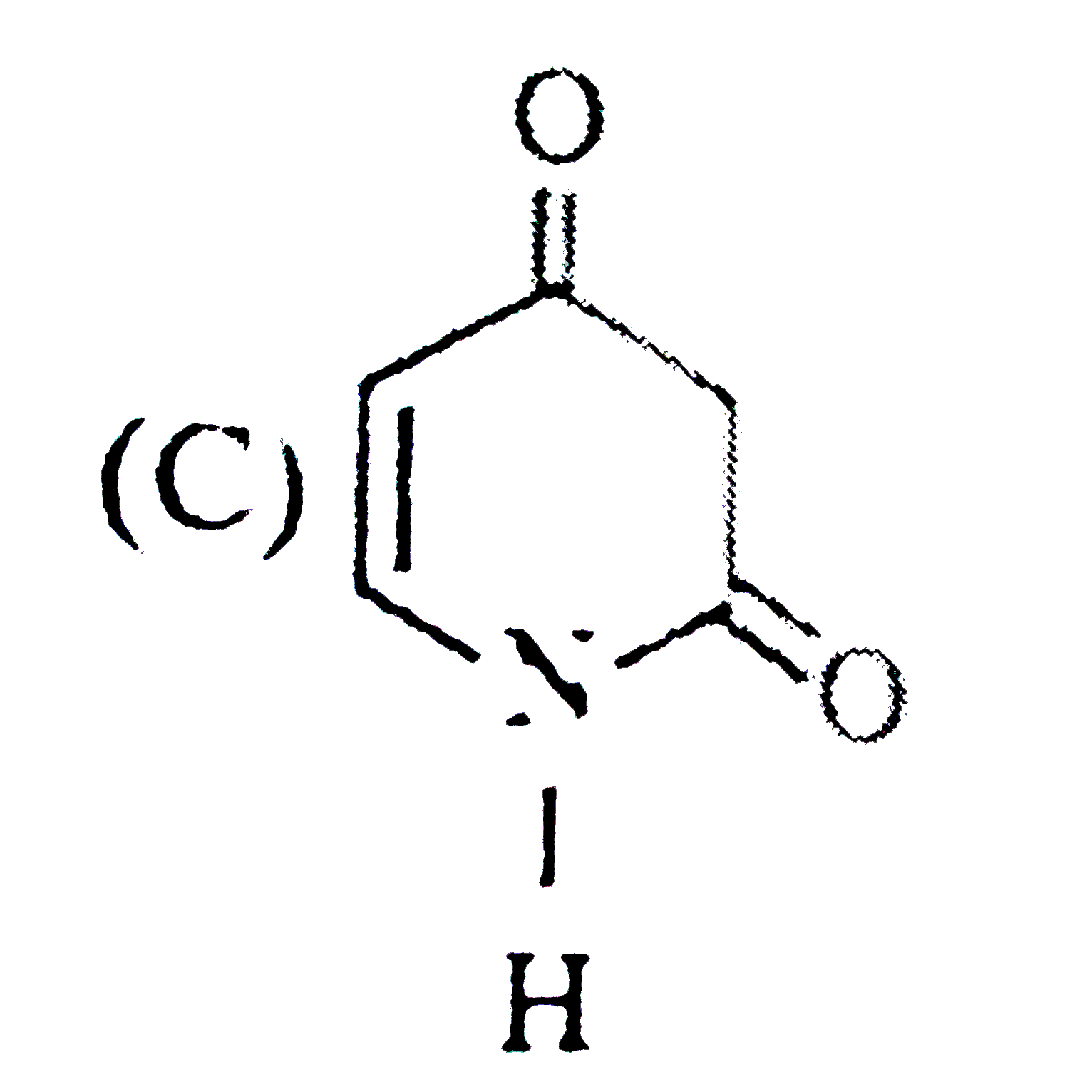Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the pair of compounds which exhibit cis-trans (geometrical) isomerism: |
|
Answer» fumaric ACID and maleic acid |
|
| 2. |
Select the organic compounds which was prepared for the first time in laboratory from in elements: |
|
Answer» UREA |
|
| 3. |
Select the organic compounds which burn with non smoky flame: |
|
Answer» `CHCl_3` |
|
| 4. |
Select the option in which heat evolved is maximum. Given : Delta_(f)H^(@) (CO_(2), g) = -75 k Cal//mol, ""Delta_(f)H^(@) (CO, g) = -25 k Cal//mol The product will be CO if excess amount of carbon is present and CO_(2) if excess O_(2) is present |
|
Answer» 10 moles of CARBON and 4.5 moles of `O_(2)` energy released `=9xx-25=-225` (b) `{:(C,+,1/2 O_(2),rarr,CO_(2),),(2,,2,,0,),(L.R.,,1,,2,DeltaH=-50),(CO+1/2 O_(2) rarrCO_(2),,,,,DeltaH=-100):}` `{:(2,,1,,2):}` Total `DeltaH=-150` (C) `{:(C,+,1/2O_(2),rarr,CO,),(4,,3.5,,0,),(L.R.,,1.5,,4,DeltaH=-25xx4=-100),(,,,,,),(CO,+,1/2O_(2),rarr,CO_(2),),(4,,1.5,,0,),(,,L.R.,,3,DeltaH-3xx50=-150),(,,,,,"Total "DeltaH=-250):}` (D) `{:(C,+,1/2O_(2),rarr,CO,),(2.5,,2.5,,0,DeltaH=-25xx2.5=62.5),(L.R.,,1.25,,2.5,),(,,,,,),(CO,+,1/2O_(2),rarr,CO_(2),),(2.5,,1.25,,0,DeltaH=-2.5xx50=125),(0,,,,2.5,),(,,,,,"total "DeltaH=62.5+125=187.5):}` |
|
| 5. |
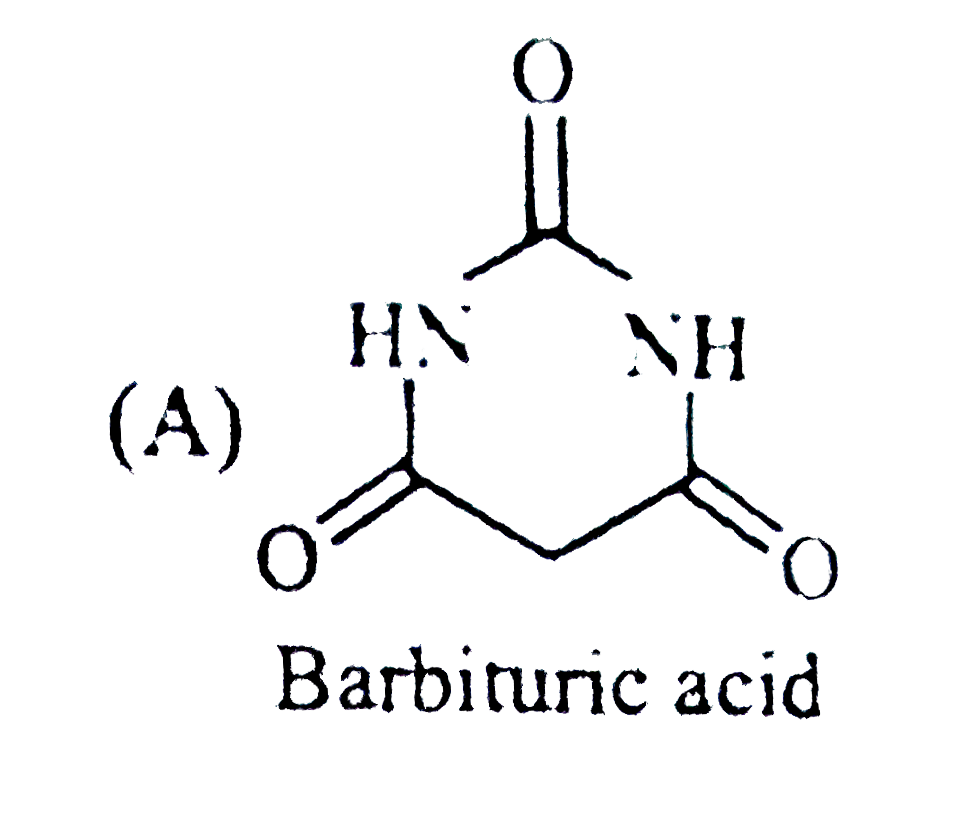
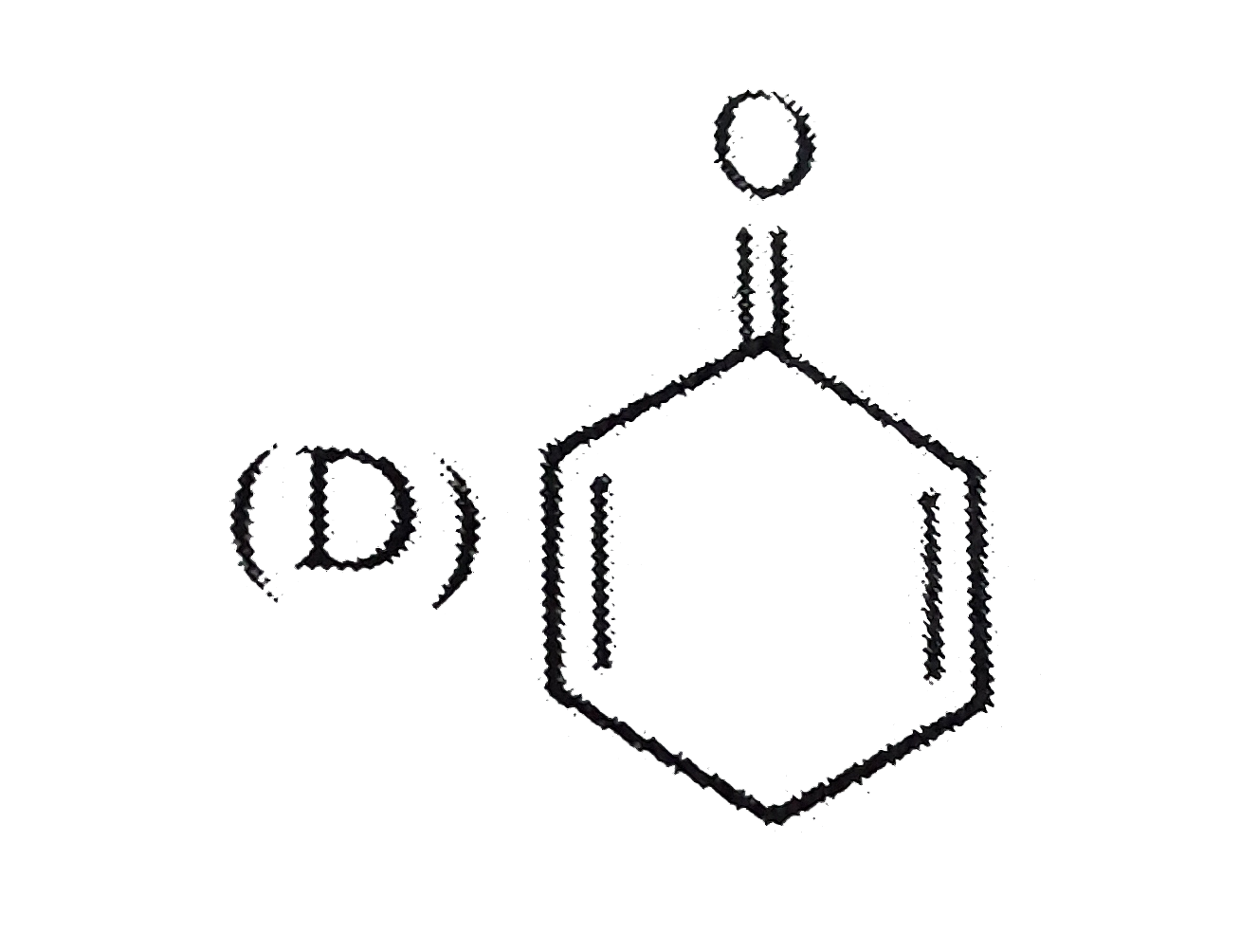
Select the optically inactive compound among the following: |
|
Answer»
|
|
| 6. |
Select the only incorrect option for gold sol. |
|
Answer» <P>(II) (ii) (P) |
|
| 7. |
Select the only incorrect option. |
|
Answer» <P>(III) (i) (P) |
|
| 8. |
Select the only incorrect option for Agl//I^(-) sol. |
|
Answer» (II) (ii) (P) |
|
| 9. |
Select the only incorrect option. |
|
Answer» (III) (II) (Q) |
|
| 10. |
Select the only INCORRECT combination among the following is |
|
Answer» (II) (III) (S) |
|
| 11. |
Select the only correct option Fe(OH)_(3) sol. |
|
Answer» (I) (III) (Q) |
|
| 12. |
