Saved Bookmarks
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
State occurence of group-18 elements. |
|
Answer» Solution :All elements of GROUP-18 except RADON and oganesson occur in the atmosphere. They are gases, (except radon and oganesson). The atmospheric abundance in dry air is 1% by volume of which argon is the major constituent. Helium and sometimes neon are found in minerals of radioactive origin e.g., pitchblende, monazite, cleveite. The main commercial SOURCE of helium is natural gas. Xenon and radon are the RAREST elements of the group. Radon is OBTAINED as decay product of `""^(226)Ra` `""_(85)^(226)Ra to ""_(86)^(222)Rn + 4/2He` Oganesson has been synthetically produced by collision of `""_(98)^(249)Cf + ""_(20)^(48)Ca to ""_(118)^(294)Og + 3(1/0)n` Oganesson has atomic mass of 294 and half life of 0.7 milliseconds. Its atomic number is 118. |
|
| 2. |
State occurence of group-17 elements. |
|
Answer» Solution :(i) Fluorine : Fluorine is present mainly as insoluble fluorides such as fluorspar (`CaF_2`), cryolite (`Na_3AlF_`6) and fluoroapatite [`3Ca_3(PO_4)_2. CaF_2`] and small quantities are present in soil, river water plants and BONES and teeth of animals. (ii) Chlorine : Sea water mainly contains 2.5% by mass of sodium chlorides. The deposits of dried up seas contain these compounds, e.g., sodium chloride (NaCl), carnallite (KCl-`MgCl_2-6H_2O`). (iii) Bromine and iodine : Apart from sodium chloride, sea water also contains bromides and iodides of potassium, magnesium and calcium. EX. : NaBr, KBr, `MgBr_2` etc. while iodine is mainly obtained as iodates (NaI03). certain marine life contain iodine in their systems, various seaweeds, for example, contain upto 0.5% of iodine and chile saltpetre contains upto 0.2% of sodium iodate. (iv) Tennessine : Tennessine is a synthetic radioactive element. (At. Mass = 294). Only small amount of the element could be prepared as its half life is in milliseconds only. In the periodic table, the Group-17 only contains the element in all three states of matter. All are reactive non-metals. Fluorine is used in toothpaste and refrigerant GASES, chlorine for bleach and chemical warfare, bromine in fire retardant material while iodine in ANTISEPTICS. |
|
| 3. |
State : Kohlrauseh's law |
| Answer» Solution :The LIMITING molar conductivity of an ELECTROLYTE can be represented as the SUM of the limiting molar conductivities of the INDIVIDUAL CATION and anion. | |
| 4. |
State Kohlrausch's law and write mathematical expression of molar conductivity of the given solution at infinite dilution. |
|
Answer» Solution :Statement of Kohlrausch's LAW : This states that at infinite dilution of the solution, each ion of an electrolyte migrates indepef!dently of its co-IONS and contributes independently to the total molar conductivity of the electrolyte, irrespective of the nature of other ions present in the solution This law of independent MIGRATION of ions is represented as `^^_(0) = lambda_(+)^(0) + lambda_(-)^(0)`. Where `^^_(0)`is the molar conductivity of the electrolyte at infinite dilution or ZERO concentration while `lambda_(+)^(0)`and `lambda_(-)^(0)`arethe molarionicconductivites of cationand anionrespectively atinfintedilution. |
|
| 5. |
State Kohlrausch law of independent migration of ions. Why does the conductivity of a solution decreases with dilution? |
|
Answer» SOLUTION :Kohlrausch law of independent migration of IONS: The law states that limiting molar CONDUCTIVITY of an electrolyte can be STATE as the sum of the individual contributions of the anion and cation of the electrolyte. On dilution, the conductivity (K) of the electrolyte decreases as the number of ions per unit volume of solution decreases. |
|
| 6. |
State Kohlrausch law of independent migration of ions. Why does the conductivity of a solution decrease with dilution ? |
|
