ii) Chemical properties : Reaction with metals and non-metals : Chlorine reacts with all non-metals EXCEPT NITROGEN, oxygen, carbon and inert gases. Chlorine with metals and non-metals form chlorides. `2Al + 3Cl_2 to2AlCl_3 , P4 + 6Cl_2 to4PCl_3 2Na + Cl_2 to 2NaCl , S_8 + 4Cl_2to 4S_(2)Cl_(2)` `2Fe + 3Cl_(2) to 2FeCl_(3)`, Affinity for hydrogen : Chlorine has very strong affinity for hydrogen. It reacts with compounds containing hydrogen to form HCl `2 + Cl_2 to2HCl` `H_2S + Cl_2 toS + 2HCl` `C_10H_16 + 8Cl_2 to 16HCl + 10C` Reaction with alkalies : With cold and dilute alkalies chlorine produces a mixture of chloride and hypochlorite but with hot and concentrated alkalies it GIVES chloride and chlorate. `2NaOH + Cl_(2) to NaCl + NaOCl + H_(2)O` `6NaOh + 3Cl_(2) to 5NaCl + NaClO_(3) + 3H_(2)O` Reaction with ammonia : With ammonia, the products obtained are ammonium chloride and dinitrogen. `3Cl_(2) (g) + 8NH_(3)(g) to N_(2)(g) + 6NH_(4)Cl(s)` With excess dichlorine the product is `NCl_3` (explosive) `NH_(3)(g) + 3Cl_(2)(g) to NCl_(3) + 3HCl(g)` Bleaching action and oxidizing action: Chlorine water on standing loses its yellow COLOUR due to the formation of HCl and HOCl. Hypochlorous acid so formed, gives nascent oxygen which is responsible for oxidising and bleaching properties of chlorine. `Cl_2 + H_2O toHOCl + HCl` `HOCl to HCl + [O]` `2FeSO_4 + H_2SO_4 + Cl_2 to Fe_2(SO_4)_3 + 2HCl` `Na_2SO_3 + Cl_2 + H_2O toNa_2SO_4 + 2HCl SO_2 + 2H_2O + Cl_2 toH_2SO_4 + 2HCl` `I_2 + 6H_2O + 5Cl_2 to2HlO_(3) + 10HCl` The bleaching action of chlorine is permanent as it takes place through oxidation. It bleaches vegetable and organic matter in presence of moisture. Coloured matter + [O] `to` colourless substance Formation of bleaching powder : With dry slaked lime, it gives bleaching powder. `2Ca(OH)_(2) + 2Cl_(2) to Ca(OCl)_(2) + CaCl_(2) + 2H_(2)O` The composition of bleaching powder is `Ca(OCl)_(2).CaCl_(2).Ca(OH)_(2).2H_(2)O` Reaction with hydrocarbons : With saturated hydrocarbons, dichlorine gives substituted products and with unsaturated hydrocarbons it gives addition products. `CH_(4) + Cl_(2) overset(hv)to underset("Methyl chloride")(CH_(3)Cl + HCl)` `C_(2)H_(4) + Cl_(2) overset("Room")underset("Temperature")to underset(1,2 "dichloroethane")underset(Cl)overset(CH_(2))| - underset(Cl)overset(CH_(2))|` Uses of dichlorine : For bleaching wood pulp (required for the manufacture of paper and rayon), cotton and textiles. In extraction of gold and platinum. In the manufacture of dyes, drugs and organic compounds such as `C Cl_4, CHCl_3`, DDT, refrigerants etc. In sterilising drinking water and in preparation of poisonous gases such as phosgene (`COCl_2`), tear gas (`C Cl_3NO_2`), mustard gas: `[Cl-CH_(2)CH_(2)-S-CH_(2)CH_(2)-Cl]` |
|
| 36. |
State physical properties of potassium permanganate and state its uses |
|
Answer» Solution :Potassium permanganate forms dark purple crystals which are isostructural with `KCIO_(4)`. The salt is not very soluble in water [6.4g/100g of water at 293K] It is diamagnetic and has intense colour. It also shows TEMPERATURE dependent weak paramagnetism Uses: It is used as oxidizing agent in preparative ORGANIC chemistry. It is used for BLEACHING of wool, cottom, silk and othe testile fibres and TEH decolourisation of oils are also dependent on its strong oxidizing power. It ACT as disinfectant and used as germicide |
|
| 37. |