Select the only correct option. |
|
Answer» <P>(I) (i) (P) |
|
| 13. |
Select the numbers with same significant figures: |
|
Answer» `6.02xx10^(23)` |
|
| 14. |
Select the number of compounds in which deprotonation gives aromatic anion: |
|
Answer»
|
|
| 15. |
Select the natural rubber from the following. |
| Answer» Solution :Isoprene | |
| 16. |
Select the most suitable alcohol and oxidant to parpare the following compounds. |
Answer» SOLUTION :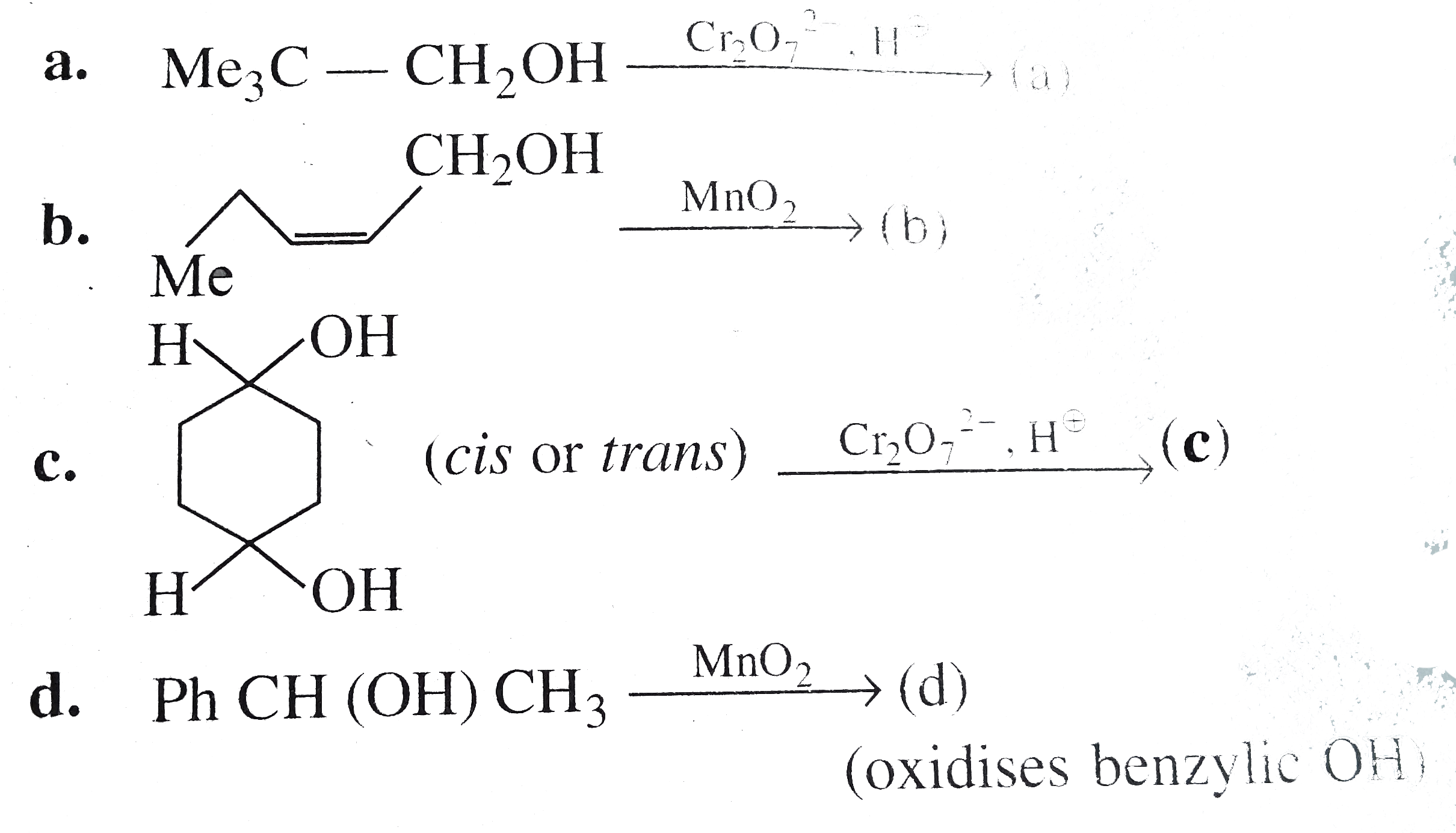
|
|
| 17. |
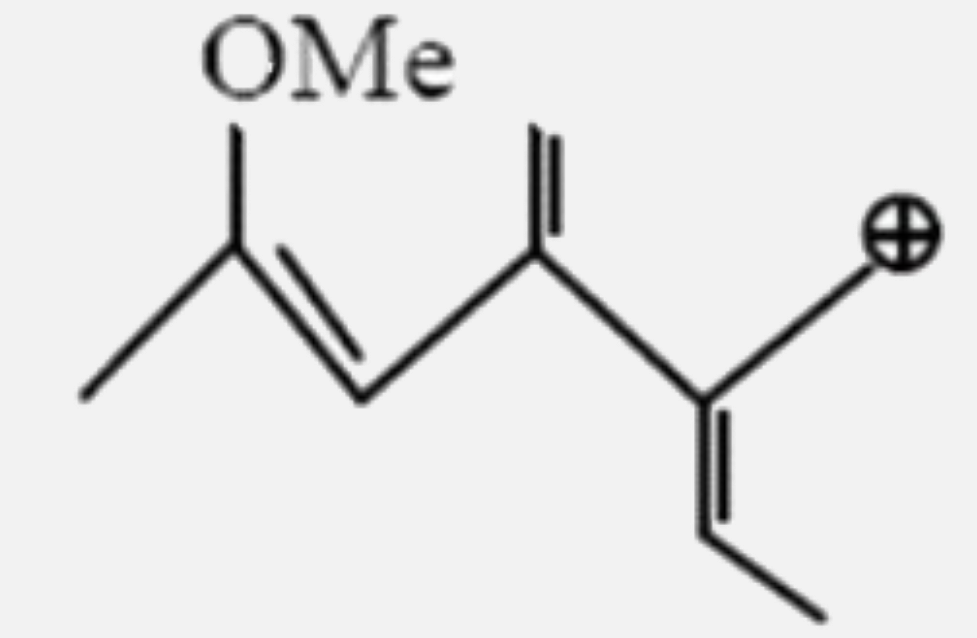
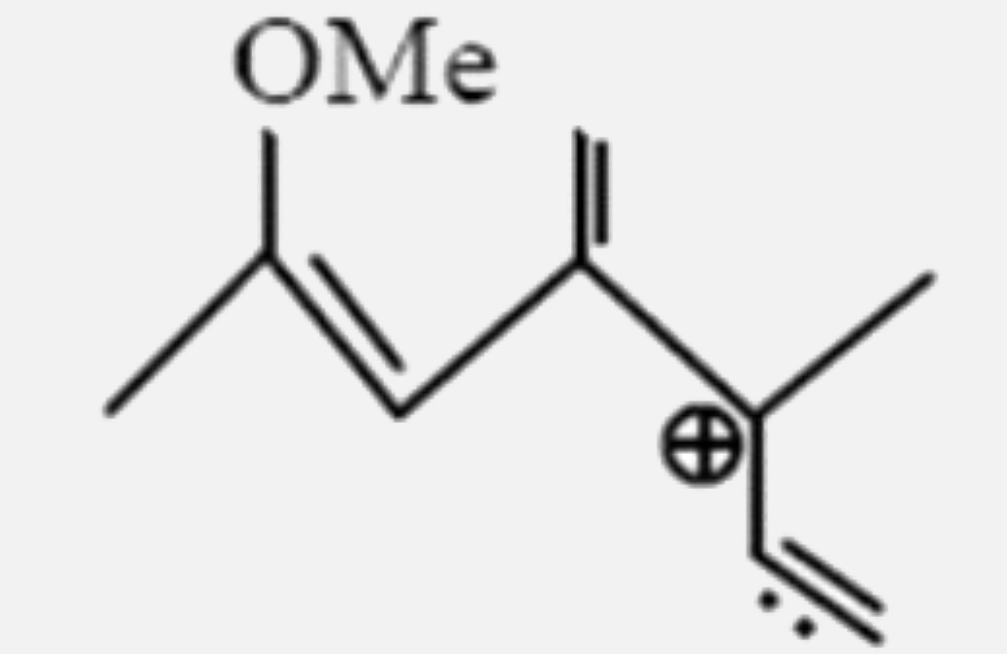
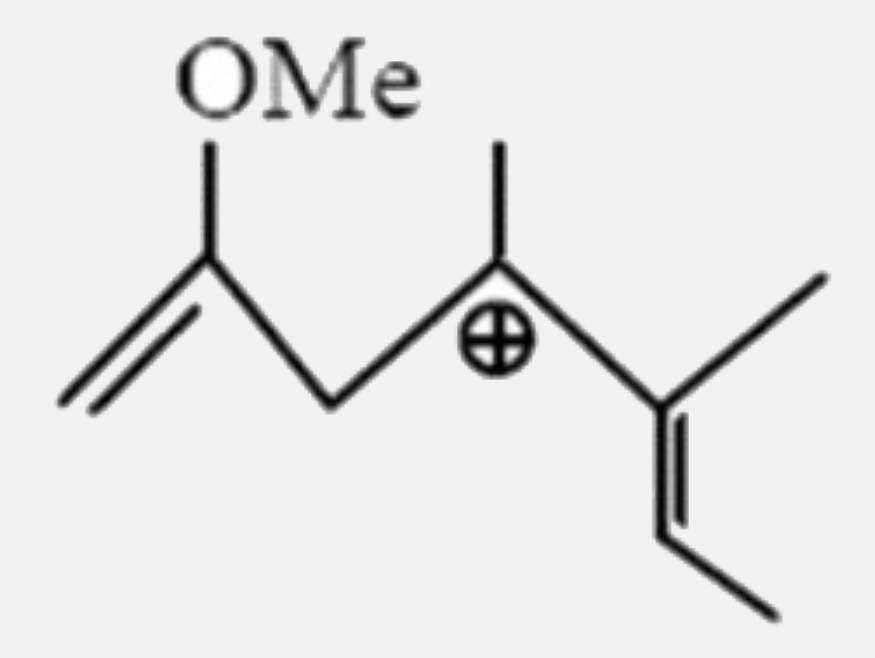
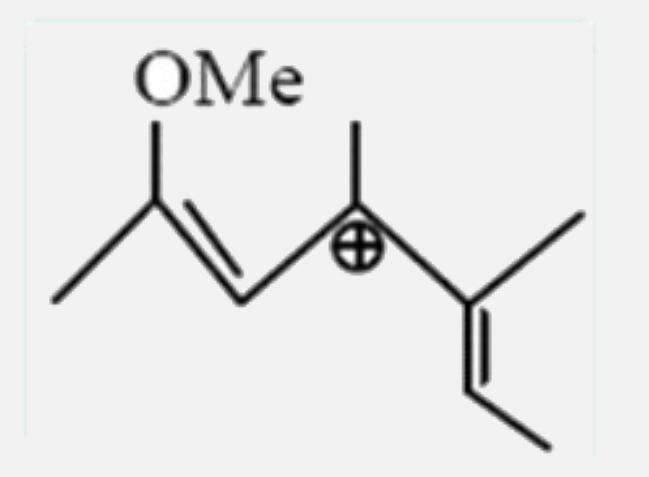
Select the most stable carbocation: |
|
Answer»
|
|
| 18. |
Select the most reactive compound towards the attack of MeMgBr followed by hydrolysis? |
|
Answer» `Me-OVERSET(O)overset(||)C-Me` |
|
| 19. |
Select the most reactive cycloalkane: |
|
Answer» Cyclopropane |
|
| 20. |