Answer» Solution : Kohlrausch LAW of independent MIGRATION of ions states that limiting molar conductivity of an electrolyte can be represented as the SUM of the individual contributions of anion and cation of the electrolyte. If `Lambda_(Na^(+))^(@)`and `Lambda_(Cl^(-))^(@)`are the limiting molar conductivities for SODIUM and chloride ions respectively, then limiting molar conductivity for sodium chloride is given by the EQUATION : `Lambda_(m)^(@) (NaCl) = lambda_(Na^(+))^(@) + lambda_(Cl^(-))^(@)` Conductivity of a solution decreases with dilution because the number of ions per unit volume of the solution decreases. |
|
| 7. |
State Kohlrausch Law. How is it useful to determine the molar conductivity of weak electrolyte at infinite dilution. |
|
Answer» Solution :Kohlrausch.s LAW: It is defined as, at infinite dilution the limiting molar conductivity of an electrolyte is equal to the sum of the limiting molar conductivities of its constituent ions. Determination of the molar conductivity of weak electrolyte at infinite dilution: It is impossible to DETERMINE the molar conductance at infinite dilution for weak electrolytes experimentally. However, the same can be calculated using Kohlraush.s Law. For EXAMPLE, the molar conductance of `CH_(3)COOH`, can be calculated using the experimentally determined molar conductivities of strong electrolytes HCl, NaCl and `CH_(3)COONa`. `Lambda^(@)""_(CH_(3)COONa)=lambda^(@)""_(Na^(+))+lambda^(@)""_(CH_(3)COO^(-))""..................(1)` `Lambda^(@)""_(HCl)=lambda^(@)""_(H^(+))+lambda^(@)""_(Cl^(-))""......................(2)` `Lambda^(@)""_(NaCl)=lambda^(@)""_(Na^(+))+lambda^(@)""_(Cl^(-))""....................(3)` Equation (1) + Equation (2) - Equation (3) gives, `(Lambda^(@)""_(CH_(3)COONa))+(Lambda^(@)""_(HCl))-(Lambda^(@)""_(NaCl))=lambda^(@)_(H+)+lambda^(@)""_(CH_(3)COO^(-))=Lambda^(@)CH_(3)COOH)` |
|
| 8. |
State Kohlrausch Law. How is it useful to determine the molar conductivity of weak electrolyte at infinite dilution? |
|
Answer» SOLUTION :Kohlraush.s law :At infinite dilution, the limiting molar conductivity of an electrolyte is equal to the some of the limiting molar CONDUCTIVITIES of its constituent ions. Calculation of molar conductance at infinite dilution of a WEAK electrolyte. It is impossible to determine the molar conductance at infinite dilution for weak electrolytes experimentally. However, the same can be calculated using Kohlraush.s Law. For EXAMPLE, the molar conductanceof `CH_(3)COOH`, can becalculated using the experimentally determined molar conductivities of strong electrolytes HCl,NaCl and `CH_(3)COONa`. `wedge_(CH_(3)COONa)^(@)=lambda_(Na^(+))^(@)+lambda_(CH_(3)COO^(-))^(@) "...(1)"` `wedge_(HCl)^(@)=lambda_(H^(+))^(@)+lambda_(Cl^(-))^(@)"...(2)"` `wedge_(NaCl)^(@)=lambda_(Na^(+))^(@)+lambda_(Cl)^(@)"...(3)"` Equation (1) + Equation (2) - Equation (3) gives. `(wedge_(CH_(3)COONa)^(@))+(wedge_(HCl)^(@))-(wedge_(NaCl)^(@))=lamda_(H^(+))^(@)+lambda_(CH_(3)COO)^(@)` `""=wedge_(CH_(3)COOH` |
|
| 9. |
State Kohlrausch law of independent migration of ions. |
| Answer» Solution :The LIMITING molar conductivity of an electrolyte can be represented as the SUM of the INDIVIDUAL contributions of the ANION and cation of the electrolyte. | |
| 10. |
State Kohlrauseh law. |
| Answer» Solution :At infinite dilution, the LIMITING MOLAR conducting of an electrolyticsolution is equal to sum of the molar CONDUCTANCES of all the IONS PRESENT in it. | |
| 11. |
State Kohlrausch law. |
| Answer» SOLUTION :Kohlrausch LAW states that the LIMITING molar conductivity of an electrolyte can be represented as the SUM ofthe individual contributions of the anion and cation of the electrolyte. | |
| 12. |
State Kohirausch Law. How is it useful to determine the molar conductivity of weak electrolyte at infinite dilution. |
|
