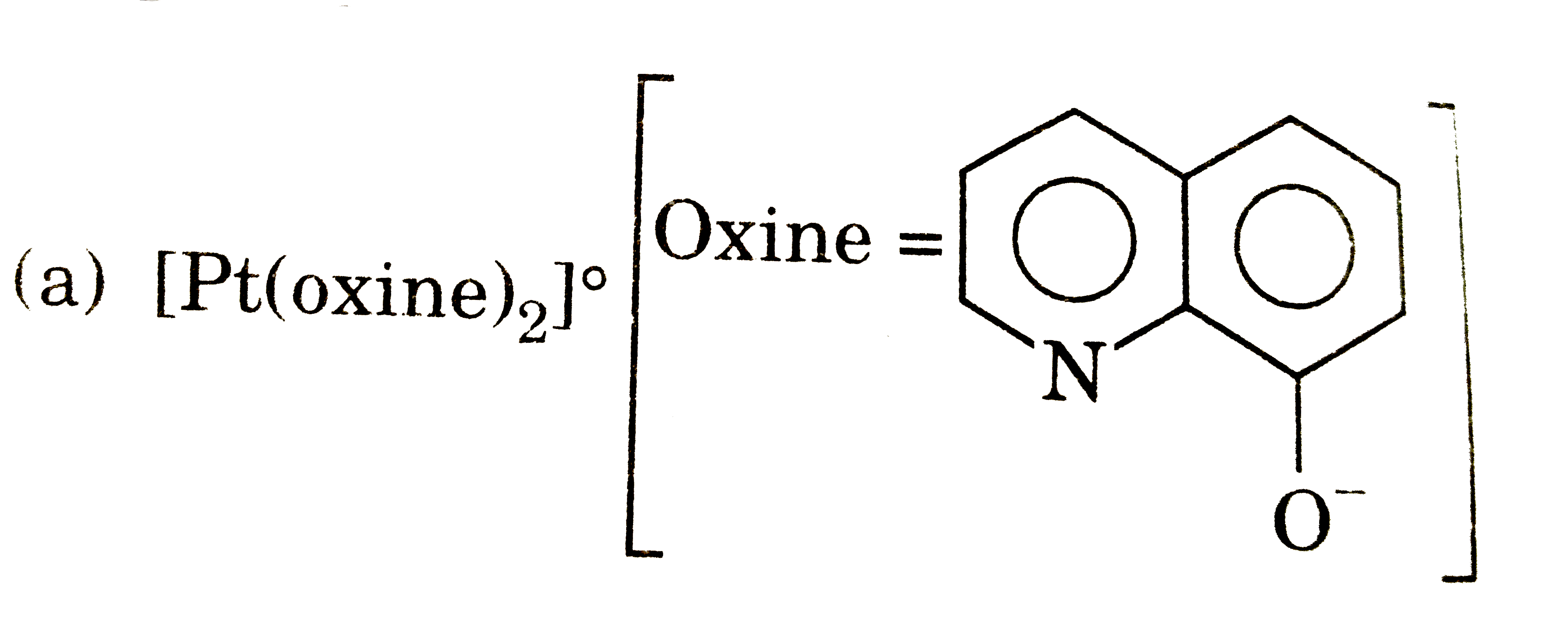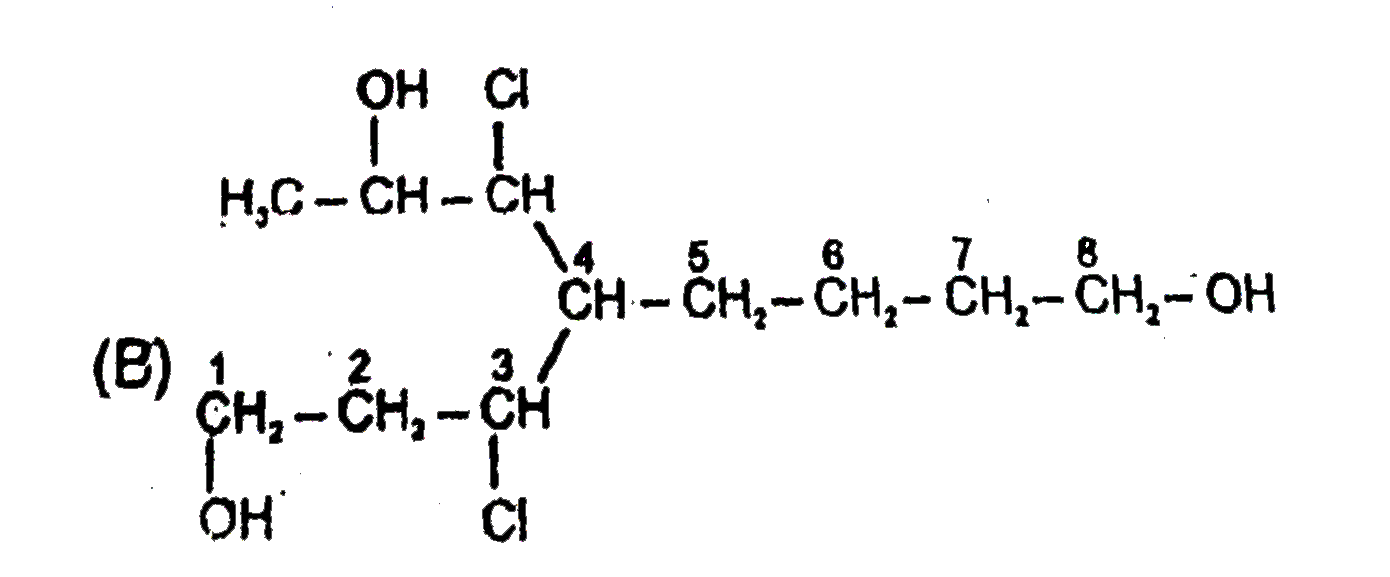Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
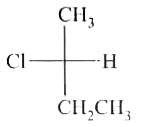
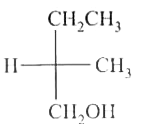
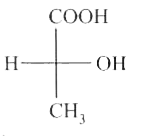
select write statement (s) (A) 8 Cs+ ions occupy the second nearest neighbour locations of a Cs+ ion (B) Each sphere is surrounded by six voids in two dimensional hexagonal close packed layer (C) If the radius of cations and amions are 0.3 Å and 0.4 Å then coordinate number of cation of the crystal is 6. (D) In AgCl, the silver ion is displaced from its lattice position to an interstitial position such a defect is called a frenkel defect. |
|
Answer» Solution :(A) 6 CS+ ION second nearest neighbour (B)  `(C)` `(r_(+))/(r_(1))`=0.75 (BCC) 8: Coordination no. (D) True |
|
| 2. |
Select write statement (s) |
|
Answer» `8 Cs^(+)` ions OCCUPY the second nearest neighbour locations of a `Cs^(+)` ION |
|
| 3. |
Select which type of overlapping is responsible for pi-character in Si-N bond N_(3)SiNCO |
|
Answer» `3p PI to 2P pi` |
