Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sodium carbonate solution in water is alkaline due to |
|
Answer» hydrolysis of `NA^(+)` `CO_(3)^(2-)+H_(2)O HARR HCO_(3)^(-)+""^(-)OH` |
|
| 2. |
Sodium carbonate solution in water is alkaline due to : |
|
Answer» HYDROLYSIS of `NA^+` |
|
| 3. |
Sodium carbonate reacts with SO_2 in aqueous solution to give: |
|
Answer» `NaHCO_3` |
|
| 4. |
Sodium carbonate on heating gives: |
|
Answer» WATER vapours |
|
| 5. |
Sodium carbonate is manufactured by solvay process. The products those are recycled are: |
|
Answer» `CO_2` and `NH_3` |
|
| 6. |
Sodium carbonate extract is acidified with HNO_(3) only in the identification of halides. Comment. |
| Answer» Solution :`HCl` and `H_(2)SO_(4)` are not used, as the acids react with the TEST reagent `AgNO_(3)` and FORM ppt. of `AgCl` and `Ag_(2)SO_(4)` respectively because of this only `HNO_(3)` is used for the purpose. | |
| 7. |
Sodium carbonate is: |
|
Answer» Efflorescent |
|
| 8. |
Sodium carbonate cannot be used in place of ammonium carbonate for the precipitation of the fifth group radicals because |
|
Answer» SODIUM ions interfere with the DETECTION of fifth group radicals |
|
| 9. |
Sodium carbonate can be manufactured by Solvay process but potassium carbonate cannot be prepared because |