Answer» Solution :Kohlrausch.s law : It is defined as, at infinite dilution the limiting molar conductivity of an electrolyte is equal to the SUM of the limiting molar conductivities of its constituent ions. Determinatin of the molar conductivity of weak electrolyte at infinite dilution: It is impossible to DETERMINE the molar conductance at infinite dilution for weak electrolytes experimentally. However, the same can be calculated USING Kohlraush.s Law. For example , the molar conductance of `CH_3COOH`, can be calculated using the experimentally determined molar conductivities of strong electrolytes HCl, NACL and `CH_3COONa`. `Lambda_(CH_3COONa)^@ = lambda_(Na^+)^@ + lambda_(CH_3COO^(-))^@""..........(1)` `Lambda_(HCl)^(@) = lambda_(H^+)^@ = lambda_(H^+)^@ + lambda_(Cl^-)^(@) "".......(2)` `Lambda_(NaCl)^@ = lambda_(Na^+)^@ + lambda_(Cl^-)^@ ""................(3)` Equation (1) + Equation (2) - Equation (3) gives `(Lambda_(CH_(3)COONa)^(@))+(Lambda_(HCl)^(@))-(Lambda_(NaCl)^(@))=Lambda_(H^(+))^(@)+lambda_(CH_(3)COO^(-))^(@)=Lambda_(CH_(3)COOH)^(@)` |
|
| 13. |
State importance of carbohydrates. |
|
Answer» Solution :Carbohydrates are essential for life in both plants and animals. They FORM major portion of our food. Honey has been used for a long time as an instant source of energy by "Vaids" in ayurvedic system of medicine. Carbohydrates are used as storage molecules as starch in plants and glycogen in animals. CELLWALL of bacteria and plants is made of CELLULOSE. The furniture is the cellulose in the form of wood and clothe ourselves with cellulose in the form of cotton fibre. Carbohydrates PROVIDE the raw materials for many important industries like textiles, paper, lacquers and breweries. Two aldo-pentoses, D-ribose and 2-deoxy- D-ribose are present in nucleic acids. Carbohydrates are found in biosystem in COMBINATION with many proteins and lipids. |
|
| 14. |
State Henry's Law. Write its one application. What is the effect of temperature on the solubility of a gas in solution. |
|
Answer» Solution :For the statement of HENRY's Law, CONSULT SECTION 3. Oxyegen to be used by deep sea divers is genrally diluted with heliuym in order to reduce painful effects during DECOMPRESSION. Solubility of a gas in a solution generally DECRESES with rise in temperature. |
|
| 15. |
State Henry's law. Write its one application. What is the effect of temperature on solubility of gases in liquid ? |
|
Answer» Solution : Henry.s law states : The solubility of a gas in a liquid is directly proportional to the pressure ofthe gas. Application : To INCREASE the solubility of `CO_2`in soft drinks and soda WATER, the bottle is SEALED under high pressure. Effect of temperature : Solubility of a gas in a liquid decreases with rise of temperature. As dissolution is an exothermic PROCESS, the solubility should decrease with rise of temperature according to Le chatelier.s PRINCIPLE. |
|
| 16. |
State Henry's law. What is the effect of temperature on the solubility of a gas in a liquid ? |
|
Answer» Solution :Henry's Law states that the SOLUBILLITY of a gas in a liquid is directly proportional to the PRESSURE of the gas. If we use the mole fraction of a gas in the solution as a measure of its solubility ,then it can be said that the mole fraction of gas in the solution is proportional to the partial pressure of the gas over the solution. The most commonly used FORM of Henry's Law states that''the partial pressure of a gas in vapour phase (p) is directly proportional to the mole fraction of the gas (X) in the solution,i.e `p propto x` `p =K_(H)x` The solubility of gases is dependent on temperature. An increase in temperature results in a decrease in gas solubility in liquid, while a decrease in temperature results in an increase of gas solubility in liquid . As increase in temperature causes an increase in kinetic energy, resulting in a more rapid motion of MOLECULES, breaking intermolecular bonds which enable molecular to escape from the solution allowing the gases dissolved to evaporate more readily. Hence causing decrease in the solubility of gases in liquids. |
|
| 17. |
State Henry's law. Give its mathematical form. |
|
Answer» Solution :At CONSTANT temperature the solubility the solubility of a GAS in a liquid is directly proportional to the PARTIAL PRESSURE of the gas present above the surface of the solution. `p=K_(H).x` p,partial pressure of the gas. x: mole fraction of the gas. `K_(H)`: HENRY's constant. |