Select the molecule with more polar bond from each of the indicated bonds among given set of molecule : (a) H_(3)Cunderset((I))(-)H,(H_(3)C)underset((II))(-)Br (b)H_(3)Cunderset((I))(-)NH_(2),underset((II))(H_(3)C)-OH (c ) H_(3)Cunderset((I))(-)OH,underset((II))(H_(3)C)-SH |
|
Answer» I,I,I |
|
| 21. |
Select the member of each pair that shows faster rate of SN2 reaction with Kl in acetone. CH_(3)-underset((I))CH_(2)-CH_(2)-ClorH_(3)C-underset((II))underset(CH_(3))underset(|)CH-CH_(2)-Cl (B) CH_(3)-underset((I))CH_(2)-CH_(2)-ClorCH_(3)-underset((II))CH_(2)-CH_2-Cl (C) CH_(3)-underset((I))underset(CH_(3))underset(|)CH-CH_(2)-CH_(2)-ClorCH_(3)-underset((II))underset(H_(3)C)underset(|)overset(H_(3)C)overset(|)CH_(2)-CH_(2)Cl CH_(3)-CH_(2)underset((I))-CH_(2)-overset(Br)overset(|)CH_(2)-CH_(3)orCH_(3)-underset(H_(3)C)underset(|)CH-underset((II))CH_(2)-overset(Br)overset(|)CH_(3) |
| Answer» SOLUTION :`(A)TOI,(B)TOII,(C)toI,(D)toI` | |
| 22. |
Select themaor product formed from reaction of 2-bromo butane and lacoholic KOH |
|
Answer» trans `CH_(3)CH=CHCH_(3)` |
|
