Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sodium metal crystallises in a body centred cubic lattice with a unit cell edge of 4.29 A. The radius of Na atom is approximately |
|
Answer» `1.86 A` |
|
| 2. |
Sodium metal crystalizes in a body centred cubic lattice with the cell edge a =4.29overset@A.The radius of sodium atom is : |
|
Answer» `1.8574overset@A` |
|
| 4. |
Sodium metal cannot be stored under : |
|
Answer» benzene |
|
| 5. |
Sodium metal cannot be stored under: |
|
Answer» Benzene |
|
| 6. |
Sodium, Magnesium and Aluminium can be obtained from their are by |
|
Answer» Electrometallurgy |
|
| 8. |
Sodium iso-propoxide + A oversetDeltato 2-ethoxy propene + NaCl The compound A is |
|
Answer» `C_2H_5ONa` |
|
| 9. |
Sodium is used in nuclear reactor as : |
|
Answer» a moderator |
|
| 10. |
Sodium hypophosphite represented as |
|
Answer» `Na_(3)PO_(4)` |
|
| 11. |
Sodium iauryl sulphate is |
|
Answer» Bio-DEGRADABLE, CATIONIC dertergent |
|
| 12. |
Sodium hydroxide is produced on a large scale |
|
Answer» by the hydrolysis of `Na_(2)CO_(3)` |
|
| 13. |
Sodium hydroxide may be prepared by any of the following methods except : |
|
Answer» NELSON cell |
|
| 14. |
Sodium ordinarily does not show an oxidation state of +2, because of its |
|
Answer» HIGH FIRST ionisation potential |
|
| 15. |
Sodium forms two oxides (A) and (B) having 25.81 % and 41.03% oxygen respectively. What is the reaction of (A) and (8) with H_(2)O: |
|
Answer» SOLUTION :`AtoNa_(2)O`,`Na_(2)O+H_(2)Oto2NaOH` `BtoNa_(2)O_(2)+2H_(2)Oto2NaOH+H_(2)O_(3)` |
|
| 16. |
Sodium formate solution on electrolysis gives ................. at anode. |
|
Answer» METHANE + `CO_2` |
|
| 17. |
Sodium formate on heating with soda lime give: |
|
Answer» `CH_4` |
|
| 18. |
Sodium extract of Lassaigne's solution is treated with FeSO_4 , FeCl_3 and dil. H_2 SO_4 to get a blood red colour . Which of the following is probable organic compound ? |
|
Answer»
|
|
| 19. |
Sodium extract prepared by using thiourea contains which ion in the solution ,mainly responsible for a characteristic test: |
| Answer» ANSWER :C | |
| 20. |
Sodium extract of Lassaigne's solution is treatedwith FeSO_4, FeCl_3 and dil. H_2SO_4 to get a blood red colour. Which of the following is probable organic compound? |
|
Answer»
|
|
| 21. |
Sodium extract is heated with concentrated HNO_(3) before testing for halogens because : |
|
Answer» Ag reacts faster with halides in ACIDIC medium. |
|
| 22. |
Sodium ethoxide reacts with ethyl iodide to yield: |
|
Answer» `CH_3CH_3` |
|
| 23. |
Sodium ethoxide reacts with ethanoyl. The compound that is produced in the above reaction is |
|
Answer» 2-butanone |
|
| 24. |