|
| 18. |
State Henry's Law for the solubility of a gas in liquid. Explain the singinficance of Henry's Law constant (K_(H). At the same temperature, hydrogen is more soluble in water than helium. Which of them has higher K_(H) value and why ? |
| Answer» SOLUTION :He has HIGHER `K_(H)` VALUE. | |
| 19. |
State Henry's law. Calculate the solubility of CO_2 in water at 298 K under 760 mm Hg. [K_(H) for CO_2 in water at 298K is 1.25 xx 10^(6) mm Hg] |
|
Answer» Solution :Henry.s law : At constant temperature, the solubility of a gas in a liquid is directly PROPORTIONAL to the partial pressure of the gas PRESENT above· the surface of the liquid or solution. `p=K_(H).x` here, `p=760 mm, K_(H)=1.25 xx 10^(6)` mm Substih1ting the values in the equation above, we have `x=(p)/( K_(H) )= (760mm)/(1.25 xx 10^(6) mm) = 608 xx 10^(-6) =6.08 xx 10^(-4)` Mole fraction of `CO_2` in WATER `=(n)/(n+55.5) = (n)/(55.5)` (n can be neglected as compared to 55.5) n is the number of moles of `CO_2` and 55.5 is the number of moles of water in one litre. `6.08 xx 10^(-4) = (n)/(55.5)` or `n=337.44 xx 10^(-4) mol` or `3.37 xx 10^(-2)` mol `L^(-1)` |
|
| 20. |
State Henry's law and mention two ofil8important applications. |
|
Answer» Solution :Henry.s law : Ref.Q. 3 of V.S.A.T.Q. Applications of Henry.s law : (i) To increase the solubility of `CO_(2)` in SODA WATER and SOFT drinks , the bottle is sealed under high pressure. (ii) At high ALTITUDE , low blood oxygen causes climber to BECOME weak and make them unable to think clearly, which are symptoms of a condition known as anoxia. |
|
| 21. |
State Henry's law and mention some important applications. |
|
Answer» Solution :Henry.s law : The solubility of the amount of a gas in a liquid is directly proportional to the pressure of gas. Applications of Henry.s law : (i) Deep SEA divers use He and `O_2` mixture for RESPIRATION in place of `N_2` and `O_2` because `N_2` being more soluble in blood at high pressure and creates the painful effect on the human body. (ii) To make soft drink bottles rich in `CO_2`, SEALING of the BOTTLE is done at high pressure. |
|
| 22. |
State Hardy-Schulze rule which deals with the coagulation of colloids by the addition of an electrolyte. |
|
Answer» As the charge on the ion INCREASE, the COAGULATING power INCREASES. |
|
| 23. |
State Hardy-Schulze rule. |
| Answer» SOLUTION :HIGHER the VALENCY of the active ion, more will be its PRECIPITATING power. | |
| 24. |
Define Hardy -Schulze rule. |
| Answer» Solution :GREATER the charge of the ACTIVE ion (or flocculating ion), greater will be its COAGULATING POWDER. | |
| 25. |
State and discuss Hardy-Schulze rule. |
| Answer» Solution :GREATER the charge of the active ION (or flocculating ion), greater will be its COAGULATING POWDER. | |
| 26. |
State Faraday's second law of electrolysis. |
| Answer» Solution :When same quantity of electricity is passed through DIFFERENT electrolytes, the masses of the SUBSTANCE DISCHARGED at the electrodes are directly PROPORTIONAL to their EQUIVALENT masses. | |
| 27. |
State Faraday's secondlawof electrolysis |
|
Answer» Solution :Second Law :When the samequantity of chargeis passedthroughthe solutionsof differentelectrolytes, the amountof substancesliberatedat therespectiveelectrodes are directlyproportionalto theirelectrochemicalequivalents. When Q coulombchargeis passedthroughthe electrolytyiccellsthe masses of silver,zinc and copperdepositedat therespectiveelectrodesbe ` m_(Ag)m_(ZN) and m_(cu)` RESPECTIVELY . ACCORDINGTO Faraday's second Law, ` m_(Ag) PROP Z_(Ag) m_(Zn) and m_(Cu) prop Z_(Cu)` or ` (M_(Ag))/(Z_(Ag)) = ( M_(Zn))/(Z_(Zn)) = (M_(Cu))/(Z_(Cu))` |
|
| 28. |
State Faraday's Laws of electrolysis. |
|