|
Answer» `K_2CO_3` is more SOLUBLE than `Na_2CO_3` |
|
| 10. |
Sodium carbonate and mixture are taken in (a)___ ratio white perparingsodiumcarbonateextract |
|
Answer» |
|
| 11. |
Sodium cannot be extracted by the electrolysis of brine solution because |
|
Answer» electrolysis cannot take place with brine solution. |
|
| 12. |
Sodium connot be extracted by electrolysis of aqueous solutions due to |
|
Answer» Sodium is less REACTIVE |
|
| 14. |
Sodium borate on reaction with H_(2)SO_(4) (conc.) and C_(2)H_(5)OH gives a compound 'A' which burns with green flame. The compound 'A' is |
|
Answer» `H_(2)B_(4)O_(7)` |
|
| 15. |
Sodium borohydride reacts with iodine in the presence of diglyme to give A. A heated at 388 K give B. A heated at 373 K in sealed tube to form C. A further heated at red hot condition to give element D. Find out A,B, C and D. Give the reactions. |
|
Answer» Solution :(i) Sodium borohydride reacts with IODINE in the presence of DIGLYME to give diborane (A). `2NaBH_(4)+I_(2)overset("diglyme")(to)underset((A))(B_(2)H_(6))+2NaI+H_(2)` (ii) Diborane (A) heated at 388 K gives PENTABORANE (11)(B) `underset((A))underset(("Diborane"))(5B_(2)H_(6))underset((U-"tube"))overset(388K)(to)underset((B))underset(("pentaborane-11"))(2B_(5)H_(11))+4H_(2)` (iii) Diborane (A) heated at 373 K in the sealed tube to form decaborane (14)(C) `underset((A))underset(("Diborane"))(5B_(2)H_(6))underset("sealed tube")overset(373K)(to)underset((C))underset(("decaborane"-14))(B_(10)H_(14)+8H_(2)` (iv) Diborane (A) heated at red hot conditions to give element boron (D). `underset((A))underset(("Diborane"))(B_(2)H_(6))overset("Red hot")(to)underset((D))underset(("Boron"))(2B)+3H_(2)` 
|
|
| 16. |
Sodium bismuthate is |
|
Answer» a POWERFUL reducing-agent `2MN^(2+)+5NaBiO_(3)+14H^(+) to 2MnO_(4)^(-)+5Na^(-)+5Bi^(3+)+7H_(2)O` (a). It is a strong oxidizing agent as shown in (b) (c) It is a stoichiometric compound (d). `BiO_(3)+6NaOH to 2Na_(3)Bi_3+3H_(2)O` |
|
| 17. |
Sodium bicarbonate reacts with salicylic acid to form |
|
Answer» `C_(6)H_(5)O Na` |
|
| 18. |
Sodium bicarbonate solution on adding to magnesium sulphate solution forms: |
|
Answer» MAGNESIUM bicarbonate |
|
| 19. |
Sodium bicarbonate is manufactured by: |
|
Answer» CYANIDE process |
|
| 22. |
Sodium benzoate is used as ____. |
| Answer» SOLUTION :FOOD PRESERVATIVE. | |
| 23. |
Sodium benzoate is the most important food preservative. Suggest reason. |
| Answer» Solution :When sodium benzoate is used as FOOD preservative it is metabolised by the CONVERSION into hippuric ACID, which is FINALLY excreated in the URINE. | |