| 23. |
Select the longest continous chain in each of the following molecules. |
Answer» 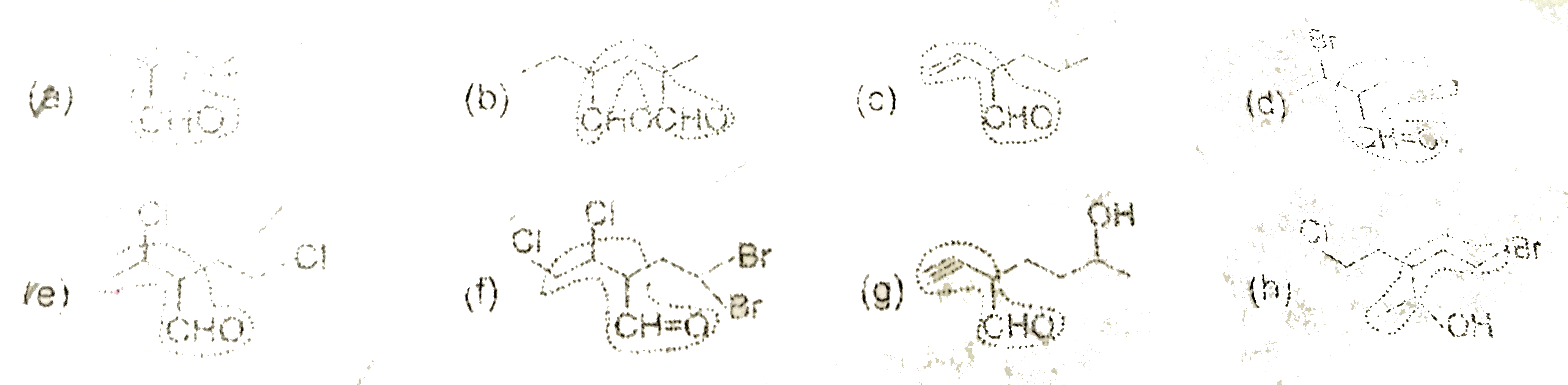
|
|
| 25. |
Select the intermediate in the following reaction mechanism O_(3(g))toO_(2(g))+O_((g))+O_(3(g))to 2O_2 |
|
Answer» `O_(3(G))` |
|
| 26. |
Select the intermediate in the following reaction mechanism:O_3(g)iffO_2(g)+O(g)O(g)+O_3(g)to2O_2 |
|
Answer» `O_3(G)` |
|
| 27. |
Select the incorrectly matched options : |
|
Answer» `R-CO-CH_(3)OVERSET(Zn//Hg)underset(conc.HCl)rarrR-CH_(2)-CH_(3)` (Clemmensen REDUCTION) |
|
| 28. |
Select the incorrect statment: |
|
Answer» The C. N. of cation OCCUPYING a tetrahedral HOLE is 4 |
|
| 29. |
Select the incorrect statemnt. |
|
Answer» `SO_(2)` GAS has oxidisingas well as reducingbehaviour . ` 2HI + H_(2)SO_(4) to2 H_(2)O + SO_(2) + I_(2)` |
|
| 30. |
Select the incorrect statements on the basis of curve given below for real gas(A). |
|
Answer» For 1 mol, `V_(C) = (48)/(3) pi r^(3) XX N_(A)` |
|
| 31. |