Sodium ethoxide is specific reagent for: |
|
Answer» Dehydration |
|
| 25. |
Sodium ethoxide is obtained by the reaction of ethyl alcohol with : |
|
Answer» NaOH |
|
| 26. |
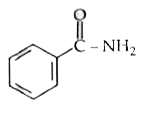
Sodium ethoxide has reacted with ethanoyl chloride . The compound that is produced in the above reaction is |
|
Answer» DIETHYL ether 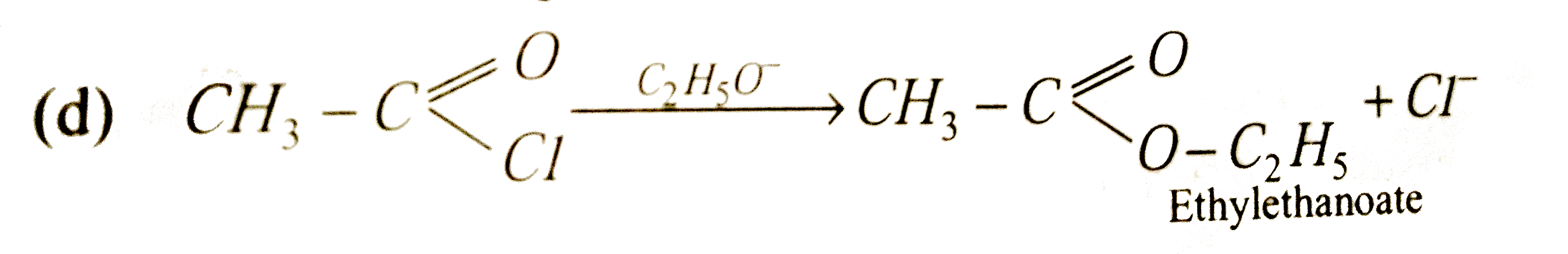
|
|
| 27. |
Sodium ethoxide (C_(2)H_(5)ONa) is a specific reagent for |
|
Answer» Dehydration |
|
| 28. |
Sodium ethoxide is a specific reagent for |
|
Answer» dehydration  ETHOXIDE ion is a strong base which ABSTRACTS proton from CARBOCATION to form alkene.  Thus, there is a loss of hydrogen and halogen both leading to dehydrohalogenation. |
|
| 29. |
Sodium ethoxide and ethyl chloride on heating will give : |
| Answer» Answer :A | |
| 30. |
Sodiumcyanideis sometimesaddedinthefrothfloatationprocess asa depressantwhen the ore contains amixtureofZnS +PbS because |
|
Answer» Pb `(CN)_2 `getsprecipitatedwithoutanyeffecton ZNS |
|
| 31. |
Sodium dodecyl benzene sulphonate is used as a: |
|
Answer» Pesticide |
|
| 33. |
Sodium cyanide solution is used to extract _______ from its ores. |
|
Answer» copper |
|
| 34. |
Sodium crystallizes in a face centred cubic lattice. The approximate number of unit cells in 5.0 g of sodium is (Atomic mass of sodium = 23 amu) |
|
Answer» `32.7 × 10^(22)` |
|
| 35. |
Sodium cyanide is added to depresses the floatation property of ZnS by forming a layer of ……. |
| Answer» Solution :`Na_(2)[Zn(CN)_(4)]`-SODIUM ZINCATE complex | |
| 36. |
Sodium crystallizes in the cubic lattice and the edge of the unit cell is 430 pm. Calculate the number of atoms in a unit cell. (Atomic mass of Na = 23.0 density = 0.9623g cm^(-3), N_A = 6.023 xx 10^(23) mol^(-1)). |
|
Answer» Solution :Length of edge = 430 pm = `430 xx 10^(-12) m=430 xx 10^(-10)` cm Volume of unit CELL = `a^3 =(430 xx 10^(-10))^3 cm^3 = 79.5 xx 10^(-24) cm^3` Mass of one atom= `("Atomic mass")/("Avogadro.s No.") = (23)/(6.023xx10^(23))=3.82 xx 10^(-23)` g Mass of unit cell = `"Volume" xx "density" = 79.5 xx 10^(-24) xx 0.9623 = 76.5 xx 10^(-24)` g No. of atoms in unit cell = `("Mass of unit cell")/("Mass of an atom") =(76.5 xx 10^(-24))/(3.82 xx 10^(-23)) = 2` |
|
| 37. |
Sodium crystallises in a bcc unit cell. Calculate the approximate number of unit cells in 9.2 g of sodium ? "[Atomic Mass of Na = 23 u]" |
|
Answer» Solution :No. of moles of sodium `=("Mass of sodium")/("Atomic mass of sodium")` `=(9.2)/(23)=0.4` ONE MOLE of sodium metal contains `=6.022xx10^(23)" atoms"` 0.4 mole of sodium metal contains `=0.4xx6.022xx10^(23)" atoms "=2.4088xx10^(23)" atoms"` One bcc UNIT cell contains = 2 atoms No. of unit cells in 9.2 g of sodium `=(2.4088xx10^(23))/(2)=1.2044xx10^(23)` |