Answer» Solution :Faraday.s law of ELECTROLYSIS : (i) FIRST law : The mass of the substance (M) liberated at an electrode during electrolysis is directly proportional to the quantity of charge (Q) passed through the cell. `M alpha Q` (ii) Second law : When the same qunatity of charge is passed through the solutions of different electrolytes, the amount of substances liberated at the RESPECTIVE ELECTRODES dand directly proportional to their electrochemical equivalents. `M alpha Z`. |
|
| 29. |
State Faraday's First law of electrolysis. Write its mathematical form using usual notations. |
|
Answer» Solution :Faraday.s first law of electrolysis states that The amount of chemical reaction which OCCURS at any electrodeduring electrolysis by a CURRENT is directly proportional to the quantity of electricity passed through the ELECTROLYTE during electrolysis. It is mathematically REPRESENTED as. W = ZIt |
|
| 30. |
State faraday's first law of electrolysis. Uses of: (a)H_(2)SO_(4)""(b)Chlorine Disitinguish between crystalline solids and amorphus solids. A solution of a substance having mass 1*8xx10^(-3)kg has the osmotic pressure of 0*52 atm at 280 K. Calculate the molar mass of the substance used. [Volume =1 dm^(3), ,R=8*314JK^(-1) mol^(-1)] |
|
Answer» Solution :Farday's first law electrolysis states that, "The amount of substacne which is DEPOSITED on an electrode during electrolysis is directly praporational to the amount of electricity which passes through the electrolyte". Use of Sulphuric acid : (a) The major use of sulphuric acid is in the production of fertilizers, for EXAMPLE, superphosphate of lime and ammonium sulphate. (b) It is wadel use in the manufacture of chemicals, such as in making hydrochloric acid, nitric acid, sulphate slats, synthetic detergents, dyes and pigments, explosives and drugs. USES of CHLORINE : (i) Chlorine is commonly used as an antiseptic and is used to make drinking water safe and to treat swimming pools. (ii) Large amounts of chlorine are used i many industrial processes, such as in the production of paper products, plastics, dyes, textiles, medicines, antiseptics, insecticides, solvents and paints. (III) We know that, osmotic pressure is given by the formula, `MB=- W_(B)RT//piV` `=(1.8xx10^(3))(8.314)280//0.52xx1` `=(4190.25xx140^(-3))//0.52` `=8058.1xx10^(-3)=8.0581g//mol` |
|
| 31. |
State Faraday's first law of electrolysis . |
| Answer» SOLUTION :Faraday's firstlaw of ELECTROLYSIS :It STATES that theamount ofthe substanceproduced due to oxidationor reductionat electrodeduring electrolysis is directlyproportional to thequantity of electricitythat passesthroughthe ELECTROLYTIC CELL. | |
| 32. |
State Farday's first law of electrolysis. For the electrode reaction Zn^(+2)+2e^(-)rarrZn_((s)), what quantity of electricity in coloumbs is required to deposite one mole of zinc. |
|
Answer» Solution :To deposit 1 MOLE of ZINC from `Zn^(2+)` ION. `96500 xx 2` COULOMBS are required. |
|
| 33. |
State : Faraday first law of electrolysis |
|
Answer» Solution :The AMOUNT of substance DEPOSITED at an electrode is DIRECTLY PROPORTIONAL to quantity of current passed through it.s solution. Mathematically `WpropQimpliesWpropItimpliesW=Z It` Where, W = mass, 1 = current in ampere, t = time in second, Z = electrochemical equivalent |
|
| 35. |
State EAN rule for co-ordination compounds. |
| Answer» Solution :A central METAL atom/ion in a co-ordination compound goes on accepting electron phase from the ligands until `ul("the total number")` of ELECTRONS around it becomes equal to the atomic `ul("number of NEXT nearest noble GAS")`. | |
| 36. |
State differences between the following pairs : (i) alpha-Helix and beta-pleated structures. (ii) Primary and secondary structure of protein. (iii) Enzymes and coenzymes. |
|