|
| 4. |
Select which is/are isomer of each other : |
|
Answer» `[Pt(NH_(3))_(4)(NO_(2))_(2)]SO_(4)` |
|
| 5. |
Select which is heteroleptic complex and show fac-mer isomerism : |
|
Answer» `PtCl_(2).2NH_(3)` It is heteroleptic and show fac-mer isomerism. |
|
| 6. |
Select which complex have structural and stereo both type of isomers : |
|
Answer»
|
|
| 7. |
Select which has maximum formal charge on central atom : |
| Answer» Answer :D | |
| 8. |
Select which complex formation reaction is/are incorrect for forward displacement: |
|
Answer» `Ag_(2)S+8CN^(-) to [Ag(CN)_(4)]^(-)+S^(2)` (d) [Cu(CN)_(4)]^(-) "is formed " |
|
| 9. |
Select true statement (S) : |
|
Answer» `I_(2)` does not react with ethane at room temperature even though `I_(2)` is more easily cleaved homolytically than the other halogens. |
|
| 10. |
Select true statement : |
|
Answer» Both B and C GIVE same name REACTION with KOH. 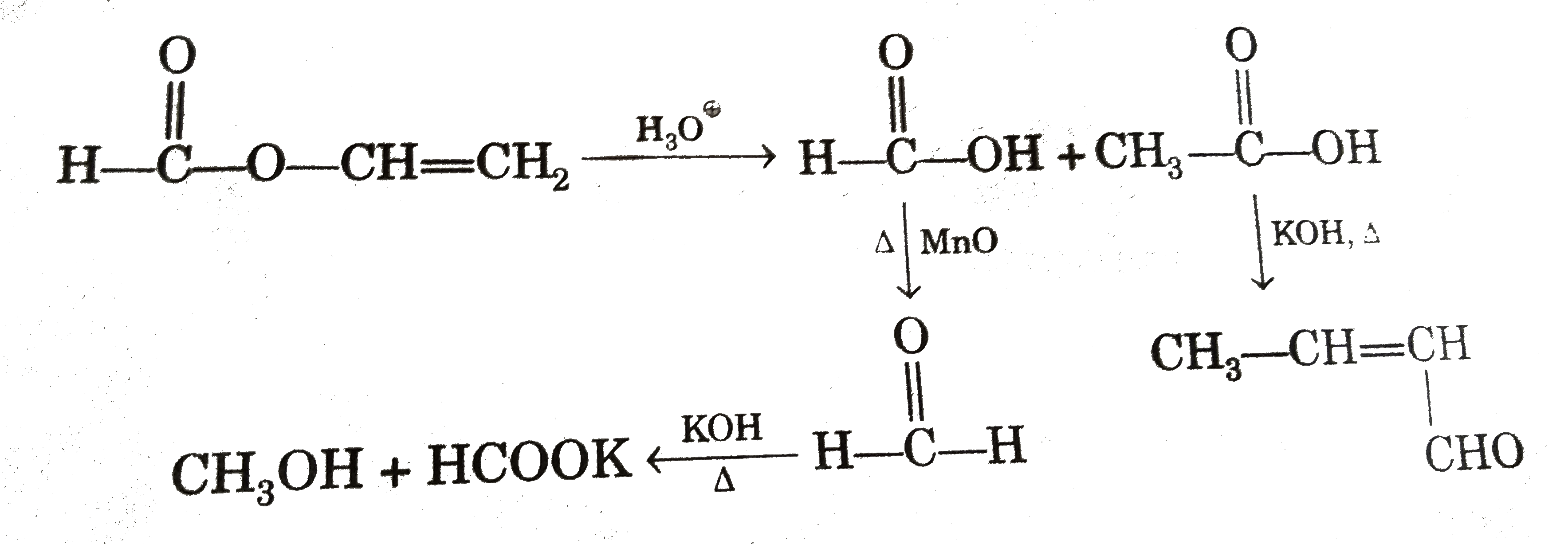
|
|
| 11. |