|
| 38. |
Sodium crystallizes in a bcc lattice, hence the coordination number of sodium in sodium metal is |
|
Answer» 0 |
|
| 39. |
Sodium chloride when heated with conc. H_(2)SO_(4) and solid potassium dichromate gives |
|
Answer» Chromic chloride |
|
| 40. |
Sodium chloride, NaCl usually crystallizes in a face centred cubic lattice. How many ions are in contact with any single Na^(+) ion ? |
|
Answer» 4 |
|
| 42. |
Sodium chloride is soluble in water not in benzene because |
|
Answer» `Delta H_("hydration") lt Delta H_("LATTICE energy in water") and Delta H_("solvation") GT Delta H_("lattice energy in BENZENE")` |
|
| 43. |
Sodium chloride is soluble in water but not in benzene because |
|
Answer» `DeltaH_("hydration")ltDeltaH_("lattice ENERGY in water")and` |
|
| 44. |
Sodium chloride is prepared in the lab by burning sodium metal in the atmosphere of chlorine. The staff produced has a slightly yellowish colour. The cause of yellow colour is due to |
|
Answer» presence of `NA^(+)` ions in the CRYSTAL LATTICE. |
|
| 45. |
Sodium chloride is known as: |
|
Answer» ROCK salt |
|
| 46. |
Sodium chloride is an ionic compound whereas hydrogen chloride is a gas because |
|
Answer» SODIUM is reactive |
|
| 47. |
Sodium chloride is an ionic compound where as hydrogen chloride gas is mainly convalent because |
|
Answer» A) electronegativity difference in the CASE of hydrogen is less 2.1 |
|
| 48. |
Sodium chloride crystallizes in face-centred cubic (f.c.c.) structure. Its density is 2.165 g cm^(-3). If the distance between Na^+ and its nearest Cl^(-) ions is 281 pm, find out the Avogadro's number (Na = 23 g "mol"^(-), Cl = 35.5 g "mol"^(-)). |
|
Answer» Solution :For a cubic unit CELL, `d=(ZM)/(a^3 10^(-30)N_A) G cm^(-3)` or `N_A=(ZM)/(10^(-30)d a^3)` When .a. is in pm. Where M=Molar MASS of NACL = 23 + 35.5 = 58.5 g `mol^(-)` `N_A`=Avogadro.s number = ? d= density = 2.165 g `cm^(-3)` Z= number of atoms per unit cell = 4 (f.c.c.) a=edge length of the unit cell in pm. For NaCl type crystal, edge length (a) = `2 xx "interionic distance"` = 2 (281) = 562 pm `:. N_A=(4 xx 58.5 g mol^(-))/(10^(-30) xx 2.165 g cm^(-3) xx (562)^3 cm^3)` `=(234.0 g mol^(-1))/(8 xx 22188041 xx 10^(-30) xx 2.165 g) = 6.09 xx 10^(23) mol^(-)` |
|
| 49. |
Sodium chloride contains 60.68% chlorine by mass. If 7.1 g chlorine is made to react with 5.2 g of sodium, the number of moles of sodium which reacted with Cl_(2) is |
|
Answer» `0.2` 60.68 CHLORINE react with sodium = 39.32 g 7.1 g of chlorine react with sodium `= ((39.32g)XX(7.1g))/(60.68)` `= 4.60 g` Moles of sodium ` = (4.60g)/(23 g MOL^(-1)) = 0.2 mol` |
|