State product and its colour when MnO_(2) reacts with KOH in presence of air? |
|
Answer» `KMnO_(4)`- PURPLE |
|
| 38. |
State physical and chemical properties of phosphine (PH_(3)) |
|
Answer» Solution :(i) PHYSICAL properties : `PH_3` is colourless gas with rotten fish smell and is highly poisonous. It is SPARINGLY soluble in water. Chemical properties : Phosphine explodes in contact with traces of oxidizing agents like `HNO_(3), Cl_2` and `Br_2` vapours. The solution of phosphine in water decomposes in presence of LIGHT giving red phosphorus and `H_2`. When absorbed in COPPER sulphate or mercuric chloride solution, the corresponding phosphides are obtained: `3CuSO_(4) + 2PH_(3) to Cu_(3)P_(2) + 3H_(2)SO_(4)` `3HgCl_(2) + 2PH_(3) to Hg_(3)P_(2) + 6HCl` Phosphine is weakly basic and like ammonia it gives phosphonium compounds with acids, e.g., `PH_(3) + HBr to underset("Phosphonium BROMIDE")(PH_(4)Br)` |
|
| 39. |
State perfect order of ionic radii. |
|
Answer» `Lu^(3+) LT Yb^(3+) lt Eu^(3+) lt La^(3+)` `""_(71)Lu lt ""_(70)Yb lt ""_(63)Eu lt ""_(57)La` |
|
| 40. |
State one use each of DDT and iodoform. |
|
Answer» SOLUTION :USE of DDT : It is USED as an INSECTICIDE. Use of IODOFORM: It is used as an antiseptic. |
|
| 41. |
State one use of acetonitrile. |
| Answer» Solution :It is USED in non-aqueous POLAR SOLVENT organic and INORGANIC COMPOUNDS. | |
| 42. |
State one use for the enzyme streptokinase in the medicine ? |
| Answer» Solution : It DISSOLVES BLOOD clots in CORONARY ARTERIES. | |
| 43. |
State one difference between an emulsion and a sol. |
| Answer» Solution :An EMULSION is a COLLOIDAL solution of a LIQUID dispersed in a liquid whereas a SOL is a colloidal solution of a solid dispersed in a liquid. | |
| 44. |
State one condition in which a bimolecular reaction may be kinetically of the first order? |
| Answer» SOLUTION :By TAKING ONE of the reactants in large excess so that it MAY not contribute towards the ORDER. | |
| 45. |
(a) (i) State Ohm's law.(ii) With the help of a circuit diagram derive the formula for the resultant resistance of three resistances connected in parallel. |
|
Answer» SOLUTION :This law can be stated as, "at constant temperature, the strength of the CURRENT flowing through a CONDUCTOR is directly proportional to the potential difference and inversely proportional to the resistance of the conductor". Thus, `I=V/R, V=RI, V="VOLTS", I="ampere", R="OHMS"`. |
|
| 47. |
State oftherole ofsilicain themetallurgyofcopper. |
|
Answer» SOLUTION :During roasting, copperpyrites are convertedintoamixtureofFeOand`Cu_ 2O`. `""underset ("Copper pyrites")(2CuFeS_2 )+O _ 2overset (Delta) toCu_2S +2Fe S +SO _ 2` `""2Cu_ 2S+3O _ 2overset (Delta ) to2 Cu_ 2O+2 SO _ 2and2 FES +3 O _ 2to2 FeS +3O _ 2to2 FeO+2 SO _2 ` ToremoveFeO(basic ), an acidicflux silicaisaddedduringsmelting. FeOthen combineswith` SiO _ 2`toformferroussilicate` (FeSiO_ 3 ) `slag whichfloatsover moltenmatteand hencecan beeasilyremoved `"" FeO+underset ("FLUX")(SiO_2) tounderset ("Slag")(FeSiO_3) ` Thus,therole ofsilica inmetallurgyofcopperis toremoveferrous OXIDE as ferroussilicateslag. |
|
| 48. |
State of hybridization of central atom and number of lone pairs over central atom in XeF_4 are : |
|
Answer» `SP, 0` |
|
| 49. |
What is meant by 'chemical equilibrium ' ? |
|
Answer» DYNAMIC |
|
| 50. |
State of chemical equilibrium is : |
|
Answer» DYNAMIC |
|