Select total true statements (1)conc. H_2SO_4 in used to acidify KMnO_4 (2)In aq. medium Fe^(3+ )is more stable than Fe^(2+) (3)conc.HNO_3is usedto acidifyKMnO_ 4(4) commonproductof heatinginKMn O_ 4&K_ 2Cr _ 2O _7isO_ 2 (g )(5 )Ce O_ 2isgoodanalyticalagent(6)Sc does not showvariablevalency Options are |
|
Answer» 1 |
|
| 12. |
Select triangular planar species among the following |
|
Answer» `ClO_(3)^(-)` |
|
| 13. |
Select to correct statements. |
|
Answer» Catalyst can CHANGE the spontaneity of reaction |
|
| 15. |
Select the wrong statement(s): |
|
Answer» `CaF_(2)` is SOLUBLE in WATER |
|
| 16. |
Select the wrong statements |
|
Answer» Hg from an amalgam with iron |
|
| 17. |
Select the wrong statement(s) : |
|
Answer» Iron does not fonn AMALGAM with MERCURY |
|
| 18. |
Select the wrong statement (S): |
|
Answer» Nuclear isomers contain the same number of protons and neutrons |
|
| 19. |
Select the wrong statement among the following |
|
Answer» Antieutrino can be detected during `BETA`-emission |
|
| 20. |
Select the wrong statement |
|
Answer» Ozone is a pale blue gas |
|
| 21. |
Select the wrong statement. |
|