Answer» Solution :(i) `alpha`-Helix structure. The PEPTIDE chain coils and the turns of the coil are held together by H-bonds between amide groups giving spiral structure called `alpha`-Helix structure. Myosin, keratin and wool have a-helix structure. `beta`-pleated structure. The peptide chains lie side by side held together by intermolecular H-bonding,resulting in the FORMATION of flat sheet open structure called `beta`-pleated structure. Silk protein fibroin has `beta`-pleated sheet structure . (ii) Primary structure of proteins refers to the sequence in which amino acids are arranged in proteins. It involves covalent bonds. Secondary structure of proteins refers to the arrangement of polypeptide chains. It involves hydrogen bonding. (iii) Enzymes are biological catalysts which are required to CATALYSE biochemical reactions e.g. amylase, STREPTOKINASE, maltase etc. All enzymes are proteins. Coenzymes are non-proteinaceous part which INCREASE the activity of enzymes e.g. metal ions like `Mg^(2+), Zn^(2+), Mn^(2+)` etc. |
|
| 37. |
State chemical tests to distinguish between the following pairs of compounds: (i) Propanal ad propanone. (ii) Phenol and bennzoic acid. |
|
Answer» Solution :(i) Propanal and propanone: The two componds can be distinguished by iodoform TEST. Propanone contains `CH_(3)CO`- group and gives iodoform test while propanal `(CH_(3)CH_(2)CHO)` does not give iodoform test. `CH_(3)COCH_(3)+3NaOItoCH_(3)COONa+2NaoH+underset("Iodoform")(CHI_(3))` (ii) Phenol and benzoic acid: The two compounds can be distinguished as under: Ferric chloride test: Phenol gives a violet colour with aqueous `FeCl_(3)` while benzoic acid gives buff coloured ppt. of ferric benzoate. < br> `underset("Phenol")(C_(6)H_(5)OH)+FeCl_(3)to` violet colour `underset("Benzoic acid")(3C_(6)H_(5)COOH)+FeCl_(3)tounderset(("Buff coloured ppt"))underset(Ferric benzoate")((C_(6)H_(6)COO)_(3)Fe+3HCl` Sodium BICARBONATE test: Phenol is a weaker acid than benzoic acid. While benzoic acid DECOMPOSES sodium bicarbonate, phenol does not. `C_(6)H_(5)COOH+NaHCO_(3)toC_(6)H_(5)COONa+CO_(2)uarr+H_(2)O` `C_(6)H_(5)OH+NaHCO_(3)to` No action. |
|
| 38. |
State clearly what are known as nucleotides and nucleosides. |
|
Answer» SOLUTION :A NUCLEOSIDE contain only TWO basic components of nucleic acids i.e., pentose sugar and nitrogenous BASE. A nucleotide contains all the three basic components of nucleic acids i.e., a phosphoric acid group, pentose sugar and nitrogenous base. |
|
| 39. |
State briefly the principle involved in the following operations in mettalurgy. Give an example. Zone refining. |
| Answer» SOLUTION :This method of refining of metals is BASED on the principle that impurities are more soluble in the melt than in the solid form of the metal. A circular MOBILE HEATER is fixed at one end of a rod of IMPURE metal. The melton zone moves along with the heater which is moved forward. As the heater moves forward, pure metal crystallises out of the melt and impurities pass on to the adjacent molten zone. This method is very useful for producing semiconductors and other metals of very high purity, e.g., germanium, silicon etc. | |
| 40. |
State briefly the principle involved in the following operations in mettalurgy. Give an example. Hydraulic washing. |
| Answer» Solution :This method of separation is based UPON th difference in GRAVITIES of the ore and the gangue particles. An UPWARD stream of running water is used to wash the powdered ore. The lighter gangue particles are washed away and the heavier ore particles are left behind. The method is used to separate impurities from the ores of IRON and copper. | |
| 41. |
State biological importance of hormones. |
|
Answer» Solution :(i) Peptide hormones : The role of insulin maintains the blood sugar level within the narrow limit. Insulin is RELEASED in response to rapid rise in glucose level in glucose. On the other hand, hormone glucagon tends to increase the glucose level in the blood. These two hormones together regulate the glucose level in the blood. (ii) Amine hormones : Norepinephrine mediate responses to external stimuli. Thyroxine hormone produced by the thyroid gland is an iodinated derivative of amino acid tyrosine. Abnormally low level of thyroxine leads to hypothyroidism which is characterised by lethargyness and obesity. Increased level of thyroxine causes hyperthyroidism. Low level of iodine in the diet may lead to hypothyroidism and enlargement of the thyroid gland. This condition