Select the incorrect statement(s)/order (s): |
|
Answer» d-orbital can accommodate 10 electrons |
|
| 32. |
Select the incorrect statements from the following: |
|
Answer» The ratio of `SIGMA` bonds to `pi` bonds in `SO_(3)` and `SO_(2)` are same |
|
| 33. |
Select the incorrect statements about the following |
|
Answer» `O_(3)` is USED as germicide for purification of AIR 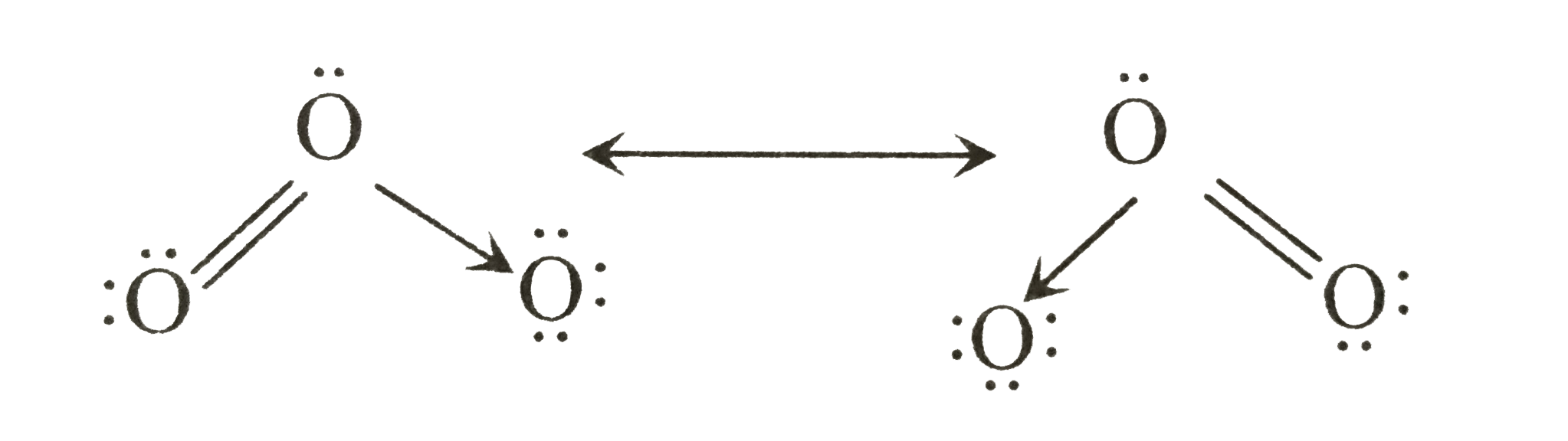
|
|
| 34. |
Select the incorrect statement(s) about C_(2) molecule |
|
Answer» It exists in vapour PHASE It exists in vapour phase and is diamagnetic in nature because it contains no unpaired electrons. Since four electrons are present in TWO `p_(i)`- molecular orbitals so the double bond contains both` pi`- bonds. |
|
| 35. |
Select the incorrect statement(s): |
|
Answer» Magnesium can be burnt in the ATMOSPHERE of `CO_(2) and SO_(2)` |
|
| 36. |
Above which temperature, hydrous calcium sulphate is converted into dead burnt plaster :- |
|
Answer» 313 K |
|
| 37. |
Select the incorrect statements : |