Answer» The C.N. of cation occupying a tetrahedral HOLE is 4. Since octahedral hole is surrounded by six nearest neighbours so C.N. of cation occupying octahedral hole is 6. In Schottky defect a pair of anion and cation leaves the lattice, so density to lattice decreases. HOWEVER in case of Frenkel defect the density remains unchanged as the ions are PRESENT in the INTERSTITIAL sites without changing the volume. |
|
| 22. |
Select the wrong statement : |
|
Answer» If a very SMALL amount of `AlCl_(3)` is added to gold sol, coagulation occurs but if a large quantity of `AlCl_(3)` is added, there is no coagulation. |
|
| 23. |
Select the wrong chemical reaction among the following: |
|
Answer» `MnO_2 + 4HCl rarr MnCl_2 + Cl_2 + 2H_2O` |
|
| 24. |
Select the weakest Bronstad base from the following bases |
|
Answer»
 is the weakest BASE due to delocalisation of a LONE pair of electrons present on the N-atom into the BENZENE ring. is the weakest BASE due to delocalisation of a LONE pair of electrons present on the N-atom into the BENZENE ring.
|
|
| 25. |
Select the true statement from the following for metal carbonyls ? |
|
Answer» `pi` back bonding strengthens M - C bond order as well as CO bond order . |
|
| 26. |
Select the true statement(s) among the following : |
|
Answer» Pure overlapping of two `d_(xy)` orbitals along x-axis results in the FORMATION of `pi`-bond 
|
|
| 27. |
Select the suitable choice by comparing Column-I with II. {:("Column-I","Column-II"),("(X) Cryolite", (M) Na[Al(OH_4)]),("(Y) Sodium-Aluminate", (N)[Al_2(OH)_(4),Si_2O_3]),("(Z) Kaolinite", (P) Na[Al_2(OH)_(3)Si_2O_3]),(,(Q) Na[Al_2(OH)_(3)Si_(2)O_3]),(,(R ) Na_3[AlF_4]):} |
|
Answer» `X to R, Y to P , Z to Q` |
|
| 28. |
Select the total number of properties which are correct with respect to K_(4)[Fe(CN)_(5)O_(2)] or any of its isomers (d^(6) configuration of central metal ion). (a) Paramagnetic (b) Inner orbital complex (c ) Low spin complex (d)Delta_(0) gt P (e ) Shows optical isomerism (f) Shows linkage isomerism |
|
Answer» Also, CONSIDER linkage isomers for (e ). |
|
| 29. |
Select the true statement |
|
Answer» Both B and C give same name eraction with KOH 
|
|
| 30. |