is largely being controlled by the Epinephrine and addition of sodium iodide to the commercial table salt (Iodised salt). (iii) Steroid hormones : Steroid hormones are produced by adrenal CORTEX and gonads (i.e., testes in males and ovaries in females). Hormones produced by the adrenal cortex plays very important role in the functions of the body. For example, glucocorticoids control the carbohydrate metabolism, modulate inflammatory reactions, and are involved in reactions to stress. The mineralocorticoids control the level of excretion of water or salt by the kidney. If adrenal cortex does not function properly then of the RESULTS may be Addison.s disease characterised by hypoglycaemia, weakness and increased susceptibility to stress. The disease is fatal unless treated by glucocorticoids and mineralocorticoids. Hormones released by gonads are responsible for development of secondary sex characters. Testosterone is a major sex hormone produced in males. It is responsible for development of secondary male characteristics such as deep voice, facial hair and general physical CONSTITUTION. Estradiol is the main female sex hormone which is responsible for the development of female characteristics and PARTICIPATES in control of menstrual cycle. Progesterone is responsible for preparing the uterus for implantation of fertilised egg. |
|
| 42. |
State biological importance of nucleic acid. |
|
Answer» Solution :It has been observed that nucleus of the LIVING CELL is responsible for the transmission of inherent characters, also called heredity. The PARTICLES in nucleus of the cell, responsible heredity are called chromosomes which are made of proteins and nucleic acids. DNA is exclusively responsible for maintaining identity of different species of organisms for million of YEARS. A DNA molecule is capable of self-duplication during the cell division and identical DNA strands are transferred to daughter cells. The nucleic acid plays an IMPORTANT role in protein synthesis. The proteins are synthesised by various RNA molecules in the cell but the message for the synthesis of a protein is present in DNA. |
|
| 43. |
State any three postulates of Werner theory of co-ordination compounds? |
|
Answer» Solution :i. In coordination compounds, METAL central metal atom or ion EXHIBIT two types of valencies. Primary valency (ionisable valency) and Secondary valency (nonionisable valency) ii. Primary valency of a metal is the charge on the metal and secondary valency is its coordination number of the complex ion that is the number of covalent BONDS formed by ligands with central metal ion. iii. Primary valencies of a metal ion are SATISFIED by negative ions and the secondary valency is satisfied by negative ions or NEUTRAL molecules which act as ligands. iv. The secondary valency of a metal is arranged in space to give a definite geometrical shape to the complex depending on the number of ligands. |
|
| 44. |
State any one characteristic of enzymatic catalysts. |
| Answer» SOLUTION :These CATALYSTS are HIGHLY SPECIFIC in their ACTION. | |
| 45. |
State any one condition under which a bimolecular reaction may be kinetically of first order. |
| Answer» Solution :A bimolecular REACTION may BECOME kinetically of first order if one of the REACTANTS is present in EXCESS. | |
| 46. |
State and explainvan't Hoff- Avogardo's law. |
|
Answer» Solution :Statement of van't Hoff-Avogadro's law : At given temperature, equal volumes of the solutions having the same osmotic pressurecontain number of solute particles. Explanation : The equal number of moles of the SUBSTANCES contain equal number of molecles. The osmotic PRESSURE, `pi`, is given by `pi = CRT` where C is the concentrationof a solution is mol `dm^(-3)`. `pi = n/V Rt :. pi prop (n)/(V)` at CONSTANT temperature . If two solutions contain, `n_(A)` and `n_(B)` moles of two SOLUTES in the equal volumes (V) of the solutions A and B at the same temperature, then `pi_(A) prop n_(A//V)` and `pi_(B)prop n_(B//V)` `:. (pi_(B))/(pi_(A)) = (n_(A))/(n_(B))` If `pi_(A) = pi_(B)`, then `n_A = n_B`. Thus, two solutions having equal osmotic pressure will contain equal number of moles and hence equal number of the molecules of the substances (solutes). This is similar to Avogadro'shypothesisfor gases. Hence, it is called van't Hoff-Avogadro's law for the solutions. |