| 24. |
Sodium benzoate, salt of sorbic acid, acetic acid, sodium bi carbonate, sodium meta sulphite. |
| Answer» SOLUTION :Sodium bi CARBONATE. It is a baking SODA WHEREAS others are food preservatives. | |
| 25. |
Sodium benzoate, sodium propionate calcium propitiate, sodium metal bisulphite, sodium thiosulphate Among them how many chemicals are not food preservatives |
|
Answer» |
|
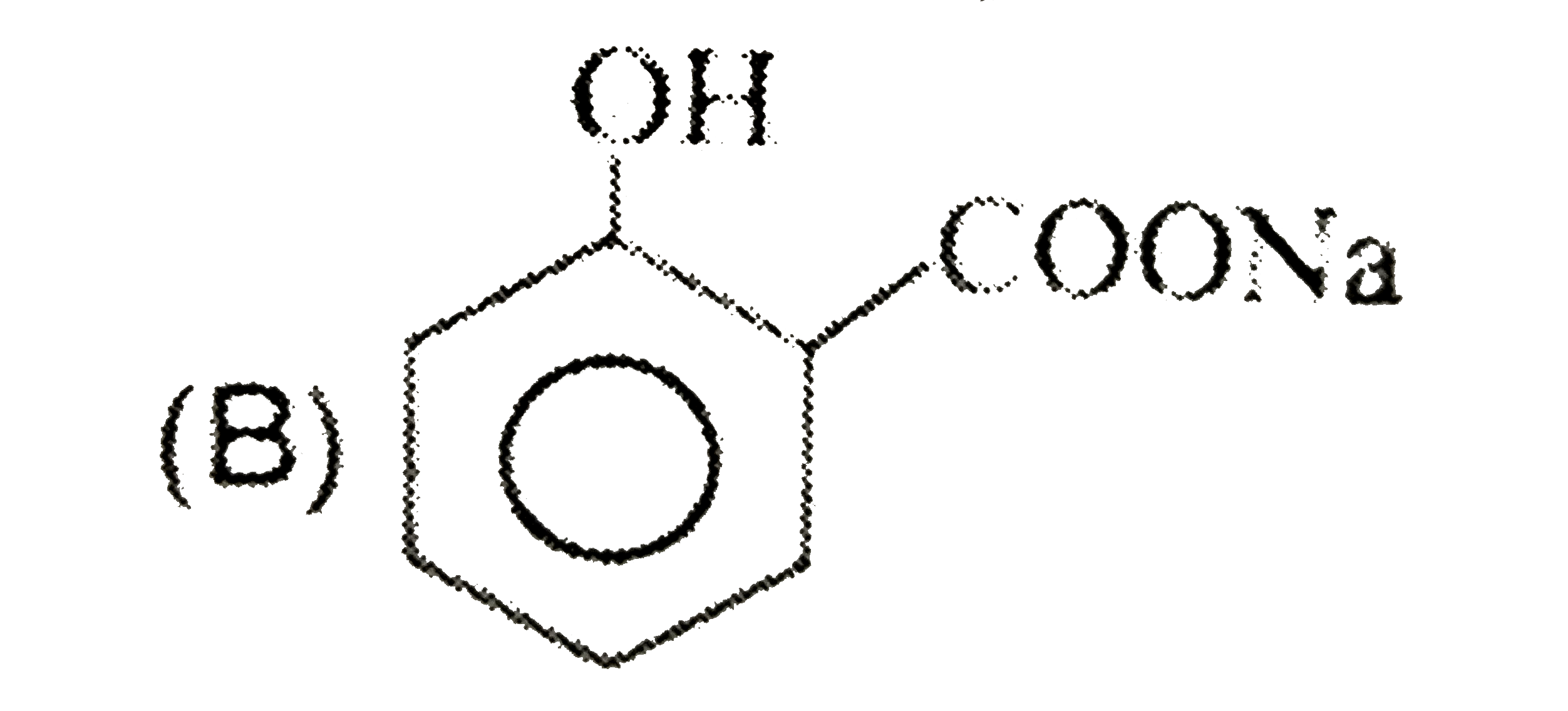
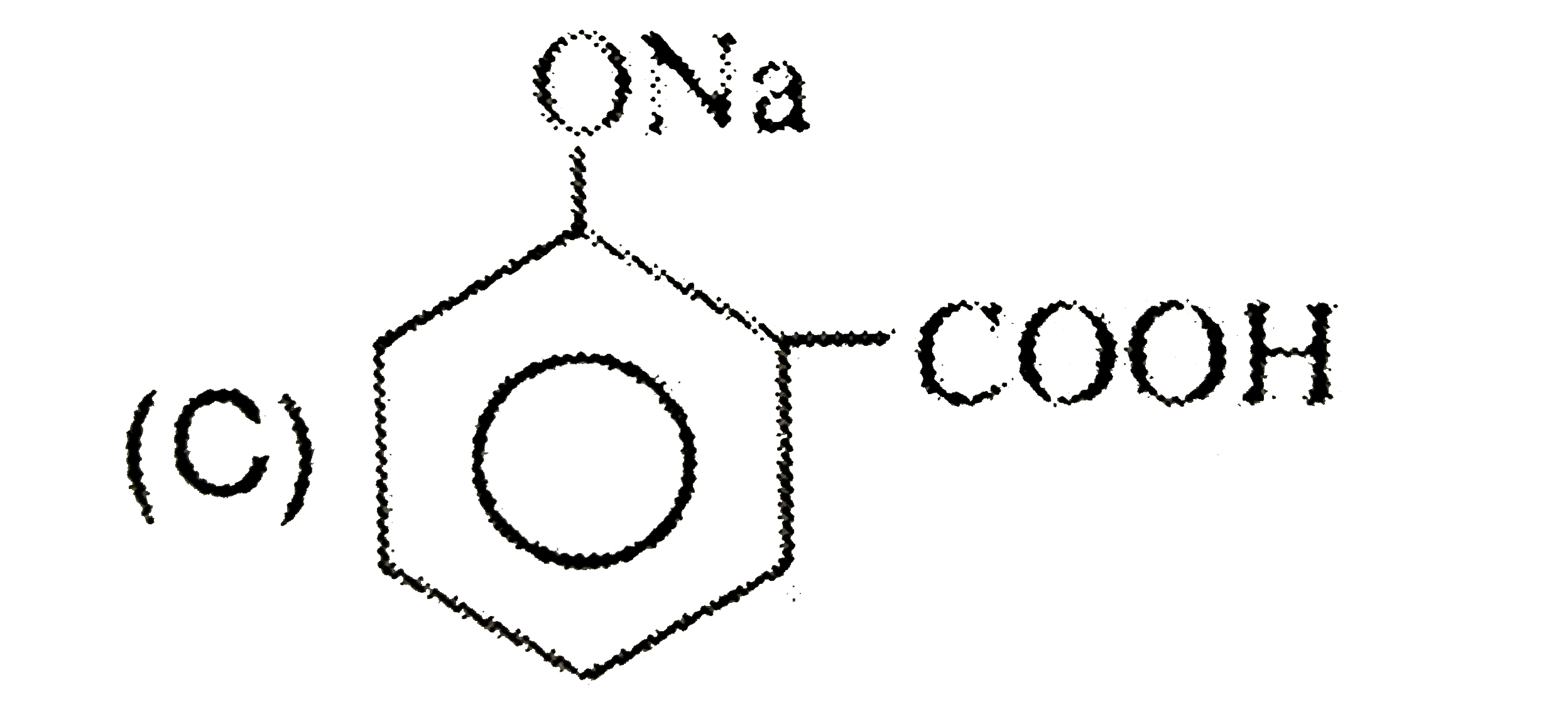
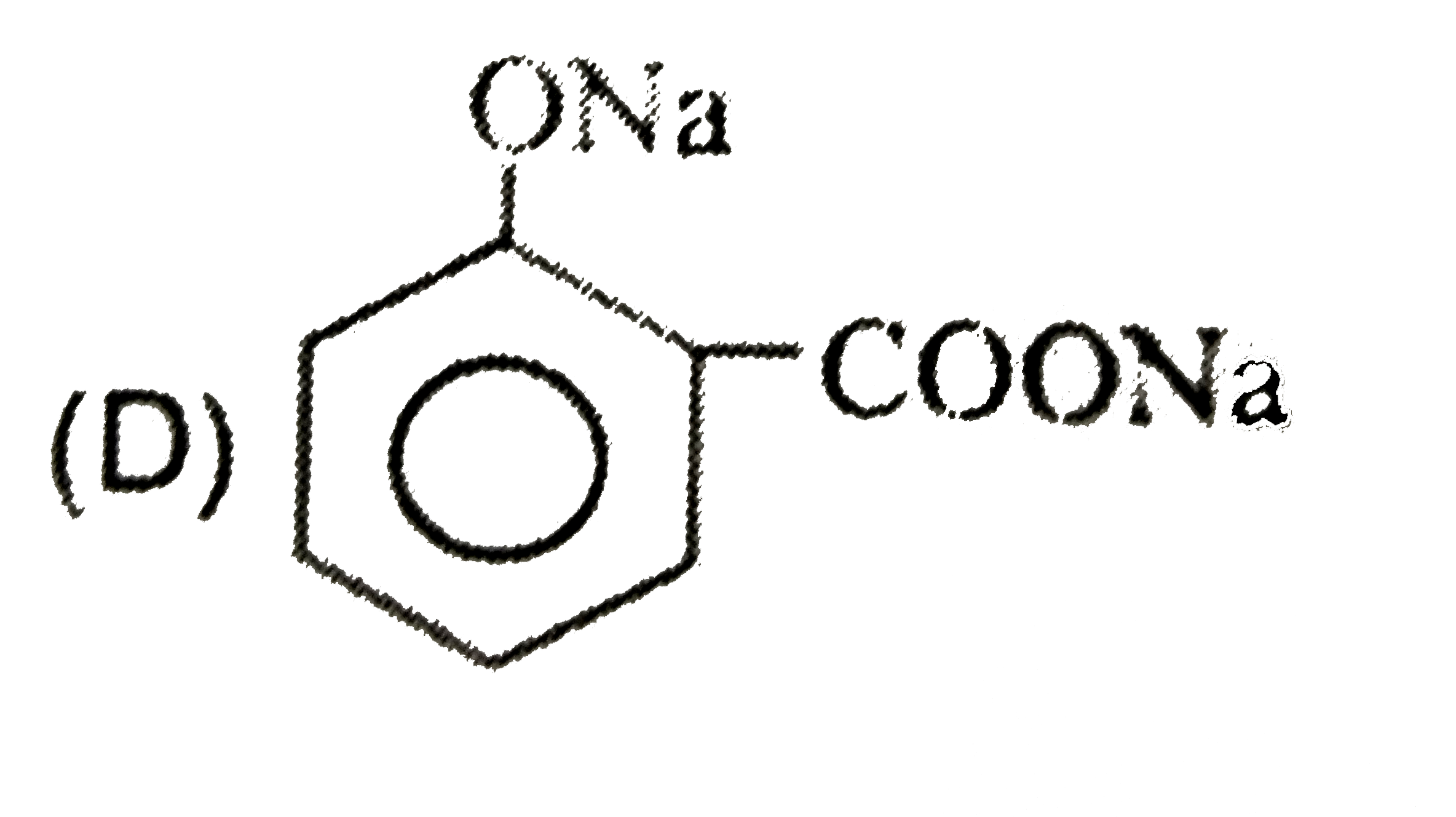
| 26. |
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis , it gives |
|
Answer» Phenol 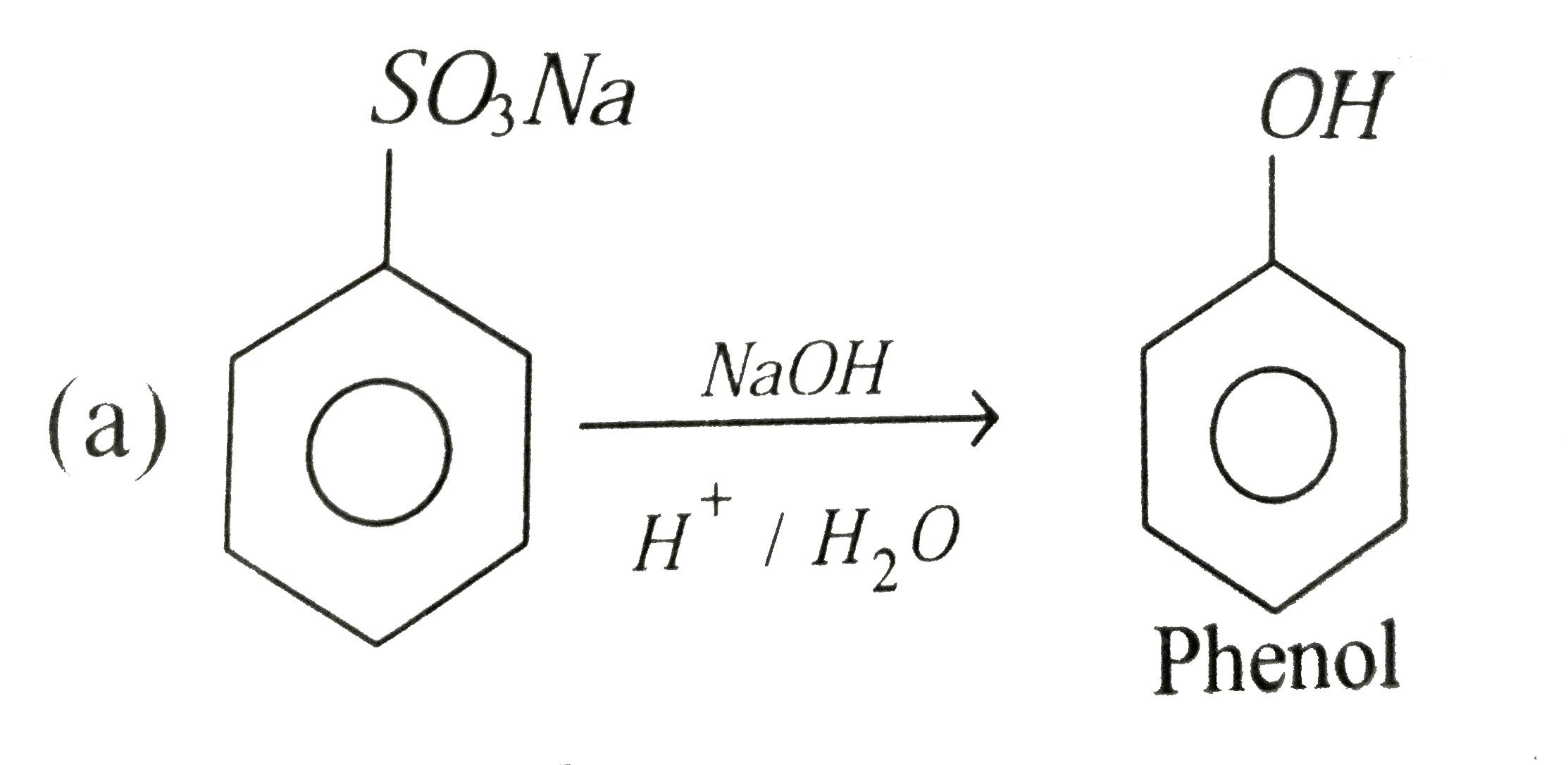
|
|
| 27. |
Sodium atom crystallizes in body centered cubic with a=4.29 overset(@)A. The radius of sodium atom is |
|
Answer» `18.6 OVERSET(@)A` |
|
| 28. |
Sodium atom reacts most readily with : |
|
Answer» `R-NH_2` |
|
| 29. |
Sodium and potassium occur: |
|
Answer» In NATIVE state |
|
| 30. |
Sodium alkyl benzene sulphonate is used as |
|
Answer» Pesticides |
|
| 31. |
Sodium acetate reacts with acetyl chloride to give |
|
Answer» acetic acid |
|
| 32. |
Sodium acetate reacts with Ethanoyl chloride to form: |
| Answer» Solution :Acetic anhydride | |
| 33. |
Sodium acetate and acetyl chloride react to give |
|