Select the most ionic and most covalent compounds respectively from the following. CrO_5, Mn_2O_7, PbO, P_4O_10,SnO_2 |
|
Answer» `CrO_(5), Mn_(2)O_(7)` |
|
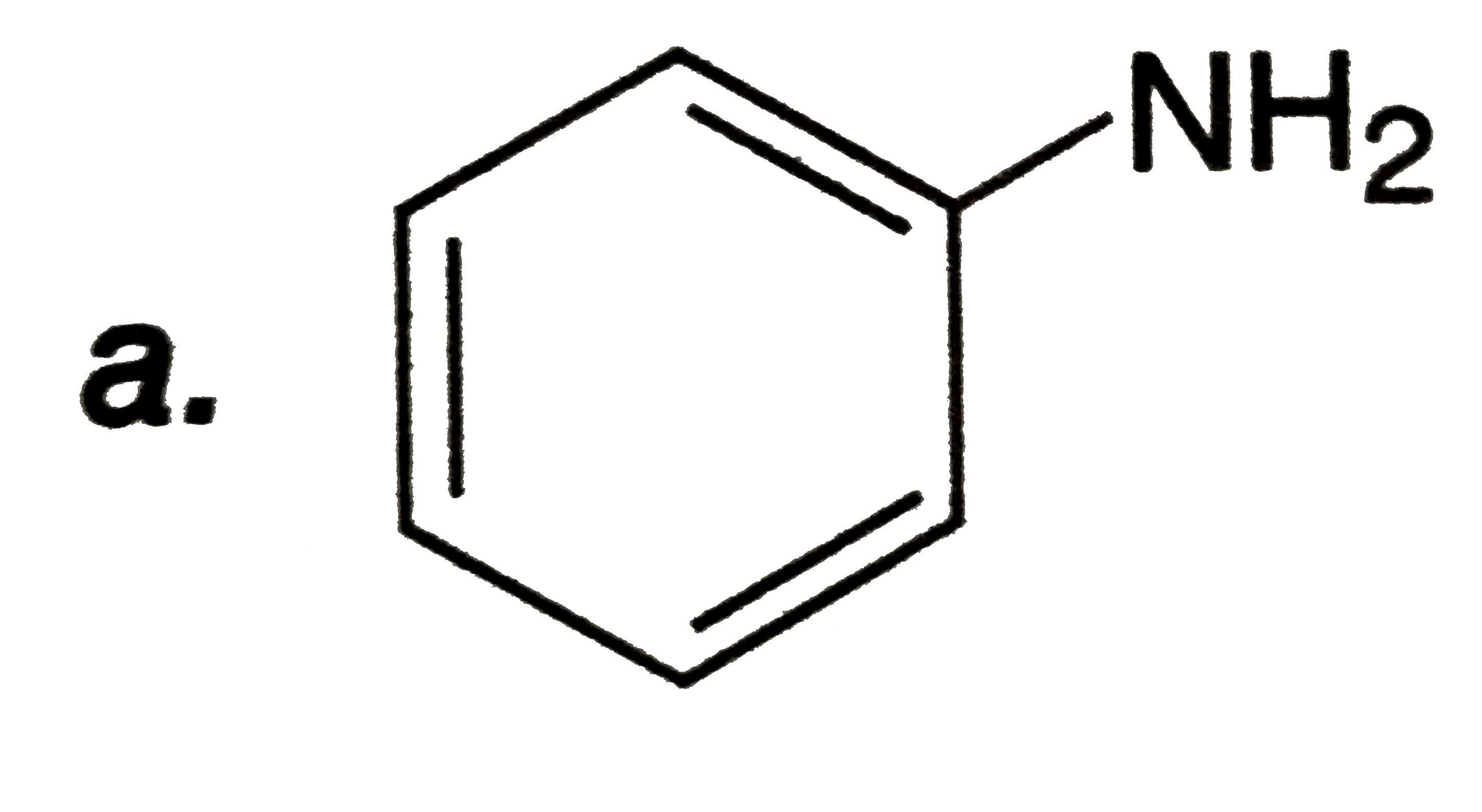
| 31. |
Select the true statement about benzene amongst the following |
|
Answer» Because of UNSATURATION BENZENE easily UNDERGOES addition reactions |
|
| 33. |
Select the strongest reducing agent from amongest the substances involved in the following half-reactions . {:(Cl_(2) (g) + 2e^(-) to 2Cl^(-) (aq) , + 1.36 V) , (Br_(2) (g) + 2e^(-) to 2 Br^(-) (aq) , +1.09V) , (Sn^(2+) + 2e^(-) to Sn , -0.14V):} |
|
Answer» `CL^(-)` |
|
| 34. |
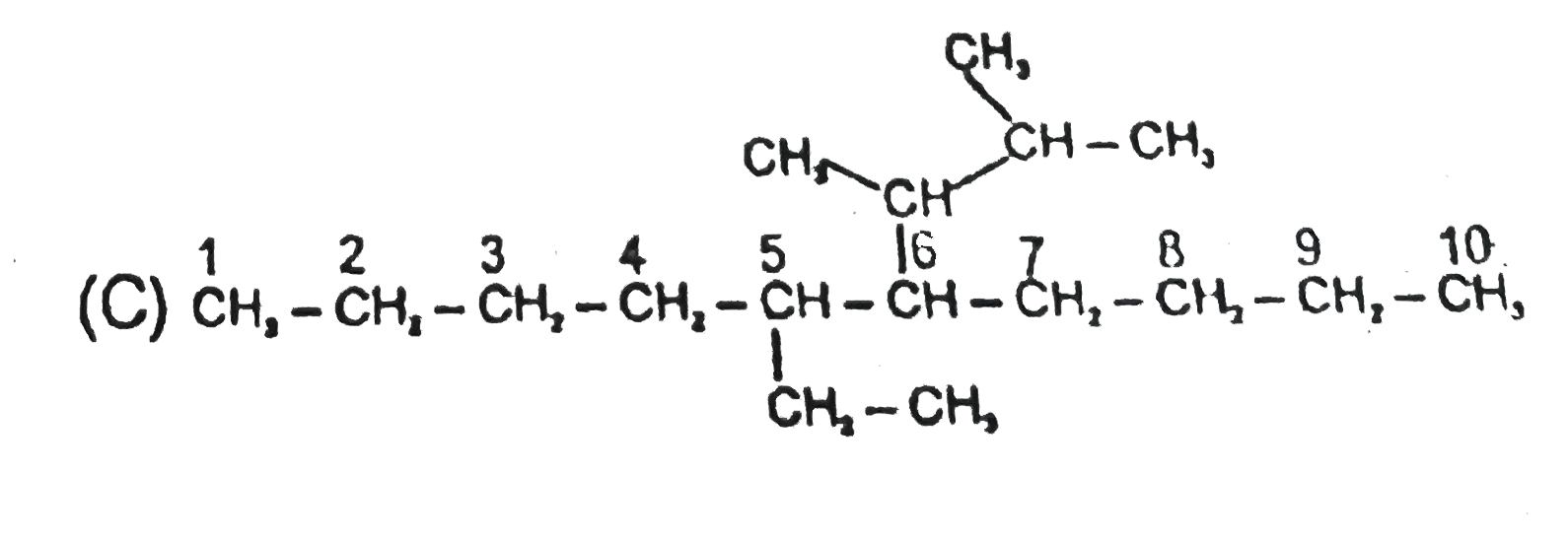
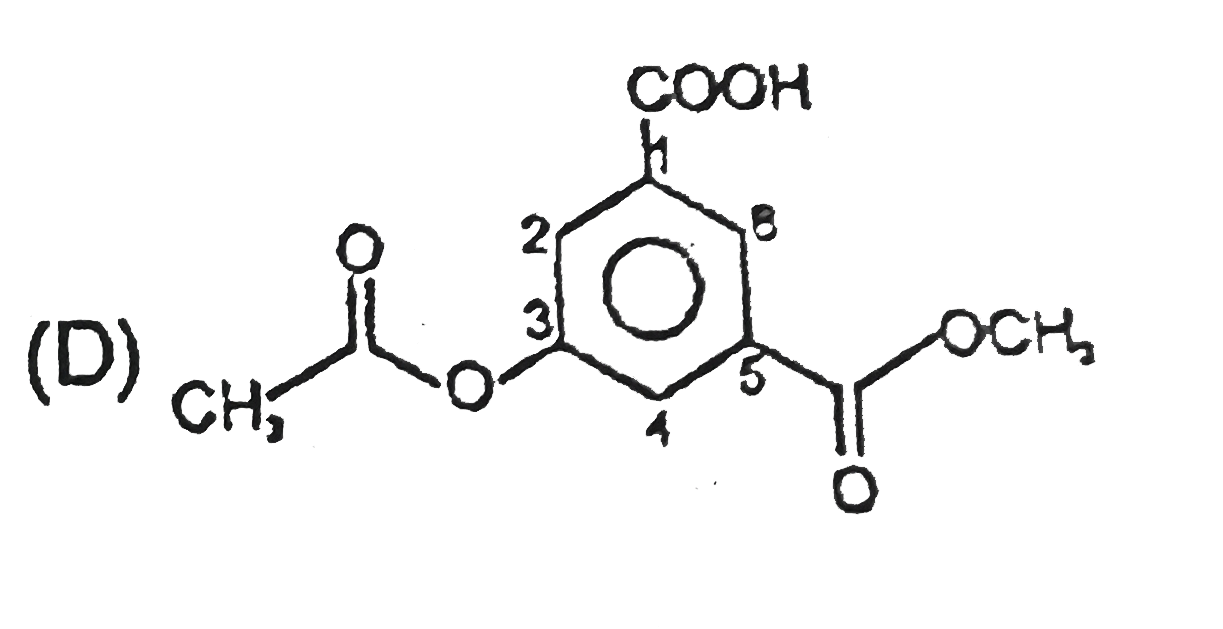
Select the structure with correct numbering for IUPAC name of the compound : |
|
Answer»
(B)Functional groups should have lowest SET of locant (C )Name of complex SIDE chain is begin from first alphabet of its complete name. (D)Ethanoyloxy COMES first than methoxy carbonyl. |
|
| 35. |
Select the state functions among the following. (i) U + PV(ii) S (iii) q(iv) w H - TS |
|
Answer» (III), (IV) and (V) only |