|
| 47. |
State and explain van't Hoff-Avogadro's law. |
|
Answer» Solution :Statement of van't Hoff-Avogadro's law : At given temperature, equal volumes of the solutions having the same osmotic pressure contain equal number of solute particles. EXPLANATION: One mole of a substance contains Avogadro's number of MOLECULES (i.e., `6.02 xx 10^(23)` molecules). Therefore, the equal number of moles of the SUBSTANCES will contain equal number of molecules. The osmotic pressure, `pi` , is given by `pi` = CRT where C is the concentration of a solution in mol`dm^(-3)`. `pi - (n)/(v) RT` `therefore piprop (n)/(V)` at constant temperature. If two solutions contain `n_(A)` and `n_(B)` moles of two solutes in the equal volumes (V) of the solutions A and Bat the same temperature, then `pi_(A) prop n_(A//V)` and `pi_(B) prop n_(B//V)` `therefore (pi_(B))/(pi_(A)) =(n_(A))/(n_(B))` If `pi_(A) = pi_(B)` , then `n_(A) = n_(B)` |
|
| 48. |
State and explain Hess's law of constant heat summation. |
|
Answer» Solution :Statement of law of constant heat summation : It states that, the heat of a reaction or the enthalpy change in a chemical reaction depends upon initial STATE of reactants and final state of products and independentof the path by which the reaction is brought about (i.e, in single step or in series of steps). Explanation : Consider the conversion of the reatant A into the final product C. Let `Delta_(r)H^(@)` be the enthalpy change for the direct conversion. `A rarr CDelta_(r)H^(0)` The same conversion can be carried out in two steps as, Enthalpy change of Reaction `{:(ARARRB,Delta_(r)H_(1)^(0)),(BrarrC,Delta_(r)H_(2)^(0)):}` They byHess's law, `Delta_(r)H^(0) = Delta_(r)H_(1)^(0) + Delta_(r)H_(2)^(theta)`. Illustration : Consider the formationof `CO_(2^((g)))`. In one step : `C_((g)) + O_(2(g)) rarr CO_(2(g)) , DeltaH = - 394 kJ` In two steps : `C_((s)) + 1/2 O_(2(g)) rarr CO_((g)) , DeltaH_(1) =-83 kJ` `CO_((g)) + 1/2O_(2(g)) rarr CO_(2(g)) , DeltaH_(2) = - 311 kJ` `:. DeltaH =DeltaH_(1) + DeltaH_(2)` `- 394 kJ = - 83 kJ + (-311) kJ` Application : (1) To calculateheat of formation , COMBUSTION , neutralisation , etc. (2) To calculate heats of extremely slow reactions. |
|
| 49. |
State and explain Markonikoff's rule with suitable example . |
Answer» Solution :Markownikoff's rule : " When an unsymmetrical alkene is treated with an unsymmetrical REAGENT (HX), then the negative PART of the reagent (-X) gets added to that unsaturated carbon of the double bond, which contains lesser no. of hydrogen atoms . " <BR> Eg ., here , an unsymmetriacal reagent HBr is added to an unsymmetrical alkene like 2 - METHYL -2 - butene , the negative part `(Br^(-))` is attached to that carbon atom which have lesser no. of hydrogen atom and forms a ocompund 2 - bromo - 2 - methylbutane.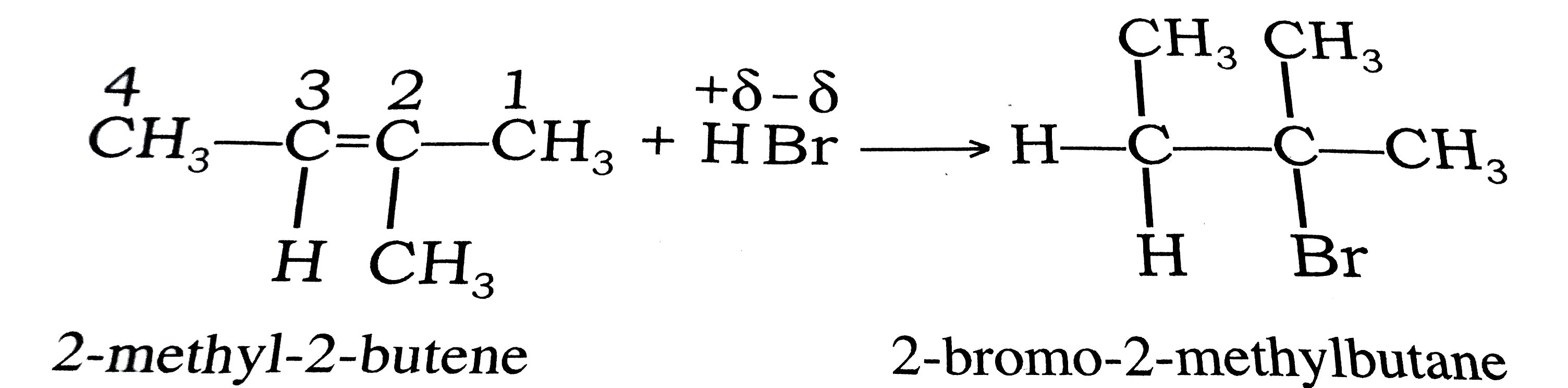 Here, Br add to `C_(2)` of 2 - methyl - 2 - butene which has lesser no. of hydrogen atoms . |
|
| 50. |
State and explain Kohlrausch's law. How does it help in the calculation of ^^_(m)^(@) for a weak electrolyte, CH_(3)COOH? |
|
Answer» Solution :Kohlrausch.s law. It states that at INFINITE dilution, the molar conductance of an electrolyte is equal to the sum of the molar conductances of its ions with molar conductance of each ion multiplied with the number of ions present in the formula of the electrolyte. e.g., `^^_(m)^(OO)(CH_(3)COOH)` `=lamda_(m)^(oo)(H^(+))+lamda_(m)^(oo)(CH_(3)COO^(-))` `^^_(m)^(oo)(NH_(4)OH)=lamda_(m)^(oo)(NH_(4)^(+))+lamda_(m)^(oo)(OH^(-))` Degree of dissociation of weak electrolyte, `alpha=(^^_(m)^(c))/(^^_(m)^(oo))` and `^^_(m)^(oo)(CH_(3)COOH)` `=^^_(m)^(oo)(CH_(3)COONa)+^^_(m)^(oo)(HCl)-^^_(m)^(oo)(NACL)` |
|