|
Answer» Size of `H^(-)`is larger than `F^(-)` |
|
| 38. |
Select the incorrect statement on basis of following conversion :- underset("Ore")underset("coloured")underset("Black")(X) underset("air")overset(KOH)tounderset("green")underset("Dark")(Y)overset(H^(o+))tounderset("agent")underset("an oxidising")(Z)+X |
|
Answer» X MAY be `MnO_(2)` |
|
| 39. |
Select the incorrect statement ( s) : |
|
Answer» At Boyle's temperature a real gas BEHAVES like an ideal gas IRRESPECTIVE of pressure. |
|
| 40. |
Select the incorrect statement regarding gamma- form of SO_(3) |
|
Answer» SULPHUR has `sp^(3)` hybridisation 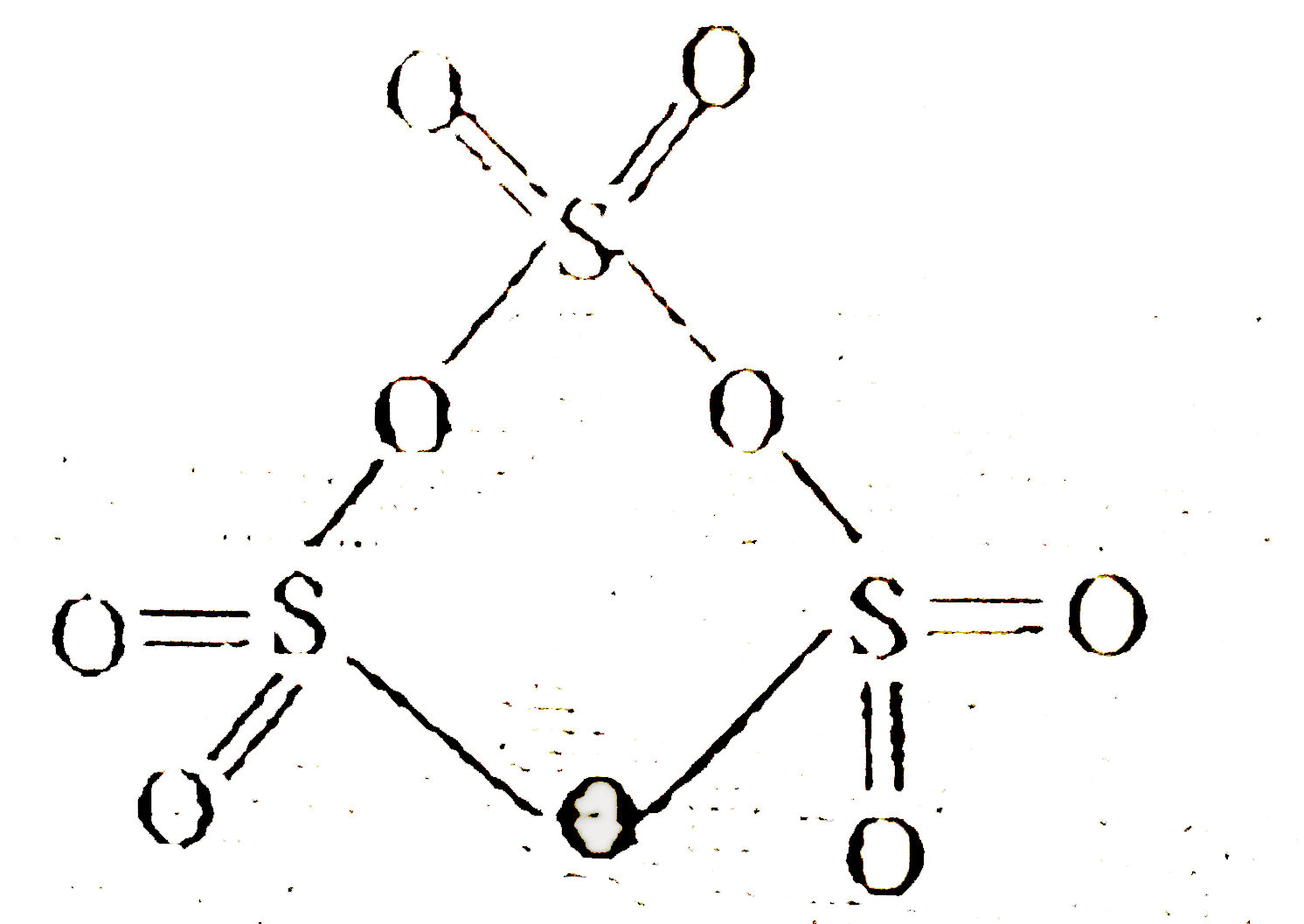 `GAMMA-` forms of `SO_(3)`-TRIMER of `SO_(3)=S_(3)O_(9)` Hybridisation `sp^(3)` Non planar Six `p pi-d pi` bonds are present All S-O bond length are not same. |
|
| 41. |
Select the incorrect statement regarding B_(2)H_(6). |
|
Answer» It CONTAINS B-B IONIC bond |
|
| 42. |
Select the incorrect statement in the gas equation, PV= nRT |
|
Answer» n is the number of molecules of a GAS |
|
| 43. |
Select the incorrect statement for the metallurgy of aluminum: |
|
Answer» Bauxite can be enriched by BAYER's process |
|
| 44. |
Select the incorrect statement amoung the following . |
|
Answer» ZEOLITES are used in petrochemical INDUSTRIES for CRACKING of hydrocarbons |
|
| 45. |
Select the incorrect statement for B_(2)H_(6) |
|
Answer» It CONTAINS B-B ionic bond |
|
| 46. |
Select the incorrect statement , among the following |
|
Answer» Haemoglobin is soluble in water |
|
| 47. |
Select the incorrect statement about the following : |
|
Answer» `O_3` molecular is angular in SHAPE |
|
| 48. |
Select the incorrect statement among the following |
|
Answer» Hemoglobin is soluble in WATER |
|
| 49. |
Select the incorrect statement about the boron. |
|
Answer» Pure FORM of the elements are obtained by the reduction of `BCl_(3)` with zinc at `900^(@)C`. |
|
| 50. |
Select the incorrect statement about nylon 2-nylon-6 : |
|
Answer» It is a copolymer. |
|