|
| 36. |
Select the strongest base in following compounds: . |
|
Answer» |
|
| 38. |
Select the starting substance and reagent for synthesis of following amines: |
Answer» SOLUTION :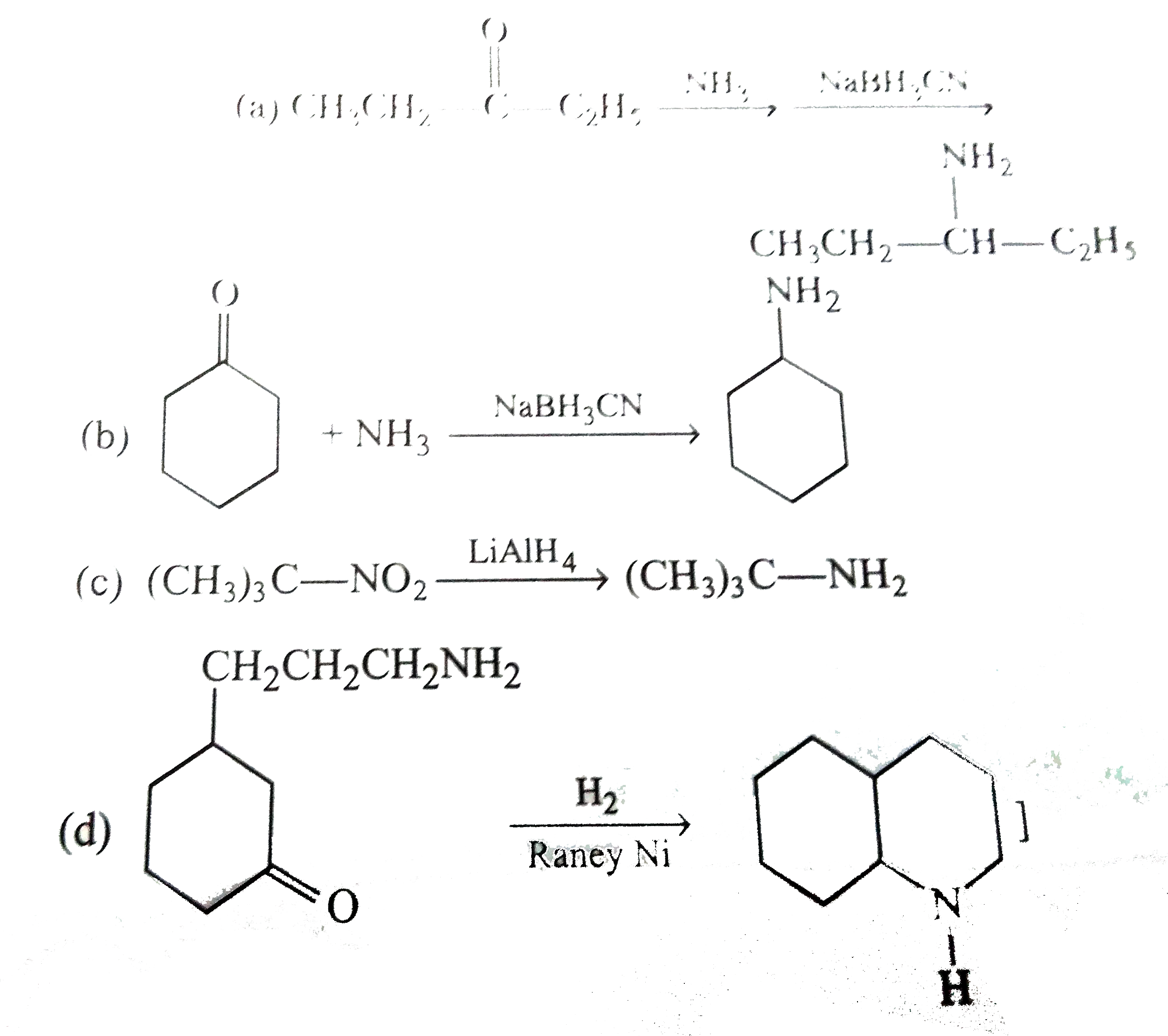
|
|
| 39. |
Select the same molecuels among the following: |
|
Answer»
|
|
| 40. |
Select the reagent for above conversion. |
| Answer» Solution :(a)Carbene formation | |
| 41. |
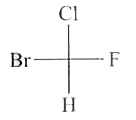
Select the S-isomer from the following: |
|
Answer»
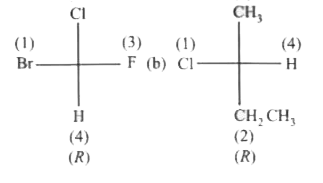 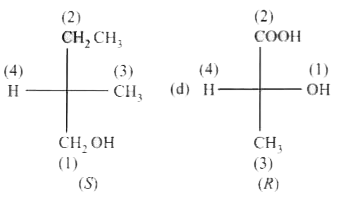
|
|
| 42. |
Select the reactions that would result in the formation of 2-bromopropane. |
|
Answer» `CH_3 CH= CH_2 + Br_2 overset(HV)(to)` |
|
| 43. |
Select the reaction for which the equilibrium constant is written as [MX_(3)]^(2)=K[MX_(2)]^(2)[X_(2)] |
|
Answer» `MX_(3)iffMX_(2)+1//2X_(2)` |
|
| 44. |
Select the proper choice by true statement by symbol "T" and false statement by symbol "F". (i) Perchloric acid is weaker than chloric acid. (ii) HF is stronger acid than HCl. (iii) NH_3 is weaker base than PH_3. (iv) All noble gases exist as monoatomic. |