Answer» ACETIC ACID |
|
| 35. |
Sodalime test for detection of nitrogen cannot be used for : |
|
Answer» acetamide |
|
| 36. |
Sodalime test is used to detect .................element in an organic compound : |
|
Answer» C |
|
| 37. |
Sodalime is extensively used in decarboxylation reaction to obtain alkanes. Sodalime is- |
|
Answer» NaOH |
|
| 38. |
Soda water is of which type solution ? |
|
Answer» SOLID solution |
|
| 39. |
Soda water is an example for ........ |
|
Answer» gel |
|
| 42. |
Soda lime is made from: |
|
Answer» `Na+CaO+H_(2)O` |
|
| 43. |
Soda-lime is a specific reagent for |
|
Answer» DECARBOXYLATION |
|
| 44. |
Soda lime is used extensively in decarboxylation reaction to obtain alkanes. Soda lime is: |
|
Answer» NaOH |
|
| 46. |
Soda lime is |
|
Answer» NaOH |
|
| 47. |
Soda extractof a salt solution in acidified with excessof dil CH_(2)COOH and CaCI_(2) soinis added Awhite pptinsolable in CH_(3)COOH confurm of |
|
Answer» `C_(2)O_(4)^(2-)` |
|
| 50. |
Sod. Salt. (A)+ dil. HCl rarrSalt (B) +gas (X). (X) is colourless with suffocating odour and can turn lime water milky. (B)+K_(2)Cr_(2)O_(7)+ conc. H_(2)SO_(4)rarr coloured gas (Y). (A)+ Sulphur rarr Compound (C ). Which of the following is // are correct ? |
|
Answer» the OXIDATION no. of SULPHUR in (C) is `+2.5` |
|