|
Answer» FTFT |
|
| 45. |
Select the product with minimum yeild obtained form the following reaction CH_(3)CHBrCH_(2)+KOH overset(alc) to |
|
Answer» 2-BUTENE |
|
| 46. |
Select the principal functional group when the following groups are present in the molecule: (a) -OH,-COOH,-NO_(2) (b) -NO_(2),-NH_(2),-CONH_(2) (C )-Br,-CH_(3),-CHO (d) Cl,-CH_(3),-OH. |
|
Answer» (C) ALDEHYDE,-CHO (d) Alcoholic, -OH. |
|
| 47. |
Select the process having exothermic process :- |
|
Answer» `S_((g))^(-)toS_((g))^(-2)` |
|
| 48. |
Select the pK_(a) value of the strongest acid from the following |
|
Answer» 1 Strong ACIDS have LOWER `pK_(a)` values. |
|
| 49. |
Select the pair of hydrate isomers in which both hydrate isomers have twostereoisomeric form : |
|
Answer» `[Co(NH_(3))_(5)(H_(2)O)](NO_(3))_(3)" and "[Co(NH_(3))_(5)(NO_(3)](NO_(3))_(2).H_(2)O`<BR>`[Pt(H_(2)O)_(4)Cl_(2)]Br_(2)" and "[Pt(H_(2)O)_(4)Br_(2)]Cl_(2)` |
|
| 50. |
Select the pair of almost same size. |
|
Answer» AL,Ca `Al,Gararr` due to o-orbital contraction. |
|